Get your patient on Foscavir - Foscarnet Sodium injection, Solution (Foscarnet Sodium)
Foscavir - Foscarnet Sodium injection, Solution prescribing information
WARNING
RENAL IMPAIRMENT IS THE MAJOR TOXICITY OF FOSCAVIR. FREQUENT MONITORING OF SERUM CREATININE, WITH DOSE ADJUSTMENT FOR CHANGES IN RENAL FUNCTION, AND ADEQUATE HYDRATION WITH ADMINISTRATION OF FOSCAVIR IS IMPERATIVE. (See ADMINISTRATION section; Hydration. )
SEIZURES, RELATED TO ALTERATIONS IN PLASMA MINERALS AND ELECTROLYTES, HAVE BEEN ASSOCIATED WITH FOSCAVIR TREATMENT. THEREFORE, PATIENTS MUST BE CAREFULLY MONITORED FOR SUCH CHANGES AND THEIR POTENTIAL SEQUELAE. MINERAL AND ELECTROLYTE SUPPLEMENTATION MAY BE REQUIRED.
FOSCAVIR IS INDICATED FOR USE ONLY IN IMMUNOCOMPROMISED PATIENTS WITH CMV RETINITIS AND MUCOCUTANEOUS ACYCLOVIR-RESISTANT HSV INFECTIONS. (See INDICATIONS section).
INDICATIONS
CMV Retinitis
FOSCAVIR is indicated for the treatment of CMV retinitis in patients with acquired immunodeficiency syndrome (AIDS). Combination therapy with FOSCAVIR and ganciclovir is indicated for patients who have relapsed after monotherapy with either drug. SAFETY AND EFFICACY OF FOSCAVIR HAVE NOT BEEN ESTABLISHED FOR TREATMENT OF OTHER CMV INFECTIONS (e.g., PNEUMONITIS, GASTROENTERITIS); CONGENITAL OR NEONATAL CMV DISEASE; OR NONIMMUNOCOMPROMISED INDIVIDUALS.
Mucocutaneous Acyclovir Resistant HSV Infections
FOSCAVIR is indicated for the treatment of acyclovir-resistant mucocutaneous HSV infections in immunocompromised patients. SAFETY AND EFFICACY OF FOSCAVIR HAVE NOT BEEN ESTABLISHED FOR TREATMENT OF OTHER HSV INFECTIONS (e.g., RETINITIS, ENCEPHALITIS); CONGENITAL OR NEONATAL HSV DISEASE; OR HSV IN NONIMMUNOCOMPROMISED INDIVIDUALS.
DOSAGE AND ADMINISTRATION
CAUTION - DO NOT ADMINISTER FOSCAVIR BY RAPID OR BOLUS INTRAVENOUS INJECTION. THE TOXICITY OF FOSCAVIR MAY BE INCREASED AS A RESULT OF EXCESSIVE PLASMA LEVELS. CARE SHOULD BE TAKEN TO AVOID UNINTENTIONAL OVERDOSE BY CAREFULLY CONTROLLING THE RATE OF INFUSION. THEREFORE, AN INFUSION PUMP MUST BE USED. IN SPITE OF THE USE OF AN INFUSION PUMP, OVERDOSES HAVE OCCURRED.
CONTRAINDICATIONS
FOSCAVIR is contraindicated in patients with clinically significant hypersensitivity to foscarnet sodium.
ADVERSE REACTIONS
THE MAJOR TOXICITY OF FOSCAVIR IS RENAL IMPAIRMENT (see WARNINGS section). Approximately 33% of 189 patients with AIDS and CMV retinitis who received FOSCAVIR (60 mg/kg TID), without adequate hydration, developed significant impairment of renal function (serum creatinine ≥ 2.0 mg/dL). The incidence of renal impairment in subsequent clinical trials in which 1000 mL of normal saline or 5% dextrose solution was given with each infusion of FOSCAVIR was 12% (34/280).
FOSCAVIR has been associated with changes in serum electrolytes including hypocalcemia (15-30%), hypophosphatemia (8–26%) and hyperphosphatemia (6%), hypomagnesemia (15–30%), and hypokalemia (16–48%) (see WARNINGS section). The higher percentages were derived from those patients receiving hydration.
FOSCAVIR treatment was associated with seizures in 18/189 (10%) AIDS patients in the initial five controlled studies (see WARNINGS section). Risk factors associated with seizures included impaired baseline renal function, low total serum calcium, and underlying CNS conditions predisposing the patient to seizures. The rate of seizures did not increase with duration of treatment. Three cases were associated with overdoses of FOSCAVIR (see OVERDOSAGE section).
