Get your patient on Gvoke Hypopen (Glucagon Injection, Solution)
Gvoke HypoPen prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Gvoke HypoPen patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
Dosage and Administration of GVOKE for Subcutaneous Use to Treat Severe Hypoglycemia in Adult and Pediatric Patients Aged 2 Years and Older with Diabetes (2.1 )
- Important Administration Instructions
- Administer GVOKE HypoPen, GVOKE PFS, or GVOKE Kit subcutaneously. These three presentations are referred to as GVOKE in this labeling
- Administer GVOKE according to the printed instructions on the foil pouch label, carton, or the Instructions for Use.
- Visually inspect GVOKE prior to administration. The solution should appear clear and colorless to pale yellow.
- Administer GVOKE subcutaneously in the lower abdomen, outer thigh, or outer upper arm.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, may administer an additional dose from a new HypoPen, GVOKE PFS, or GVOKE Kit while waiting for emergency assistance.
- When the patient has responded to GVOKE, give oral carbohydrates.
- Do not attempt to reuse GVOKE. Each GVOKE device or vial contains a single dose of glucagon. Do not reuse and discard any unused portion.
- Recommended Dosage
- Recommended dose for adult and pediatric patients aged 12 years of age and older is 1 mg.
- Recommended dose for adult and pediatric patients aged 12 years of age and older is 1 mg.
- The recommended dose for pediatric patients aged 2 to less than 12 years of age who weigh:
- Less than 45 kg, is 0.5 mg
- 45 kg or greater, is 1 mg
Dosage and Administration of GVOKE VialDx for Intravenous Use as a Diagnostic Aid in Adults (2.2 )
- GVOKE VialDx is only for intravenous use under medical supervision.
- The recommended diagnostic dose for relaxation of the stomach, duodenal bulb, duodenum, and small bulb is 0.2 mg to 0.5 mg; the recommended dose to relax the colon is 0.5 mg to 0.75 mg.
- See the Full Prescribing Information for administration instructions for GVOKE VialDx.
2.1 Dosage and Administration of GVOKE for Subcutaneous Use to Treat Severe Hypoglycemia in Adult and Pediatric Patients Aged 2 Years and Older with Diabetes
Important Administration Instructions
To treat severe hypoglycemia in adult and pediatric patients aged 2 years and older with diabetes, administer GVOKE HypoPen, GVOKE PFS, or GVOKE Kit subcutaneously. These three presentations are only for subcutaneous use and are referred to as GVOKE in this labeling.
- For GVOKE HypoPen or GVOKE PFS: Do not open foil pouch until ready to administer.
- For GVOKE Kit: Store in original carton until ready to administer.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires the help of others to recover, instruct the patient to inform those around them about GVOKE and its Instructions for Use. Administer GVOKE subcutaneously as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for GVOKE.
- Visually inspect GVOKE prior to administration. The solution should be clear, colorless to pale yellow
- Withdraw the correct dose (see below for dosage recommendations).
- Administer subcutaneously in the lower abdomen, outer thigh, or outer upper arm, according to the printed instructions on the foil pouch label, carton, or the Instructions for Use.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose from a new GVOKE HypoPen, GVOKE PFS, or GVOKE Kit may be administered while waiting for emergency assistance.
- When the patient has responded to GVOKE, give oral carbohydrates to restore the liver glycogen and prevent recurrence of hypoglycemia.
- Each GVOKE HypoPen, GVOKE PFS, or GVOKE Kit product contains a single dose of glucagon. Do not reuse and discard any unused portion.
Recommended Dosage in Adult and Pediatric Patients Aged 12 Years and Older to Treat Severe Hypoglycemia
To treat severe hypoglycemia in patients 12 years of age and older with diabetes, use GVOKE HypoPen, GVOKE PFS, or GVOKE Kit:
- The recommended dose is 1 mg administered by subcutaneous injection into lower abdomen, outer thigh, or outer upper arm.
- If there has been no response after 15 minutes, an additional 1 mg dose from a new GVOKE HypoPen, GVOKE PFS, or GVOKE Kit may be administered while waiting for emergency assistance
Recommended Dosage in Pediatric Patients Aged 2 to less than 12 Years of Age to Treat Severe Hypoglycemia
To treat severe hypoglycemia in pediatric patients aged 2 to less than 12 years of age with diabetes, use GVOKE HypoPen, GVOKE PFS, or GVOKE Kit.
- The recommended dose in those who weigh:
- Less than 45 kg is 0.5 mg administered by subcutaneous injection into the lower abdomen, outer thigh, or outer upper arm.
- 45 kg or greater is 1 mg administered by subcutaneous injection into the lower abdomen, outer thigh, or outer upper arm.
- If there has been no response after 15 minutes, an additional 0.5 mg dose (for those who weigh less than 45 kg) or 1 mg dose (for those who weigh 45 kg or greater) from a new GVOKE HypoPen, GVOKE PFS, or GVOKE Kit may be administered while waiting for emergency assistance.
Dosage and Administration of GVOKE VialDx for Intravenous Use as a Diagnostic Aid in Adults
Important Administration Information
For use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract, use GVOKE VialDx. GVOKE VialDx is only for intravenous use under medical supervision
- Must be diluted with 0.9% Sodium Chloride Injection prior to administration. Using a syringe, draw 0.2 mL from the GVOKE VialDx vial and dispense into a separate empty sterile container containing 2 mL of 0.9% Sodium Chloride Injection.
- Gently swirl the container until the solution is thoroughly mixed. The mixed solution should be clear and colorless to pale yellow. Inspect visually for particulate matter and discoloration.
- The final concentration of the diluted solution is 0.45 mg/mL of glucagon. Draw the required dose from the container into a syringe for administration (see below for dosage recommendations).
- If not used immediately, diluted GVOKE VialDx may be stored for up to 8 hours after initial dilution.
- Inject the solution intravenously via a 1-minute slow push using consistent pressure.
- After the end of the diagnostic procedure, give oral carbohydrates to patients who have been fasting, if this is compatible with the diagnostic procedure.
- GVOKE VialDx contains a single dose of glucagon. Do not reuse. Discard any unused portion.
Recommended Dosage in Adults as a Diagnostic Aid
For use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract, use GVOKE VialDx. The recommended intravenous dose for relaxation of the [see Clinical Pharmacology (12.2 )]:
- Stomach, duodenal bulb, duodenum, and small bowel is 0.2 mg to 0.5 mg
- Colon is 0.5 mg to 0.75 mg
The onset of action after an injection will depend on the organ under examination [see Clinical Pharmacology (12.2 )] .

Gvoke HypoPen prescribing information
INDICATIONS AND USAGE
- GVOKE is an antihypoglycemic agent indicated for subcutaneous use for the treatment of severe hypoglycemia in adult and pediatric patients aged two years and older with diabetes (1.1 )
- GVOKE VialDx is a gastrointestinal motility inhibitor indicated for intravenous use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract in adult patients (1.2 )
1.1 Severe Hypoglycemia in Adult and Pediatric Patients Aged 2 Years and Older with Diabetes
GVOKE is indicated for subcutaneous use for the treatment of severe hypoglycemia in adult and pediatric patients aged two and older with diabetes.
1.2 Diagnostic Aid in Adults
GVOKE VialDx is indicated for intravenous use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract in adult patients.
