Fluorouracil - Fluorouracil solution prescribing information
INDICATIONS AND USAGE:
Fluorouracil is recommended for the topical treatment of multiple actinic or solar keratoses. In the 5% strength, it is also useful in the treatment of superficial basal cell carcinomas when conventional methods are impractical, such as with multiple lesions or difficult treatment sites. Safety and efficacy in other indications have not been established.
The diagnosis should be established prior to treatment, since this method has not been proven effective in other types of basal cell carcinomas. With isolated, easily accessible basal cell carcinomas, surgery is preferred since success with such lesions is almost 100%. The success rate with fluorouracil solution and a different dosage form is approximately 93%, based on 113 lesions in 54 patients. Twenty-five lesions treated with the solution produced 1 failure and 88 lesions treated with a different dosage form produced 7 failures.
DOSAGE AND ADMINISTRATION:
When fluorouracil topical solution USP, 5% is applied to a lesion, a response occurs with the following sequence: erythema, usually followed by vesiculation, desquamation, erosion, and re-epithelialization. Fluorouracil topical solution USP, 5% should be applied preferably with a nonmetal applicator or suitable glove. If fluorouracil is applied with the fingers, the hands should be washed immediately afterward.
Actinic or Solar Keratosis: Apply solution twice daily in an amount sufficient to cover the lesions. Medication should be continued until the inflammatory response reaches the erosion stage, at which time use of the drug should be terminated. The usual duration of therapy is from 2 to 4 weeks. Complete healing of the lesions may not be evident for 1 to 2 months following cessation of fluorouracil therapy.
Superficial Basal Cell Carcinomas: Only the 5% strength is recommended . Apply solution twice daily in an amount sufficient to cover the lesions. Treatment should be continued for at least 3 to 6 weeks. Therapy may be required for as long as 10 to 12 weeks before the lesions are obliterated. As in any neoplastic condition, the patient should be followed for a reasonable period of time to determine if a cure has been obtained.
CONTRAINDICATIONS:
Fluorouracil may cause fetal harm when administered to a pregnant woman. There are no adequate and well-controlled studies in pregnant women with either the topical or the parenteral forms of fluorouracil. One birth defect (cleft lip and palate) has been reported in the newborn of a patient using fluorouracil as recommended. One birth defect (ventricular septal defect) and cases of miscarriage have been reported when fluorouracil was applied to mucous membrane areas. Multiple birth defects have been reported in a fetus of a patient treated with intravenous fluorouracil. Animal reproduction studies have not been conducted with fluorouracil. Fluorouracil administered parenterally has been shown to be teratogenic in mice, rats, and hamsters when given at doses equivalent to the usual human intravenous dose; however, the amount of fluorouracil absorbed systemically after topical administration to actinic keratoses is minimal (see CLINICAL PHARMACOLOGY ).
Fluorouracil exhibited maximum teratogenicity when given to mice as single intraperitoneal injections of 10 to 40 mg/kg on Day 10 or 12 of gestation. Similarly, intraperitoneal doses of 12 to 37 mg/kg given to rats between Days 9 and 12 of gestation and intramuscular doses of 3 to 9 mg/kg given to hamsters between Days 8 and 11 of gestation were teratogenic and/or embryotoxic (i.e., resulted in increased resorptions or embryolethality). In monkeys, divided doses of 40 mg/kg given between Days 20 and 24 of gestation were not teratogenic. Doses higher than 40 mg/kg resulted in abortion.
Fluorouracil should not be used in patients with dihydropyrimidine dehydrogenase (DPD) enzyme deficiency. A large percentage of fluorouracil is catabolized by the DPD enzyme. DPD enzyme deficiency can result in shunting of fluorouracil to the anabolic pathway, leading to cytotoxic activity and potential toxicities.
Fluorouracil is contraindicated in women who are or may become pregnant during therapy. If this drug is used during pregnancy, or if the patient becomes pregnant while using this drug, the patient should be apprised of the potential hazard to the fetus.
Fluorouracil is also contraindicated in patients with known hypersensitivity to any of its components.