In five controlled U.S. clinical trials the most frequently reported adverse events in patients with AIDS and CMV retinitis are shown in Table 9 . These figures were calculated without reference to drug relationship or severity.
| n = 189 | n = 189 | ||
| Fever | 65% | Abnormal Renal Function | 27% |
| Nausea | 47% | Vomiting | 26% |
| Anemia | 33% | Headache | 26% |
| Diarrhea | 30% | Seizures | 10% |
From the same controlled studies, adverse events categorized by investigator as “severe” are shown in Table 10 . Although death was specifically attributed to FOSCAVIR in only one case, other complications of FOSCAVIR (i.e., renal impairment, electrolyte abnormalities, and seizures) may have contributed to patient deaths (see WARNINGS section).
| n = 189 | |
| Death | 14% |
| Abnormal Renal Function | 14% |
| Marrow Suppression | 10% |
| Anemia | 9% |
| Seizures | 7% |
From the five initial U.S. controlled trials of FOSCAVIR, the following list of adverse events has been compiled regardless of causal relationship to FOSCAVIR. Evaluation of these reports was difficult because of the diverse manifestations of the underlying disease and because most patients received numerous concomitant medications.
Incidence of 5% or Greater
Body as a Whole: fever, fatigue, rigors, asthenia, malaise, pain, infection, sepsis, death
Central and Peripheral Nervous System: headache, paresthesia, dizziness, involuntary muscle contractions, hypoesthesia, neuropathy, seizures including grand mal seizures (see WARNINGS )
Gastrointestinal System: anorexia, nausea, diarrhea, vomiting, abdominal pain
Hematologic: anemia, granulocytopenia, leukopenia, neutropenia (see PRECAUTIONS )
Metabolic and Nutritional: mineral and electrolyte imbalances (see WARNINGS ) including hypokalemia, hypocalcemia, hypomagnesemia, hypophosphatemia, hyperphosphatemia
Psychiatric: depression, confusion, anxiety
Respiratory System: coughing, dyspnea
Skin and Appendages: rash, increased sweating
Urinary System: alterations in renal function including increased serum creatinine, decreased creatinine clearance, and abnormal renal function (see WARNINGS )
Special Senses: vision abnormalities
Incidence between 1% and 5%
Application Site: injection site pain, injection site inflammation
Body as a Whole: back pain, chest pain (including reports of transient chest pain as part of infusion reactions), edema, influenza-like symptoms, bacterial infections, moniliasis, fungal infections, abscess
Cardiovascular: hypertension, palpitations, ECG abnormalities including sinus tachycardia, first degree AV block and non-specific ST-T segment changes, hypotension, flushing, cerebrovascular disorder (see WARNINGS )
Central and Peripheral Nervous System: tremor, ataxia, dementia, stupor, generalized spasms, sensory disturbances, meningitis, aphasia, abnormal coordination, leg cramps, EEG abnormalities (see WARNINGS )
Gastrointestinal: constipation, dysphagia, dyspepsia, rectal hemorrhage, dry mouth, melena, flatulence, ulcerative stomatitis, pancreatitis
Hematologic: thrombocytopenia, platelet abnormalities, thrombosis, white blood cell abnormalities, lymphadenopathy
Liver and Biliary: abnormal A-G ratio, abnormal hepatic function, increased SGPT, increased SGOT
Metabolic and Nutritional: hyponatremia, decreased weight, increased alkaline phosphatase, increased LDH, increased BUN, acidosis, cachexia, thirst
Musculo-Skeletal: arthralgia, myalgia
Neoplasms: lymphoma-like disorder, sarcoma
Psychiatric: insomnia, somnolence, nervousness, amnesia, agitation, aggressive reaction, hallucination
Respiratory System: pneumonia, sinusitis, pharyngitis, rhinitis, respiratory disorders, respiratory insufficiency, pulmonary infiltration, stridor, pneumothorax, hemoptysis, bronchospasm
Skin and Appendages: pruritus, skin ulceration, seborrhea, erythematous rash, maculo-papular rash, skin discoloration
Special Senses: taste perversions, eye abnormalities, eye pain, conjunctivitis
Urinary System: albuminuria, dysuria, polyuria, urethral disorder, urinary retention, urinary tract infections, acute renal failure, nocturia, facial edema
Selected adverse events occurring at a rate of less than 1% in the five initial U.S. controlled clinical trials of FOSCAVIR include: syndrome of inappropriate antidiuretic hormone secretion, pancytopenia, hematuria, dehydration, hypoproteinemia, increases in amylase and creatinine phosphokinase, cardiac arrest, coma, and other cardiovascular and neurologic complications.