DOSAGE AND ADMINISTRATION
Dosage and Administration of GVOKE for Subcutaneous Use to Treat Severe Hypoglycemia in Adult and Pediatric Patients Aged 2 Years and Older with Diabetes (2.1 )
- Important Administration Instructions
- Administer GVOKE HypoPen, GVOKE PFS, or GVOKE Kit subcutaneously. These three presentations are referred to as GVOKE in this labeling
- Administer GVOKE according to the printed instructions on the foil pouch label, carton, or the Instructions for Use.
- Visually inspect GVOKE prior to administration. The solution should appear clear and colorless to pale yellow.
- Administer GVOKE subcutaneously in the lower abdomen, outer thigh, or outer upper arm.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, may administer an additional dose from a new HypoPen, GVOKE PFS, or GVOKE Kit while waiting for emergency assistance.
- When the patient has responded to GVOKE, give oral carbohydrates.
- Do not attempt to reuse GVOKE. Each GVOKE device or vial contains a single dose of glucagon. Do not reuse and discard any unused portion.
- Recommended Dosage
- Recommended dose for adult and pediatric patients aged 12 years of age and older is 1 mg.
- Recommended dose for adult and pediatric patients aged 12 years of age and older is 1 mg.
- The recommended dose for pediatric patients aged 2 to less than 12 years of age who weigh:
- Less than 45 kg, is 0.5 mg
- 45 kg or greater, is 1 mg
Dosage and Administration of GVOKE VialDx for Intravenous Use as a Diagnostic Aid in Adults (2.2 )
- GVOKE VialDx is only for intravenous use under medical supervision.
- The recommended diagnostic dose for relaxation of the stomach, duodenal bulb, duodenum, and small bulb is 0.2 mg to 0.5 mg; the recommended dose to relax the colon is 0.5 mg to 0.75 mg.
- See the Full Prescribing Information for administration instructions for GVOKE VialDx.
2.1 Dosage and Administration of GVOKE for Subcutaneous Use to Treat Severe Hypoglycemia in Adult and Pediatric Patients Aged 2 Years and Older with Diabetes
Important Administration Instructions
To treat severe hypoglycemia in adult and pediatric patients aged 2 years and older with diabetes, administer GVOKE HypoPen, GVOKE PFS, or GVOKE Kit subcutaneously. These three presentations are only for subcutaneous use and are referred to as GVOKE in this labeling.
- For GVOKE HypoPen or GVOKE PFS: Do not open foil pouch until ready to administer.
- For GVOKE Kit: Store in original carton until ready to administer.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires the help of others to recover, instruct the patient to inform those around them about GVOKE and its Instructions for Use. Administer GVOKE subcutaneously as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for GVOKE.
- Visually inspect GVOKE prior to administration. The solution should be clear, colorless to pale yellow
- Withdraw the correct dose (see below for dosage recommendations).
- Administer subcutaneously in the lower abdomen, outer thigh, or outer upper arm, according to the printed instructions on the foil pouch label, carton, or the Instructions for Use.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose from a new GVOKE HypoPen, GVOKE PFS, or GVOKE Kit may be administered while waiting for emergency assistance.
- When the patient has responded to GVOKE, give oral carbohydrates to restore the liver glycogen and prevent recurrence of hypoglycemia.
- Each GVOKE HypoPen, GVOKE PFS, or GVOKE Kit product contains a single dose of glucagon. Do not reuse and discard any unused portion.
Recommended Dosage in Adult and Pediatric Patients Aged 12 Years and Older to Treat Severe Hypoglycemia
To treat severe hypoglycemia in patients 12 years of age and older with diabetes, use GVOKE HypoPen, GVOKE PFS, or GVOKE Kit:
- The recommended dose is 1 mg administered by subcutaneous injection into lower abdomen, outer thigh, or outer upper arm.
- If there has been no response after 15 minutes, an additional 1 mg dose from a new GVOKE HypoPen, GVOKE PFS, or GVOKE Kit may be administered while waiting for emergency assistance
Recommended Dosage in Pediatric Patients Aged 2 to less than 12 Years of Age to Treat Severe Hypoglycemia
To treat severe hypoglycemia in pediatric patients aged 2 to less than 12 years of age with diabetes, use GVOKE HypoPen, GVOKE PFS, or GVOKE Kit.
- The recommended dose in those who weigh:
- Less than 45 kg is 0.5 mg administered by subcutaneous injection into the lower abdomen, outer thigh, or outer upper arm.
- 45 kg or greater is 1 mg administered by subcutaneous injection into the lower abdomen, outer thigh, or outer upper arm.
- If there has been no response after 15 minutes, an additional 0.5 mg dose (for those who weigh less than 45 kg) or 1 mg dose (for those who weigh 45 kg or greater) from a new GVOKE HypoPen, GVOKE PFS, or GVOKE Kit may be administered while waiting for emergency assistance.
Dosage and Administration of GVOKE VialDx for Intravenous Use as a Diagnostic Aid in Adults
Important Administration Information
For use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract, use GVOKE VialDx. GVOKE VialDx is only for intravenous use under medical supervision
- Must be diluted with 0.9% Sodium Chloride Injection prior to administration. Using a syringe, draw 0.2 mL from the GVOKE VialDx vial and dispense into a separate empty sterile container containing 2 mL of 0.9% Sodium Chloride Injection.
- Gently swirl the container until the solution is thoroughly mixed. The mixed solution should be clear and colorless to pale yellow. Inspect visually for particulate matter and discoloration.
- The final concentration of the diluted solution is 0.45 mg/mL of glucagon. Draw the required dose from the container into a syringe for administration (see below for dosage recommendations).
- If not used immediately, diluted GVOKE VialDx may be stored for up to 8 hours after initial dilution.
- Inject the solution intravenously via a 1-minute slow push using consistent pressure.
- After the end of the diagnostic procedure, give oral carbohydrates to patients who have been fasting, if this is compatible with the diagnostic procedure.
- GVOKE VialDx contains a single dose of glucagon. Do not reuse. Discard any unused portion.
Recommended Dosage in Adults as a Diagnostic Aid
For use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract, use GVOKE VialDx. The recommended intravenous dose for relaxation of the [see Clinical Pharmacology (12.2 )]:
- Stomach, duodenal bulb, duodenum, and small bowel is 0.2 mg to 0.5 mg
- Colon is 0.5 mg to 0.75 mg
The onset of action after an injection will depend on the organ under examination [see Clinical Pharmacology (12.2 )] .
DOSAGE FORMS AND STRENGTHS
GVOKE HypoPen, GVOKE PFS, GVOKE Kit (these three presentations are also referred to as GVOKE in this labeling) , and GVOKE VialDx are clear, colorless to pale yellow solutions.
Subcutaneous Injection
- Injection: 0.5 mg/0.1 mL single-dose prefilled autoinjector (GVOKE HypoPen)
- Injection: 1 mg/0.2 mL single-dose prefilled autoinjector (GVOKE HypoPen)
- Injection: 1 mg/0.2 mL single-dose prefilled syringe (GVOKE PFS)
- Injection: 1 mg/0.2 mL single-dose vial and syringe kit (GVOKE Kit)
Intravenous Injection:
- Injection: 1 mg/0.2 mL single-dose vial (GVOKE VialDx)
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Available data from case reports and a small number of observational studies with glucagon use in pregnant women over decades of use have not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Multiple small studies have demonstrated a lack of transfer of pancreatic glucagon across the human placental barrier during early gestation. In a rat reproduction study, no embryofetal toxicity was observed with glucagon administered by injection during the period of organogenesis at doses representing up to 40 times the human dose, based on body surface area (mg/m 2 ) (see Data ) .
