Get your patient on Flarex - Fluorometholone Acetate suspension/ Drops (Fluorometholone Acetate)
Flarex - Fluorometholone Acetate suspension/ Drops prescribing information
INDICATIONS AND USAGE
FLAREX (fluorometholone acetate ophthalmic suspension) 0.1% is indicated for use in the treatment of steroid responsive inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the eye.
DOSAGE AND ADMINISTRATION
Shake Well Before Using. One to two drops instilled into the conjunctival sac(s) four times daily. During the initial 24 to 48 hours the dosage may be safely increased to two drops every two hours. If no improvement after two weeks, consult physician. Care should be taken not to discontinue therapy prematurely.
Not more than one bottle should be prescribed initially, and the prescription should not be refilled without further evaluation [see PRECAUTIONS ] .
CONTRAINDICATIONS
Contraindicated in acute superficial herpes simplex keratitis, vaccinia, varicella, and most other viral diseases of cornea and conjunctiva; mycobacterial infection of the eye; fungal diseases; acute purulent untreated infections, which like other diseases caused by microorganisms, may be masked or enhanced by the presence of the steroid; and in those persons who have known hypersensitivity to any component of this preparation.
ADVERSE REACTIONS
Glaucoma with optic nerve damage, visual acuity and field defects, cataract formation, secondary ocular infection following suppression of host response, and perforation of the globe may occur.
Postmarketing Experience
The following reaction has been identified during post-marketing use of FLAREX ® (fluorometholone acetate ophthalmic suspension) 0.1% in clinical practice. Because reactions are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The reaction, which has been chosen for inclusion due to either its seriousness, frequency of reporting, possible causal connection to FLAREX, or a combination of these factors, includes: dysgeusia.
The following rare adverse reactions have been reported: Cushing's syndrome and adrenal suppression may occur after very frequent use of topical ophthalmic corticosteroids, particularly in very young children.
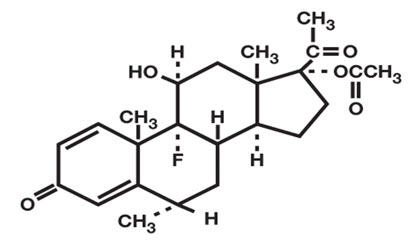
DESCRIPTION
FLAREX ® (fluorometholone acetate ophthalmic suspension) 0.1% is a corticosteroid prepared as a sterile topical ophthalmic suspension. The active ingredient, fluorometholone acetate, is a white to creamy white powder with an empirical formula of C 24 H 31 FO 5 and a molecular weight of 418.5. Its chemical name is 9-fluoro-11β, 17-dihydroxy-6α-methylpregna-1, 4-diene-3, 20-dione 17-acetate. The chemical structure of Fluorometholone Acetate is presented below:
Each mL of FLAREX (fluorometholone acetate ophthalmic suspension) 0.1% contains: Active: fluorometholone acetate 1 mg (0.1%). Preservative: benzalkonium chloride 0.01%. Inactives: sodium chloride, monobasic sodium phosphate, edetate disodium, hydroxyethyl cellulose, tyloxapol, hydrochloric acid and/or sodium hydroxide (to adjust pH), and purified water. The pH of the suspension is approximately 7.3, with an osmolality of approximately 300 mOsm/kg.
CLINICAL PHARMACOLOGY
Corticosteroids suppress the inflammatory response to inciting agents of mechanical, chemical or immunological nature. No generally accepted explanation of this steroid property has been advanced. Corticosteroids cause a rise in intraocular pressure (IOP) in susceptible individuals. In a small study, FLAREX (fluorometholone acetate ophthalmic suspension) 0.1% demonstrated a significantly longer average time to produce a rise in IOP than did dexamethasone phosphate; however, the ultimate magnitude of the rise was equivalent for both drugs and in a small percentage of individuals a significant rise in IOP occurred within three days.
HOW SUPPLIED
FLAREX (fluorometholone acetate ophthalmic suspension) 0.1% is supplied in white low density polyethylene (LDPE) bottles, with natural LDPE dispensing plugs and pink polypropylene closures. The product is supplied as 5 mL in an 8 mL bottle.
| 5 mL: | NDC 82667-010-01 |
STORAGE
Store upright between 2°C to 25°C (36°F to 77°F). After opening, FLAREX can be used until the expiration date on the bottle.
Protect from freezing.