Selected adverse event data from the Foscarnet vs. Ganciclovir CMV Retinitis Trial (FGCRT), performed by the Studies of the Ocular Complications of AIDS (SOCA) Research Group, are shown in Table 11 (see CLINICAL TRIALS section).
• Values for the treatment groups refer only to patients who completed at least one follow-up visit – i.e., 133 to 119 patients in the ganciclovir group and 93 to 100 in the foscarnet group. “Events” denotes all events observed and “patients” the number of patients with one or more of the indicated events. | ||||||
†Per person-year at risk | ||||||
‡Final frozen SOCA I database dated October 1991 | ||||||
| EVENT | GANCICLOVIR | FOSCARNET | ||||
| No. of Events | No. of Patients | Rates† | No. of Events | No. of Patients | Rates† | |
| Absolute neutrophil count decreasing to <0.50 x 10 9 per liter | 63 | 41 | 1.30 | 31 | 17 | 0.72 |
| Serum creatinine increasing to >260 μmol per liter (>2.9 mg/dL) | 6 | 4 | 0.12 | 13 | 9 | 0.30 |
| Seizure ‡ | 21 | 13 | 0.37 | 19 | 13 | 0.37 |
| Catheterization-related infection | 49 | 27 | 1.26 | 51 | 28 | 1.46 |
| Hospitalization | 209 | 91 | 4.74 | 202 | 75 | 5.03 |
Selected adverse events from ACTG Study 228 (CRRT) comparing combination therapy with FOSCAVIR or ganciclovir monotherapy are shown in Table 12 . The most common reason for a treatment change in patients assigned to either FOSCAVIR or ganciclovir was retinitis progression. The most frequent reason for a treatment change in the combination treatment group was toxicity.
• Pts. = patients with event; †Rate = events/person/year; ‡ANC = absolute neutrophil count | |||||||||
| Foscavir N=88 | Ganciclovir N=93 | Combination N=93 | |||||||
| No. Events | No. Pts.• | Rate† | No. Events | No. Pts.• | Rate† | No. Events | No. Pts.• | Rate† | |
| Anemia (Hgb <70g/L) | 11 | 7 | 0.20 | 9 | 7 | 0.14 | 19 | 15 | 0.33 |
| Neutropenia‡ ANC <0.75 x 10 9 cells/L ANC <0.50 x 10 9 cells/L | 86 50 | 32 25 | 1.53 0.91 | 95 49 | 41 28 | 1.51 0.80 | 107 50 | 51 28 | 1.91 0.85 |
| Thrombocytopenia Platelets <50 x 10 9 /L Platelets <20 x 10 9 /L | 28 1 | 14 1 | 0.50 0.01 | 19 6 | 8 2 | 0.43 0.05 | 40 7 | 15 6 | 0.56 0.18 |
| Nephrotoxicity Creatinine >260 μmol/L (>2.9 mg/dL) | 9 | 7 | 0.15 | 10 | 7 | 0.17 | 11 | 10 | 0.20 |
| Seizures | 6 | 6 | 0.17 | 7 | 6 | 0.15 | 10 | 5 | 0.18 |
| Hospitalizations | 86 | 53 | 1.86 | 111 | 59 | 2.36 | 118 | 64 | 2.36 |
Adverse events that have been reported in post-marketing surveillance include: administration site extravasation, localized edema, hypersensitivity reactions (including anaphylactic shock, urticaria and angioedema) (see WARNINGS section), gastrointestinal hemorrhage, increased lipase, glomerulonephritis, nephrotic syndrome, proteinuria, status epilepticus, ventricular arrhythmia, prolongation of QT interval, torsade de pointes (see WARNINGS section), gamma GT increased, diabetes insipidus (usually nephrogenic), renal calculus, Fanconi syndrome acquired, renal tubular acidosis, renal tubular necrosis, crystal-induced nephropathy, hypercalcemia, hypernatremia, esophageal ulceration and muscle disorders including myopathy, myositis, muscle weakness and rare cases of rhabdomyolysis. Cases of vesiculobullous eruptions including erythema multiforme, toxic epidermal necrolysis, and Stevens-Johnson syndrome have been reported. In most cases, patients were taking other medications that have been associated with toxic epidermal necrolysis or Stevens-Johnson syndrome.