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
In pregnant rats given animal sourced glucagon twice-daily by injection at doses up to 2 mg/kg (up to 40 times the human dose based on body surface area extrapolation, mg/m 2 ) during the period of organogenesis, there was no evidence of increased malformations or embryofetal lethality.
Lactation
Risk Summary
There is no information available on the presence of glucagon in human or animal milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. However, glucagon is a peptide and would be expected to be broken down to its constituent amino acids in the infant's digestive tract and is therefore, unlikely to cause harm to an exposed infant.
Pediatric Use
The safety and effectiveness of GVOKE for subcutaneous use for the treatment of severe hypoglycemia in patients with diabetes have been established in pediatric patients aged 2 years and older. Use of GVOKE for this indication is supported by evidence from two adequate and well-controlled studies in adults with type-1 diabetes mellitus [see Clinical Studies (14.1 )] and from a study in 31 pediatric patients ages 2 and older with type 1 diabetes mellitus [see Clinical Studies (14.2 )] .
The safety and effectiveness of GVOKE for subcutaneous use for the treatment of severe hypoglycemia in patients with diabetes have not been established in pediatric patients younger than 2 years of age.
Safety and effectiveness of GVOKE VialDx for intravenous use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract in pediatric patients have not been established.
Geriatric Use
Clinical studies of GVOKE and GVOKE VialDx did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger adult patients.
CONTRAINDICATIONS
GVOKE and GVOKE VialDx are contraindicated in patients with:
- Pheochromocytoma because of the risk of substantial increase in blood pressure [see Warnings and Precautions (5.1 )]
- Insulinoma because of the risk of hypoglycemia [see Warnings and Precautions (5.2 )]
- Prior hypersensitivity reaction to glucagon or to any of the excipients in GVOKE or GVOKE VialDx. Serious hypersensitivity reactions have been reported with glucagon and include anaphylactic shock with breathing difficulties and hypotension [see Warnings and Precautions (5.3 )] .
GVOKE VialDx for use as a diagnostic aid is also contraindicated in patients with glucagonoma because of risk of hypoglycemia [see Warnings and Precautions (5.8 )]
WARNINGS AND PRECAUTIONS
- Substantial Increase in Blood Pressure in Patients with Pheochromocytoma: Contraindicated in patients with pheochromocytoma because glucagon may stimulate the release of catecholamines from the tumor. (5.1 )
- Hypoglycemia in Patients with Insulinoma: In patients with insulinoma, administration may produce an initial increase in blood glucose; however, glucagon may stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. If a patient develops symptoms of hypoglycemia after a dose of GVOKE or GVOKE VialDx, give glucose orally or intravenously. (5.2 )
- Serious Hypersensitivity Reactions: Serious hypersensitivity reactions have been reported with glucagon products, including generalized rash, and in some cases anaphylactic shock with breathing difficulties, and hypotension.(5.3 )
- Lack of Efficacy with Subcutaneous Use for Severe Hypoglycemia in Patients with Decreased Hepatic Glycogen: Patients with insufficient hepatic stores of glycogen may not respond to GVOKE for subcutaneous use for the treatment of severe hypoglycemia. Insufficient hepatic stores of glycogen may be present in conditions such as states of starvation, or in patients with adrenal insufficiency or chronic hypoglycemia. (5.4 )
- Necrolytic Migratory Erythema (NME): a skin rash, has been reported postmarketing following continuous glucagon infusion and resolved with discontinuation of the glucagon. GVOKE and GVOKE VialDx are not approved for continuous infusion. Should NME occur, consider whether the benefits of continuous glucagon infusion outweigh the risks. (5.5 )
- Hyperglycemia with Intravenous Use as a Diagnostic Aid in Patients with Diabetes Mellitus: GVOKE VialDx in patients with diabetes mellitus may cause hyperglycemia. Monitor patients with diabetes for changes in blood glucose levels during treatment and treat hyperglycemia if indicated. (5.6 )
- Blood Pressure and Heart Rate Increases with Intravenous Use as a Diagnostic Aid in Patients with Cardiac Disease: GVOKE VialDx may increase myocardial oxygen demand, blood pressure, and pulse rate. Cardiac monitoring is recommended in patients with cardiac disease during use of GVOKE VialDx as a diagnostic aid, and an increase in blood pressure and pulse rate may require therapy. (5.7 )
- Hypoglycemia in Patients with Glucagonoma with Intravenous Use as a Diagnostic Aid: GVOKE VialDx administered to patients with glucagonoma may cause secondary hypoglycemia. Test patients suspected of having glucagonoma for blood levels of glucagon prior to treatment and monitor for changes in blood glucose levels during treatment. (5.8 )
Substantial Increase in Blood Pressure in Patients with Pheochromocytoma
GVOKE and GVOKE VialDx are contraindicated in patients with pheochromocytoma because glucagon may stimulate the release of catecholamines from the tumor [see Contraindications (4 )] . If the patient develops a substantial increase in blood pressure and a previously undiagnosed pheochromocytoma is suspected, 5 to 10 mg of phentolamine mesylate, administered intravenously, has been shown to be effective in lowering blood pressure.
Hypoglycemia in Patients with Insulinoma
In patients with insulinoma, administration of glucagon may produce an initial increase in blood glucose; however, glucagon administration may directly or indirectly (through an initial rise in blood glucose) stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. GVOKE and GVOKE VialDx are contraindicated in patients with insulinoma [see Contraindications (4 )] . If a patient develops symptoms of hypoglycemia after a dose of GVOKE or GVOKE VialDx, give glucose orally or intravenously.
Serious Hypersensitivity Reactions
Serious hypersensitivity reactions have been reported with glucagon products, including generalized rash, and in some cases anaphylactic shock with breathing difficulties and hypotension. Discontinue GVOKE or GVOKE VialDx if symptoms of serious hypersensitivity reactions occur. Advise patients and/or caregivers to seek immediate medical attention if the patient experiences any symptoms of serious hypersensitivity reactions.GVOKE and GVOKE VialDx are contraindicated in patients with a prior hypersensitivity reaction to glucagon, or any of the excipients in GVOKE and GVOKE VialDx [see Contraindications (4 )] .
Lack of Efficacy with Subcutaneous Use for Severe Hypoglycemia in Patients with Decreased Hepatic Glycogen
Patients with insufficient hepatic stores of glycogen may not respond to GVOKE for the treatment of severe hypoglycemia [see Clinical Pharmacology (12.2 )] . Insufficient hepatic stores of glycogen may be present in conditions such as states of starvation, or in patients with adrenal insufficiency or chronic hypoglycemia.
Necrolytic Migratory Erythema
Necrolytic migratory erythema (NME), a skin rash commonly associated with glucagonomas (glucagon-producing tumors) and characterized by scaly, pruritic erythematous plaques, bullae, and erosions, has been reported postmarketing following continuous glucagon infusion.GVOKE and GVOKE VialDx are not approved for continuous infusion. NME lesions may affect the face, groin, perineum and legs or be more widespread. In the reported cases NME resolved with discontinuation of the glucagon, and treatment with corticosteroids was not effective. Should NME occur, consider whether the benefits of continuous glucagon infusion outweigh the risks.