ADVERSE REACTIONS:
The most frequent adverse reactions to fluorouracil occur locally and are often related to an extension of the pharmacological activity of the drug. These include burning, crusting, allergic contact dermatitis, pruritus, scarring, rash, soreness and ulceration. Ulcerations, other local reactions, cases of miscarriage, and a birth defect (ventricular septal defect) have been reported when fluorouracil topical solution was applied to mucous membrane areas. Leukocytosis is the most frequent hematological side effect. Although a causal relationship is remote, other adverse reactions which have been reported infrequently are: Central Nervous System: Emotional upset, insomnia, irritability. Gastrointestinal: Medicinal taste, stomatitis. Hematological: Eosinophilia, thrombocytopenia, toxic granulation. Integumentary: Alopecia, blistering, bullous pemphigoid, discomfort, ichthyosis, scaling, suppuration, swelling, telangiectasia, tenderness, urticaria, skin rash. Special Senses: Conjunctival reaction, corneal reaction, lacrimation, nasal irritation. Miscellaneous: Herpes simplex. To report SUSPECTED ADVERSE REACTIONS, contact Encube Ethicals Private Limited at 1-833-285-4151 and/or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION:
Fluorouracil topical solution is a topical preparation containing the fluorinated pyrimidine 5- fluorouracil, an antineoplastic antimetabolite.
Fluorouracil Topical Solution USP, 5% consists of 5% fluorouracil on a weight/weight basis, compounded with edetate disodium, hydroxypropyl cellulose, methylparaben, propylparaben, propylene glycol, purified water and tromethamine.
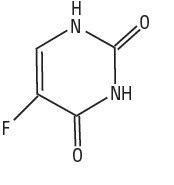
Chemically, fluorouracil is a 5-fluoro-2,4(1H,3H)-pyrimidinedione. It is a white to practically white, crystalline powder which is sparingly soluble in water and slightly soluble in alcohol. One gram of fluorouracil is soluble in 100 mL of propylene glycol. The molecular weight of 5-fluorouracil is 130.08 and the structural formula is:
CLINICAL PHARMACOLOGY:
There is evidence that the metabolism of fluorouracil in the anabolic pathway blocks the methylation reaction of deoxyuridylic acid to thymidylic acid. In this manner, fluorouracil interferes with the synthesis of deoxyribonucleic acid (DNA) and to a lesser extent inhibits the formation of ribonucleic acid (RNA). Since DNA and RNA are essential for cell division and growth, the effect of fluorouracil may be to create a thymine deficiency which provokes unbalanced growth and death of the cell. The effects of DNA and RNA deprivation are most marked on those cells which grow more rapidly and take up fluorouracil at a more rapid rate. The catabolic metabolism of fluorouracil results in degradation products (e.g., CO 2 , urea, α-fluoro-β-alanine) which are inactive.
Systemic absorption studies of topically applied fluorouracil have been performed on patients with actinic keratoses using tracer amounts of 14 C-labeled fluorouracil added to a 5% preparation. All patients had been receiving nonlabeled fluorouracil until the peak of the inflammatory reaction occurred (2 to 3 weeks), ensuring that the time of maximum absorption was used for measurement. One gram of labeled preparation was applied to the entire face and neck and left in place for 12 hours. Urine samples were collected. At the end of 3 days, the total recovery ranged between 0.48% and 0.94% with an average of 0.76%, indicating that approximately 5.98% of the topical dose was absorbed systemically. If applied twice daily, this would indicate systemic absorption of topical fluorouracil to be in the range of 5 to 6 mg per daily dose of 100 mg. In an additional study, negligible amounts of labeled material were found in plasma, urine, and expired CO 2 after 3 days of treatment with topically applied 14 C- labeled fluorouracil.
HOW SUPPLIED:
Fluorouracil Topical Solution USP, 5% is available in 10 mL drop dispensers containing 5% (NDC 21922-041-12) fluorouracil on a weight/weight basis compounded with edetate disodium, hydroxypropyl cellulose, methylparaben, propylparaben, propylene glycol, purified water and tromethamine. Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F).
Manufactured by: Encube Ethicals Pvt. Ltd. Plot No. C-1, Madkaim Industrial Estate, Madkaim, Post: Mardol, Ponda, Goa-403 404, India.
Distributed by: Encube Ethicals, Inc.
200 Meredith Drive, Suite 202, Durham, NC 27713 USA
Rev. 11/2023