Drug Interactions
A possible drug interaction of FOSCAVIR and intravenous pentamidine has been described. Concomitant treatment of four patients in the United Kingdom with FOSCAVIR and intravenous pentamidine may have caused hypocalcemia; one patient died with severe hypocalcemia. Toxicity associated with concomitant use of aerosolized pentamidine has not been reported. Because FOSCAVIR can reduce serum levels of ionized calcium, extreme caution is advised when used concurrently with other drugs known to influence serum calcium levels (e.g., intravenous pentamidine). Renal impairment and symptomatic hypocalcemia have been observed during concurrent treatment with FOSCAVIR and intravenous pentamidine.
Because of foscarnet's tendency to cause renal impairment, the use of FOSCAVIR should be avoided in combination with potentially nephrotoxic drugs such as aminoglycosides, amphotericin B, cyclosporine, acyclovir, methotrexate, tacrolimus and intravenous pentamidine (see above) unless the potential benefits outweigh the risks to the patient.
When diuretics are indicated, thiazides are recommended over loop diuretics because the latter inhibit renal tubular secretion, and may impair elimination of FOSCAVIR, potentially leading to toxicity.
Abnormal renal function has been observed in clinical practice during the use of FOSCAVIR and ritonavir, or FOSCAVIR, ritonavir, and saquinavir. (See DOSAGE and ADMINISTRATION. )
Because of the risk of QT prolongation and the potential for torsades de pointes, the use of FOSCAVIR should be avoided in combination with agents known to prolong the QT interval including Class IA (e.g., quinidine or procainamide) or Class III (e.g., dofetilide, amiodarone, sotalol) antiarrhythmic agents, phenothiazines, tricyclic antidepressants, and certain macrolides and fluoroquinolones.
DESCRIPTION
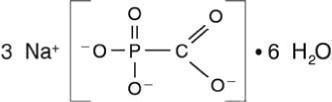
FOSCAVIR is the brand name for foscarnet sodium. The chemical name of foscarnet sodium is phosphonoformic acid, trisodium salt. Foscarnet sodium is a white, crystalline powder containing 6 equivalents of water of hydration with an empirical formula of Na 3 CO 5 P• 6 H 2 O and a molecular weight of 300.1. The structural formula is:
FOSCAVIR has the potential to chelate divalent metal ions, such as calcium and magnesium, to form stable coordination compounds. FOSCAVIR INJECTION is a sterile, isotonic aqueous solution for intravenous administration only. The solution is clear and colorless. Each milliliter of FOSCAVIR contains 24 mg of foscarnet sodium hexahydrate in Water for Injection, USP. Hydrochloric acid may have been added to adjust the pH of the solution to 7.4. FOSCAVIR INJECTION contains no preservatives.
CLINICAL PHARMACOLOGY
Pharmacokinetics
The pharmacokinetics of foscarnet has been determined after administration as an intermittent intravenous infusion during induction therapy in AIDS patients with CMV retinitis. Observed plasma foscarnet concentrations in four studies (FOS-01, ACTG-015, FP48PK, FP49PK) are summarized in Table 7 :
•Values expressed as mean S.D. (number of subjects studied) for each parameter | ||
†50 mg/kg Q8h for 28 days, samples taken 3 hrs after end of 1 hr infusion (Astra Report 815-04 AC025-1) | ||
‡90 mg/kg Q12hr for 28 days, samples taken 1 hr after end of 2 hr infusion (Hengge et al., 1993) | ||
| Parameter | 60 mg/kg Q8h | 90 mg/kg Q12h |
| C max at steady-state (μM) | 589 ± 192 (24) | 623 ± 132 (19) |
| C trough at steady-state (μM) | 114 ± 91 (24) | 63 ± 57 (17) |
| Volume of distribution (L/kg) | 0.41 ± 0.13 (12) | 0.52 ± 0.20 (18) |
| Plasma half-life (hr) | 4.0 ± 2.0 (24) | 3.3 ± 1.4 (18) |
| Systemic clearance (L/hr) | 6.2 ± 2.1 (24) | 7.1 ± 2.7 (18) |
| Renal clearance (L/hr) | 5.6 ± 1.9 (5) | 6.4 ± 2.5 (13) |
| CSF: plasma ratio | 0.69 ± 0.19 (9) † | 0.66 ± 0.11 (5) ‡ |
Distribution
In vitro studies have shown that 14 – 17% of foscarnet is protein bound at plasma drug concentrations of 1 – 1000 μM.