5.6 Hyperglycemia with Intravenous Use as a Diagnostic Aid in Patients with Diabetes Mellitus
GVOKE VialDx in patients with diabetes mellitus may cause hyperglycemia. Monitor patients with diabetes for changes in blood glucose levels during treatment with GVOKE VialDx and treat hyperglycemia, if indicated.
5.7 Blood Pressure and Heart Rate Increases with Intravenous Use as a Diagnostic Aid in Patients with Cardiac Disease
GVOKE VialDx may increase myocardial oxygen demand, blood pressure, and pulse rate which may be life threatening in patients with cardiac disease. Cardiac monitoring is recommended in patients with cardiac disease during use of GVOKE VialDx as a diagnostic aid, and an increase in blood pressure and pulse rate may require therapy.
5.8 Hypoglycemia in Patients with Glucagonoma with Intravenous Use as a Diagnostic Aid
Use of GVOKE VialDx in patients with glucagonoma may cause secondary hypoglycemia. GVOKE VialDx is contraindicated in patients with glucagonoma when used as a diagnostic aid [see Contraindications (4 )]. Test patients suspected of having glucagonoma for blood levels of glucagon prior to administration of GVOKE VialDx, and monitor for changes in blood glucose levels during treatment. If a patient develops symptoms of hypoglycemia after administration of GVOKE VialDx, administer glucose orally or intravenously.
ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in labeling:
- Substantial Increase in Blood Pressure in Patients with Pheochromocytoma [see Warnings and Precautions (5.1 )] .
- Hypoglycemia in Patients with Insulinoma [see Warnings and Precautions (5.2 )] .
- Serious Hypersensitivity Reactions [see Warnings and Precautions (5.3 )]
- Lack of Efficacy With Subcutaneous Use for Severe Hypoglycemia in Patients with Decreased Hepatic Glycogen [see Warnings and Precautions (5.4 )]
- Necrolytic Migratory Erythema [see Warnings and Precautions (5.5 )] .
- Hyperglycemia with Intravenous Use as a Diagnostic Aid in Patients with Diabetes Mellitus [see Warnings and Precautions (5.6 )]
- Blood Pressure and Heart Rate Increases with Intravenous Use as a Diagnostic Aid in Patients with Cardiac Disease [see Warnings and Precautions (5.7 )]
- Hypoglycemia in Patients with Glucagonoma with Intravenous Use as a Diagnostic Aid [see Warnings and Precautions (5.8 )]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of GVOKE and GVOKE VialDx cannot be directly compared to rates in the clinical trials of other drugs and may not reflect the rates observed in practice.
GVOKE for Subcutaneous Use for the Treatment of Severe Hypoglycemia in Adult and Pediatric Patients 2 Aged Years and Older with Type 1 Diabetes Mellitus
Adverse Reactions in Adult Patients
The safety of GVOKE for subcutaneous use for the treatment of severe hypoglycemia in adults with diabetes was evaluated in two randomized, blinded, 2-way crossover studies conducted in adults with type 1 diabetes mellitus. In total, 154 patients received a subcutaneous injection of GVOKE [see Clinical Studies (14.1 )] .
The most common adverse reactions that occurred in 2% or more of adults treated with GVOKE during these two clinical trials are listed in Table 1 .
| a Adverse Reactions that occurred within 12 hours. | |
GVOKE 1 mg dose (N = 154) | |
Nausea | 30% |
Vomiting | 16% |
Injection site edema raised 1 mm or greater | 7% |
Headache | 5% |
Injection site pain was reported by 1% of GVOKE-treated patients.
Hypertension and tachycardia have occurred with glucagon treatment.
Adverse Reactions in Pediatric Patients Aged 2 Years and Older
The safety of GVOKE for the treatment of severe hypoglycemia in patients with diabetes was evaluated in one single-arm, open-label, study in 31 pediatric patients with type 1 diabetes mellitus [see Clinical Studies (14.2 )] .
The data in Table 2 reflect the exposure of 31 pediatric patients to 0.5 mg or 1 mg of GVOKE given subcutaneously. The most common adverse reactions that occurred in ≥2% of GVOKE-treated pediatric patients aged 2 years and older are listed in Table 2 .
| a Adverse Reactions that occurred within 12 hours. | ||||
2 to 6 years of age (0.5 mg dose) N =7 | 6 to 12 years of age (0.5 mg dose) N = 13 | 12 to 18 years of age (1 mg dose) N = 11 | Total N = 31 | |
Nausea | 43% | 54% | 36% | 45% |
Hypoglycemia | 29% | 54% | 27% | 39% |
Vomiting | 14% | 23% | 18% | 19% |
Headache | 0% | 15% | 0% | 7% |
Abdominal pain | 0% | 8% | 0% | 3% |
Hyperglycemia | 14% | 8% | 0% | 7% |
Injection site discomfort | 0% | 8% | 0% | 3% |
Injection site reaction | 0% | 0% | 9% | 3% |
Urticaria | 0% | 8% | 0% | 3% |
GVOKE VialDx for Intravenous Use As a Diagnostic Aid in Adults The safety of GVOKE VialDx for intravenous use as a diagnostic aid in adults was evaluated in an open-label single-dose study in 83 adult healthy volunteers. Table 3 displays the most common adverse reactions that occurred in 5% or greater in healthy volunteers who received 0.75 mg of GVOKE VialDx intravenously.
N=83 | |
Nausea | 37.3% |
Dysgeusia | 18.1% |
Headache | 10.8% |
Hot Flush | 9.6% |
Dizziness | 8.4% |
Postmarketing Experience
Additional adverse reactions have been identified during post-approval use of glucagon. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Necrolytic migratory erythema (NME) cases have been reported postmarketing in patients receiving continuous infusion of glucagon.
- Hypoglycemia and hypoglycemic coma. Patients taking indomethacin may be more likely to experience hypoglycemia following glucagon administration [see Drug Interactions (7 )] .
DRUG INTERACTIONS
Beta-Blockers | |
Clincial Impact: | Patients taking beta-blockers may have a transient increase in pulse and blood pressure when given GVOKE or GVOKE VialDx. |
Intervention | The increase in blood pressure and heart rate may require therapy in patients with coronary artery disease. |
Insulin | |
Clincial Impact: | Insulin acts antagonistically to glucagon. |
Intervention | Monitor blood glucose when GVOKE VialDx is used as a diagnostic aid in patients receiving insulin. |
Indomethacin | |
Clincial Impact: | In patients taking indomethacin, GVOKE may lose its ability to raise blood glucose or may even produce hypoglycemia. |
Intervention | Monitor blood glucose levels during glucagon treatment of patients taking indomethacin. |
Anticholinergic Drugs | |
Clincial Impact: | The concomitant use of anticholinergic drugs and GVOKE VialDx increases the risk of gastrointestinal adverse reactions due to additive effects on inhibition of gastrointestinal motility. |
Intervention | Concomitant use of anticholinergic drugs with GVOKE VialDx is not recommended. |
Warfarin | |
Clincial Impact: | GVOKE and GVOKE Vial Dx may increase the anticoagulant effect of warfarin. |
Intervention | Monitor patients for unusual bruising or bleeding, as adjustments in warfarin dosage may be required. |
DESCRIPTION
Glucagon is an antihypoglycemic agent and a gastrointestinal motility inhibitor. Glucagon is a single chain containing 29 amino acid residues and has a molecular weight of 3483 and is identical to human glucagon. Glucagon is of synthetic origin produced by solid phase synthesis.
