Get your patient on Flurbiprofen Sodium - Flurbiprofen Sodium solution/ Drops (Flurbiprofen Sodium)
Flurbiprofen Sodium - Flurbiprofen Sodium solution/ Drops prescribing information
INDICATIONS AND USAGE
Flurbiprofen sodium ophthalmic solution is indicated for the inhibition of intraoperative miosis.
DOSAGE AND ADMINISTRATION
A total of four (4) drops of flurbiprofen sodium ophthalmic solution should be administered by instilling one (1) drop approximately every 1/2 hour beginning 2 hours before surgery.
CONTRAINDICATIONS
Flurbiprofen sodium ophthalmic solution is contraindicated in individuals who are hypersensitive to any components of the medication.
ADVERSE REACTIONS
Transient burning and stinging upon instillation and other minor symptoms of ocular irritation have been reported with the use of flurbiprofen sodium ophthalmic solution. Other adverse reactions reported with the use of flurbiprofen sodium ophthalmic solution include: fibrosis, hyphema, miosis, mydriasis, and ocular hyperemia.
Increased bleeding tendency of ocular tissues in conjunction with ocular surgery has also been reported (see Warnings ).
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Interaction of flurbiprofen sodium ophthalmic solution with other topical ophthalmic medications has not been fully investigated.
Although clinical studies with acetylcholine chloride and animal studies with acetylcholine chloride or carbachol revealed no interference, and there is no known pharmacological basis for an interaction, there have been reports that acetylcholine chloride and carbachol have been ineffective when used in patients treated with flurbiprofen sodium ophthalmic solution.
DESCRIPTION
Flurbiprofen sodium ophthalmic solution, USP 0.03% is a sterile topical nonsteroidal anti-inflammatory product for ophthalmic use.
Chemical Name: Sodium (±)-2-(2-fluoro-4-biphenylyl)propionate dihydrate.
Structural Formula:
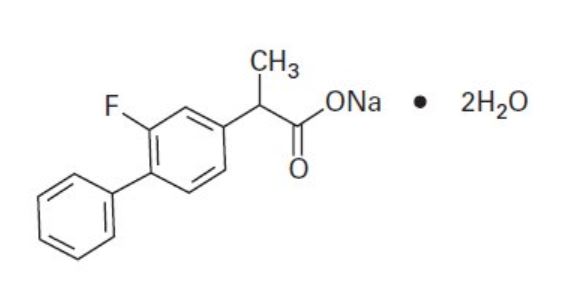
C 15 H 12 FNaO 2 •2H 2 O Mol. Wt. 302.27
Each mL contains:
Active: flurbiprofen sodium 0.03%.
Inactives: citric acid, edetate disodium, polyvinyl alcohol 1.4%, potassium chloride, purified water, sodium chloride, sodium citrate. hydrochloric acid and/or sodium hydroxide may be added to adjust pH (6.0 – 7.0).
Preservative: thimerosal 0.005%.
CLINICAL PHARMACOLOGY
Flurbiprofen sodium is one of a series of phenylalkanoic acids that have shown analgesic, antipyretic, and anti-inflammatory activity in animal inflammatory diseases. Its mechanism of action is believed to be through inhibition of the cyclo-oxygenase enzyme that is essential in the biosynthesis of prostaglandins.
Prostaglandins have been shown in many animal models to be mediators of certain kinds of intraocular inflammation. In studies performed on animal eyes, prostaglandins have been shown to produce disruption of the blood-aqueous humor barrier, vasodilatation, increased vascular permeability, leukocytosis, and increased intraocular pressure.
Prostaglandins also appear to play a role in the miotic response produced during ocular surgery by constricting the iris sphincter independently of cholinergic mechanisms. In clinical studies, flurbiprofen sodium ophthalmic solution has been shown to inhibit the miosis induced during the course of cataract surgery.
Results from clinical studies indicate that flurbiprofen sodium has no significant effect upon intraocular pressure.
HOW SUPPLIED
Flurbiprofen sodium ophthalmic solution USP, 0.03% is supplied in a plastic bottle with a controlled drop tip in the following size:
2.5 mL - NDC 69292-722-25
DO NOT USE IF IMPRINTED “Protective Seal” WITH YELLOW  IS NOT INTACT.
IS NOT INTACT.