The foscarnet terminal half-life determined by urinary excretion was 87.5 ± 41.8 hours, possibly due to release of foscarnet from bone. Postmortem data on several patients in European clinical trials provide evidence that foscarnet does accumulate in bone in humans; however, the extent to which this occurs has not been determined.
Special Populations
Adults with Impaired Renal Function: The pharmacokinetic properties of foscarnet have been determined in a small group of adult subjects with normal and impaired renal function, as summarized in Table 8 :
•Group 1 patients had normal renal function defined as a creatinine clearance (CrCl) of >80 mL/min, Group 2 CrCl was 50 – 80 mL/min, Group 3 CrCl was 25 – 49 mL/min and Group 4 CrCl was 10 – 24 mL/min. | ||||
| Parameter | Group 1 (N=6) | Group 2 (N=6) | Group 3 (N=6) | Group 4 (N=4) |
| Creatinine clearance (mL/min) | 108 ± 16 | 68 ± 8 | 34 ± 9 | 20 ± 4 |
| Foscarnet CL (mL/min/kg) | 2.13 ± 0.71 | 1.33 ± 0.43 | 0.46 ± 0.14 | 0.43 ± 0.26 |
| Foscarnet half-life (hr) | 1.93 ± 0.12 | 3.35 ± 0.87 | 13.0 ± 4.05 | 25.3 ± 18.7 |
Total systemic clearance (CL) of foscarnet decreased and half-life increased with diminishing renal function (as expressed by creatinine clearance). Based on these observations, it is necessary to modify the dosage of foscarnet in patients with renal impairment (see DOSAGE AND ADMINISTRATION ).
Drug Interaction
The pharmacokinetics of foscarnet and ganciclovir were not altered in 13 patients receiving either concomitant therapy or daily alternating therapy for maintenance of CMV disease.
There is no clinically significant interaction with zidovudine (AZT), or probenecid.
HOW SUPPLIED
FOSCAVIR (foscarnet sodium) INJECTION, 24 mg/mL for intravenous infusion, is supplied in 250 mL glass bottles or polypropylene based infusion bags containing 6000 mg foscarnet sodium (24 mg/mL) as follows:
NDC 76310-024-15 250 mL bottle NDC 76310-024-25 250 mL bottles, cases of 10 NDC 76310-024-41 250 mL infusion bag NDC 76310-024-45 250 mL infusion bag, cases of 10
Single-dose. Discard unused portion.
Store between 20° and 25°C (68° and 77°F) [See USP Controlled Room Temperature]. Protect from excessive heat (above 40°C) and from freezing. If refrigerated or exposed to temperatures below the freezing point, precipitation may occur. By keeping the bottle or infusion bag at room temperature with repeated shaking, the precipitate can be brought into solution again.
FOSCAVIR INJECTION should be used only if the bottle or infusion bag and its seal(s) are intact, a vacuum is present, and the solution is clear and colorless. Do not remove the infusion bag from the overwrap until ready for use.

Manufactured for: Clinigen Healthcare Ltd., DE14 2WW, UK
Distributed by: Hospira, Inc., Lake Forest, IL 60045 USA
FOSCAVIR is a trademark of Clinigen Healthcare Ltd.
Date of revision: October 2020
Mechanism of Action
Foscarnet exerts its antiviral activity by a selective inhibition at the pyrophosphate binding site on virus-specific DNA polymerases at concentrations that do not affect cellular DNA polymerases. Foscarnet does not require activation (phosphorylation) by thymidine kinase or other kinases.
Foscavir - Foscarnet Sodium injection, Solution PubMed™ news
- Journal Article • 2026 AprManaging refractory cytomegalovirus in an immunosuppressed patient with sarcoidosis: a case report on maribavir therapy.
- Journal Article • 2026 AprIntravitreal foscarnet is an effective adjunct after systemic therapy failure in pediatric antiviral-resistant cytomegalovirus retinitis.
- Journal Article • 2026 AprImpact of antiviral therapies on survival in patients with HHV-6B encephalitis after cord blood transplantation: a real-world study.
- Journal Article • 2026 AprRapid HHV-6 Clearance Following Preemptive Short-Course Foscarnet in Allogeneic Stem Cell Transplant Recipients.
- Journal Article • 2026 AprA real-world pharmacovigilance analysis of uveitis from systemic medications.