Its molecular formula is C 153 H 225 N 43 O 49 S with the following structure:

GVOKE HypoPen, GVOKE PFS, and GVOKE Kit
GVOKE HypoPen (glucagon) injection, GVOKE PFS (glucagon) injection, and GVOKE Kit (glucagon) injection (these three presentations are also referred to as GVOKE (glucagon) injection in this labeling) are clear, colorless to pale yellow, sterile solutions for subcutaneous injection.
- GVOKE HypoPen and GVOKE PFS: Each 0.2 mL contains 1 mg of glucagon, 11.1 mg of trehalose dihydrate NF, and 1.2 mg of 1N sulfuric acid in 209 mg dimethyl sulfoxide diluent.
- GVOKE HypoPen: Each 0.1 mL contains 0.5 mg of glucagon, 5.6 mg of trehalose dihydrate NF, and 0.6 mg of 1N sulfuric acid in 104 mg dimethyl sulfoxide diluent.
- GVOKE Kit: Each 0.2 mL contains 1 mg of glucagon, 11.1 mg of trehalose dihydrate NF, 5.8 mg of mannitol USP, and 1.32 mg of 1N sulfuric acid in 205 mg dimethyl sulfoxide diluent.
GVOKE VialDx
GVOKE VialDx (glucagon) injection is a clear, colorless to pale yellow, sterile solution for intravenous injection available in 1 mg per 0.2 mL vial.
Each 0.2 mL of GVOKE VialDx contains 1 mg of glucagon, 11.1 mg of trehalose dihydrate NF, 5.8 mg of mannitol USP, and 1.32 mg of 1N sulfuric acid in 205 mg dimethyl sulfoxide diluent prior to dilution with 0.9% Sodium Chloride [for dilution instructions, see Dosage and Administration (2.2) ] .
The diluted solution contains 0.45 mg per mL glucagon, 5 mg per mL trehalose dihydrate, 93.2 mg per mL dimethyl sulfoxide, 2.6 mg per mL mannitol, and 0.60 mg per mL 1 N sulfuric acid.
CLINICAL PHARMACOLOGY
Mechanism of Action
Glucagon increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for glucagon to produce an antihypoglycemic effect. Extrahepatic effects of glucagon include relaxation of the smooth muscle of the stomach, duodenum, small bowel, and colon.
Pharmacodynamics
GVOKE for Subcutaneous Use for the Treatment of Severe Hypoglycemia in Patients with Type 1 Diabetes Mellitus:
After subcutaneous administration of 1 mg GVOKE in adult patients with type 1 diabetes, the mean maximum glucose increase from baseline was 176 mg/dL (see Figure 1 ).
Plasma Glucose (mean ± standard error of the mean) vs. Time After Subcutaneous Administration of 1 mg of GVOKE in Adult Patients with Type 1 Diabetes Mellitus
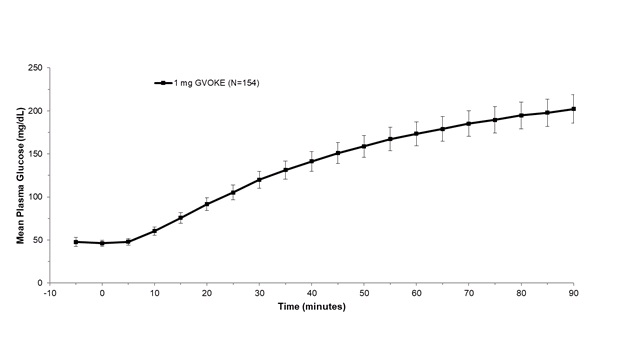
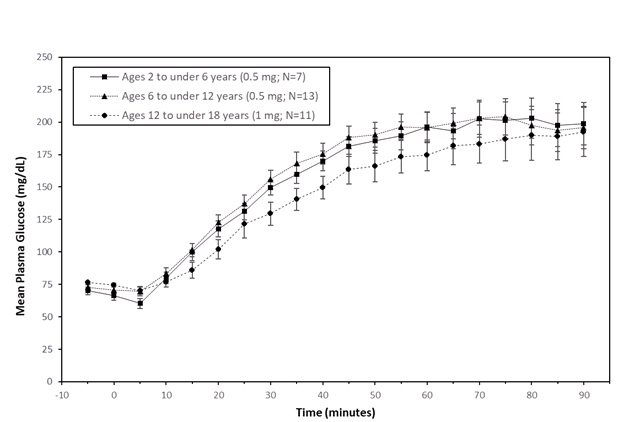
In pediatric patients with type 1 diabetes mellitus (2 to less than 18 years), the mean maximum glucose increase from baseline was 134 mg/dL (2 to less than 6 years), 145 mg/dL (6 to less than 12 years), and 123 mg/dL (12 to less than 18 years) (see Figure 2 ).
Figure 2: Plasma Glucose (mean ± standard error of the mean) vs. Time After Subcutaneous Administration of GVOKE in Pediatric Patients with Type 1 Diabetes Mellitus
GVOKE VialDx Table 5 presents the pharmacodynamic properties of another glucagon product after intravenous administration
| a Dose is determined based on the length of the procedure | ||||
Route of Administration | Dose a | Time of Maximal Glucose Concentration | Time of Onset of Action for GI Smooth Muscle Relaxation | Duration of Smooth Muscle Relaxation |
Intravenous | 0.25 to 0.5 mg | 5 to 20 minutes | 45 seconds | 9 to 17 minutes |
Pharmacokinetics
Absorption
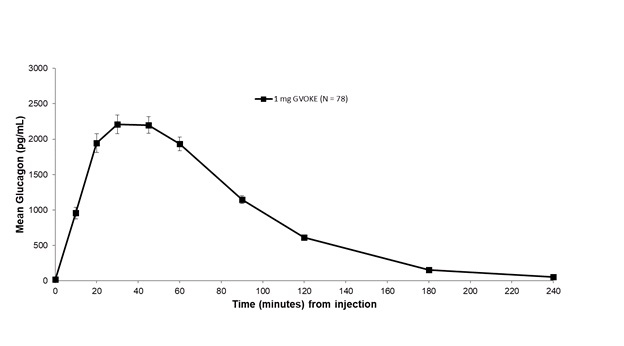
Subcutaneous injection of 1 mg GVOKE in adult type 1 diabetes mellitus patients resulted in a mean glucagon C max of 2481.3 pg/mL, t max of 50 minutes and AUC 0‑240min of 3454.6 pg•min/mL.See Figure 3 .
Figure 3: : Plasma Glucagon Concentration (mean ± standard error of the mean) vs. Time After Subcutaneous Administration of 1 mg of GVOKE in Adults with Type 1 Diabetes Mellitus
Distribution
The apparent volume of distribution was in the range of 137-2425 L.
Elimination
The half-life of GVOKE was determined to be 32 minutes.
Metabolism
Glucagon is extensively degraded in liver, kidney, and plasma.
Excretion
Urinary excretion of intact glucagon has not been measured.
Specific Populations
Pediatrics
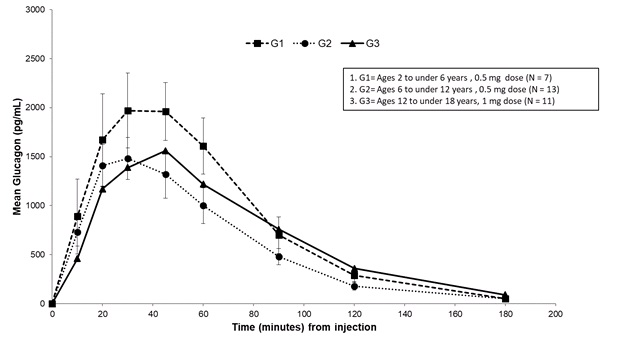
Subcutaneous injection of 0.5 mg GVOKE in patients with type 1 diabetes mellitus ages 2 to under 6 years resulted in a mean glucagon C max of 2300 pg/mL, t max of 41 minutes, and AUC 0‑180min of 138900 pg/mL•min. Subcutaneous injection of 0.5 mg GVOKE in patients with type 1 diabetes ages 6 to under 12 years resulted in a mean C max of 1600 pg/mL, median t max of 34 minutes and AUC 0‑180min of 104700 pg/mL•min. Subcutaneous injection of 1 mg GVOKE in patients with type 1 diabetes ages 12 to less than 18 years resulted in a mean C max of 1900 pg/mL, t max of 51 minutes AUC 0‑180min of 134300 pg/mL•min. Mean plasma glucagon levels were similar across the age groups following age appropriate doses of GVOKE. See Figure 4 .
Figure 4: Plasma Glucagon Concentration (mean ± standard error of the mean) vs. Time After Subcutaneous Administration of GVOKE in Pediatric Patients with Type 1 Diabetes Mellitus
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals to evaluate carcinogenic potential have not been performed. Recombinant glucagon was positive in the bacterial Ames assay. It was determined that an increase in colony counts was related to technical difficulties in running this assay with peptides. Studies in rats have shown that glucagon does not cause impaired fertility.
CLINICAL STUDIES
Adult Patients with Type 1 Diabetes Mellitus for the Treatment of Severe Hypoglycemia
GVOKE was evaluated in adult patients aged 18 to 74 years with type 1 diabetes mellitus in two multi-center 2-way crossover studies: Study A was double-blinded with 80 patients, and Study B was single-blinded with 81 patients. Both studies involved 2 clinic visits 7 to 28 days apart, with random assignment to receive GVOKE 1 mg subcutaneous injection during one session and glucagon 1 mg subcutaneous injection (subcutaneous glucagon) during the other. In these studies, 154 patients received GVOKE and 157 patients received subcutaneous glucagon. A total of 152 patients received both GVOKE and subcutaneous glucagon.
The efficacy of GVOKE was compared to subcutaneous glucagon in patients who were in a state of insulin- induced hypoglycemia via insulin infusion with target plasma glucose less than 50 mg/dL. In Study A, mean plasma glucose at time of glucagon administration was 44.8 mg/dL and 45.2 mg/dL for the GVOKE and subcutaneous glucagon groups, respectively. In Study B, mean plasma glucose at time of glucagon administration was 47.7 mg/dL and 48.7 mg/dL for the GVOKE and subcutaneous glucagon groups, respectively.
Treatment “success” was defined as plasma glucose increase from mean value at time of glucagon administration to absolute value greater than 70 mg/dL or relative increase of 20 mg/dL or greater, at 30 minutes after glucagon administration. In a pooled analysis of Study A and Study B, the proportion of patients who achieved treatment “success” was 98.7 % in the GVOKE group and 100% in the subcutaneous glucagon group and the comparison between groups met the pre-specified non-inferiority margin. A summary of treatment “success” rates is shown in Table 6 .
The mean time to treatment "success" was 13.8 minutes in the GVOKE group and 10 minutes the subcutaneous glucagon group.
| a Treatment success was defined as blood glucose greater than 70 mg/dL or an increase of blood glucose by 20 mg/dL or greater from baseline. The efficacy analysis population consisted of all patients who received both doses of the study drug. | ||||||
| b Percentage based on number of patients from both studies. | ||||||
Study A (n=80) | Study B (n=81) | |||||
GVOKE | subcutaneous glucagon | GVOKE | subcutaneous glucagon | |||
Treatment Success-n (%) a | 76 (97 %) | 79 (100%) | 76 (100%) | 78 (100%) | ||
Glucose criteria met- n (%) | ||||||
Greater than 70 mg/dL | 74 (95%) | 79 (100%) | 76 (100%) | 78 (100%) | ||
20 mg/dL or greater increase from baseline | 76 (97%) | 79 (100%) | 76 (100%) | 78 (100%) | ||
Pediatric Patients Aged 2 Years and Older with Type 1 Diabetes Mellitus for the Treatment of Severe Hypoglycemia
GVOKE was evaluated in a study in 31 pediatric patients with type 1 diabetes mellitus. Pediatric patients were administered insulin to induce a plasma glucose of less than 80 mg/dL, following which patients ages 2 to under 12 years of age received a 0.5 mg subcutaneous dose of GVOKE and patients ages 12 and older received a 0.5 mg or 1 mg subcutaneous dose of GVOKE.
All evaluable pediatric patients (30/30) achieved a target glucose increase of at least 25 mg/dL. Following administration, plasma glucose levels over time showed similar glucose responses for patients in each age group. A summary of plasma glucose results are shown in Table 7 .
| SD=standard deviation | ||||
Age Group | GVOKE Dose | Plasma Glucose (mg/dL) Mean (SD) | ||
Baseline | 30 minutes | Change | ||
2 to under 6 years (n=7) | 0.5 mg | 68.1 (8.3) | 149.6 (15.2) | 81.4 (18.3) |
6 to under 12 years (n=13) | 0.5 mg | 71.6 (7.6) | 155.8 (26.5) | 84.2 (25.3) |
12 to under 18 years (n=11) | 0.5 mg | 75.2(2.1) | 128.1(20.46) | 52.9(19.88) |
1 mg | 74.5(4.84) | 129.5 (29.5) | 55 (27.3) | |
HOW SUPPLIED/STORAGE AND HANDLING
GVOKE glucagon injection is supplied as a clear, colorless to pale yellow solution in the following configurations:
Strength | Package Size | NDC number |
For Subcutaneous Use | ||
0.5 mg per 0.1 mL | 1 single-dose Gvoke HypoPen auto-injector | 72065-120-11 |
0.5 mg per 0.1 mL | 2 single-dose Gvoke HypoPen auto-injectors | 72065-120-12 |
1 mg per 0.2 mL | 1 single-dose Gvoke HypoPen auto-injector | 72065-121-11 |
1 mg per 0.2 mL | 2 single-dose Gvoke HypoPen auto-injectors (HypoPen) | 72065-121-12 |
1 mg per 0.2 mL | 1 single-dose GVOKE PFS pre-filled syringe | 72065-131-11 |
1 mg per 0.2 mL | 2 single-dose GVOKE PFS pre-filled syringes | 72065-131-12 |
1 mg per 0.2 mL | 1 single-dose GVOKE Kit vial and syringe kit | 72065-140-11 |
For Intravaneous Use | ||
1 mg per 0.2 mL | 1 single-dose GVOKE VialDx vial | 0517-2901-01 |
1 mg per 0.2 mL | 10 single-dose GVOKE VialDx vials | 0517-2901-10 |
Store GVOKE HypoPen, GVOKE PFS, and GVOKE Kit (these three presentations are referred to as GVOKE in this labeling), and GVOKE VialDx at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). Do not refrigerate or freeze. Do not expose to extreme temperatures.
- Store the GVOKE HypoPen and GVOKE PFS in the original sealed foil pouch until time of use
- Store the GVOKE Kit vial and pouched syringe together in original carton until time of use.
- Store GVOKE VialDx vials in original carton until time of use. Discard any unused portion
INSTRUCTIONS FOR USE
GVOKE® PFS [GEE-voke P-F-S ] (glucagon injection) pre-filled syringe for subcutaneous use
This “Instructions for Use” contains information on how to inject GVOKE® PFS
Section headings and other template-related items found in this IFU are for organization of the document only and are not intended for use in corresponding artwork/patient-facing IFU files. Figure numbers in this content document may not correspond to figure numbers in corresponding artwork/patient-facing IFU files.
Appropriate trademark symbol (™ or ®) should be used upon first use of a trademarked name on each page; usage may differ from this content file and final artwork based on page layout.
1. GVOKE® PFS VISUAL
Understanding GVOKE PFS
GVOKE PFS contains a 1 mg dose of glucagon and is in a foil pouch. Below is a picture of the pouch. See the GVOKE PFS package for a full view of the Quick-Use Guide.
GVOKE PFS (1 mg dose)
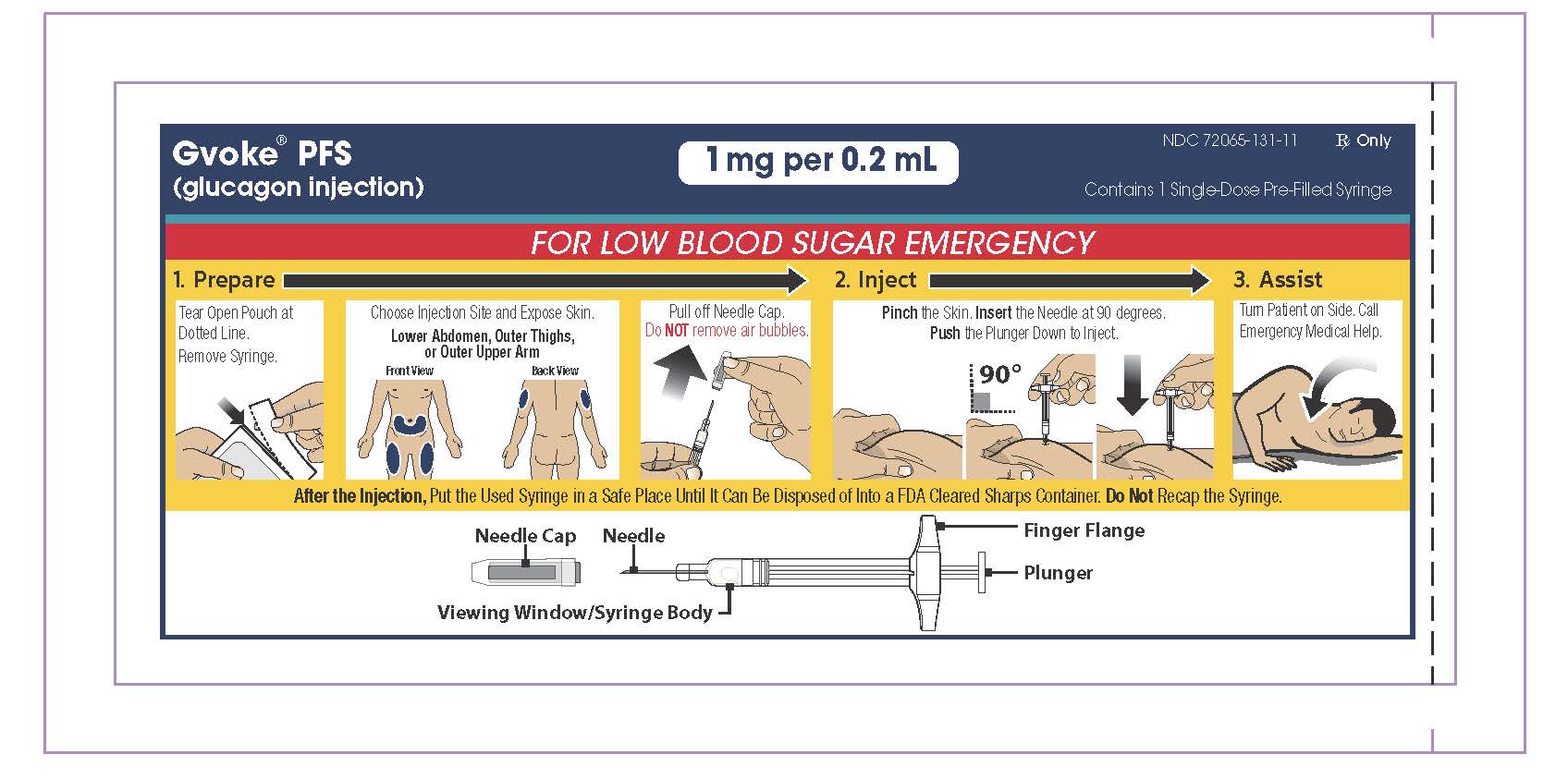
PFS Device
Note: GVOKE PFS should be used one time and then thrown away (discarded).
2. IMPORTANT INFORMATION YOU NEED TO KNOW BEFORE INJECTING GVOKE® PFS
- Become familiar with the following instructions before an emergency happens.
- Do not use this product past the expiration date printed on the device. Replace GVOKE PFS before the expiration date on the box.
- If you have questions regarding the use of this product, talk to a healthcare provider or pharmacist.
Make sure that relatives, close friends, or caregivers know that if you become unconscious, they should call for emergency medical help right away. GVOKE PFS may have been prescribed so that relatives, close friends, and caregivers can give the injection if you become hypoglycemic (severe low blood sugar) and are unable to take sugar by mouth. If you are unconscious, GVOKE PFS can be given while awaiting medical assistance.
Show your relatives, close friends, or caregivers where you store GVOKE PFS and how to use it. They need to know how to use GVOKE PFS before an emergency situation happens.
Indications for Use
GVOKE PFS is for the treatment of severe hypoglycemia in pediatric and adult patients with diabetes ages 2 years and above. Symptoms of severe hypoglycemia include unconsciousness, and seizures or convulsions.
Give GVOKE PFS if:
- the patient is unconscious,
- the patient is unable to eat sugar or a sugar-sweetened product,
- the patient is having a seizure, or
- you have tried to give the patient sugar or drinks that are high in sugar such as a regular soft drink (soda) or fruit juice and the patient does not get better.
Milder cases of hypoglycemia should be treated promptly by eating sugar or a sugar sweetened product. (See Information on Hypoglycemia for more information on the symptoms of low blood sugar.) GVOKE PFS will not work when taken by mouth (orally).
Information on Hypoglycemia
Early symptoms of hypoglycemia (low blood sugar) include:
|
|
If not treated, the patient may progress to severe hypoglycemia which can include:
- confusion
- seizures
- unconsciousness
- death
The occurrence of early symptoms calls for quick and, if necessary, repeated administration of some form of carbohydrate. Patients should always carry a quick source of sugar, such as candy mints or glucose tablets. The prompt treatment of mild hypoglycemic symptoms can prevent severe hypoglycemic reactions. If the patient does not improve or if administration of carbohydrate is impossible, GVOKE® PFS should be given or the patient should be treated with intravenous glucose by a medical professional.
Possible Problems with GVOKE PFS Treatment
Common side effects in adults and pediatric patients are nausea and vomiting. The product may cause serious side effects including serious allergic reactions, fast heart beat and high blood pressure.
People may be allergic to glucagon or to one of the inactive ingredients in GVOKE PFS, or may experience fast heart-beat for a short while.
If you experience any other reactions that may have been caused by GVOKE PFS, please contact your healthcare provider.
Important:
- Act quickly. Prolonged unconsciousness may be harmful.
- After the injection is complete, turn the unconscious patient on his or her side to prevent them from choking in case they throw up (vomit).
- Carefully read and follow these instructions. Have a healthcare provider show you the right way to use GVOKE PFS.
Important Warnings
- Do not open pouch until time of use.
- Do not use after the expiration date has passed.
- Do not use if the needle cap has been removed or is damaged.
- Do not remove the needle cap until you are ready to inject.
- Do not remove the finger flange from the syringe.
- Call a healthcare provider as soon as GVOKE® PFS has been injected.
- If the patient does not wake up within 15 minutes, give another dose of GVOKE PFS and call for emergency medical help right away.
- Feed the patient as soon as he or she wakes up and is able to swallow.
Read and become familiar with the following instructions before an emergency happens. If you have questions about using GVOKE PFS, talk with your healthcare provider or pharmacist.
3. PREPARING TO INJECT GVOKE® PFS
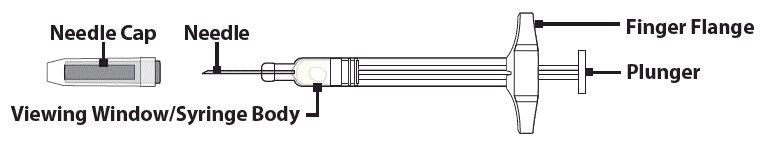
Step 1. Remove GVOKE PFS from Foil Pouch
- Tear open pouch at the dotted line and carefully remove GVOKE PFS (see Figure 1 ).
Figure 1
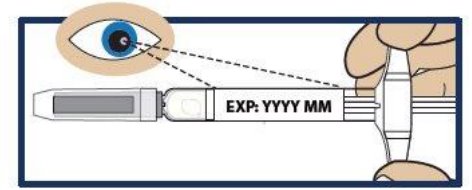
Step 2. Check the Expiration Date
- Check the expiration date printed on the label of GVOKE PFS (see Figure 2 ).
- Important: Do not use GVOKE PFS if the expiration date has passed. If GVOKE PFS is expired, throw it away in an FDA cleared sharps container and use a new GVOKE PFS.
Figure 2
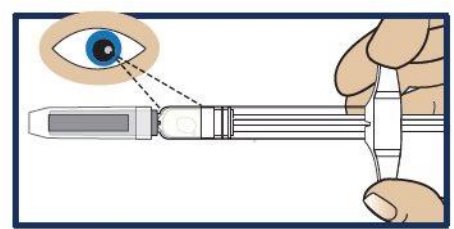
Step 3. Inspect the Solution
- Look at the liquid medicine through the viewing window. It must be clear and colorless, or a pale yellow (see Figure 3 ).
- It is normal to see air bubbles in the medicine.
- Important: Do not try to remove air bubbles before injecting.
- Do not use GVOKE® PFS or inject if the liquid contains lumps, flakes, or particles.
- Do not inject if solution is not visible in the viewing window.
- If you do not have another GVOKE PFS to use, call for emergency help right away.
Figure 3
4. INJECTING GVOKE® PFS
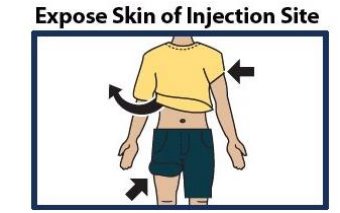
Step 4. Choose Injection Site and Expose Bare Skin
- Choose the lower abdomen, outer thigh, or outer upper arm for your injection site (see Figure 4 ).
- Remove any clothing covering the injection site (see Figure 5 ). The injection must be performed straight into the skin.
- Important: Do not inject through clothing
Figure 4
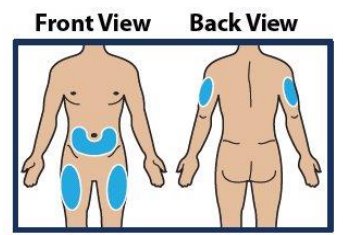
Figure 5
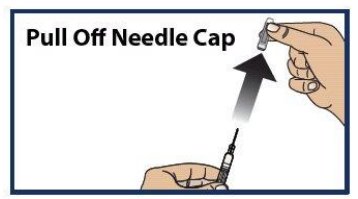
Step 5. Pull off the Needle Cap
- Pull the needle cap straight off the syringe (see Figure 6 ).
- Important: Do not put your thumb, fingers, or hand on or near the needle to help prevent accidental needle sticks.
Figure 6
Step 6. Pinch, Insert and Push to Start Injection
- Pinch the skin directly around the chosen injection site and keep pinching for the entire injection (see Figure 7 ). This is recommended to make sure a subcutaneous (under the skin) injection is given and to prevent injection into the muscle.
- Without touching the plunger, insert the needle into the skin at the injection site at a 90-degree angle (see Figure 8 ).
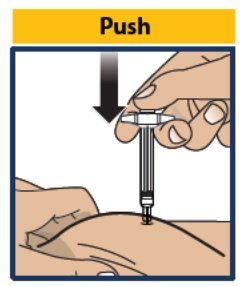
- Push the plunger down as far as it will go to inject all of the liquid medicine into the skin (see Figure 9 ). You want to inject the medicine very fast to help decrease the pain.
- Important: Do not aspirate (pull back on plunger rod) after inserting the needle. Push the plunger down as far as it will go. Do not lift up GVOKE® PFS until the injection is complete.
Figure 7
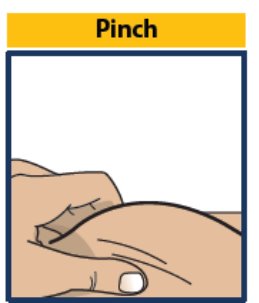
Figure 8
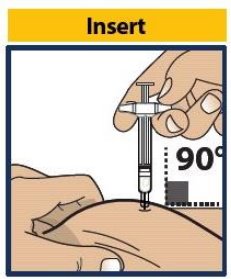
Figure 9
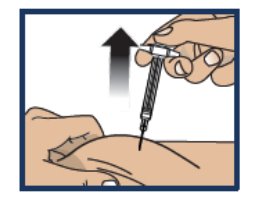
Step 7. Lift Away from Skin
- Lift the syringe straight up from the injection site (see Figure 10 ).
- Important: Do not re-cap the syringe.
Figure 10
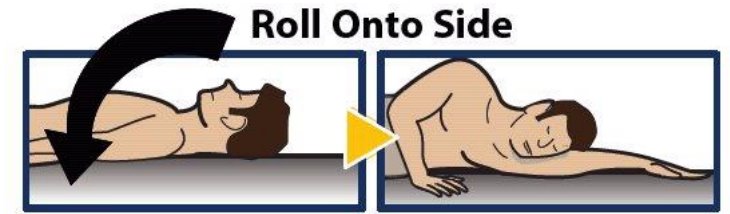
Step 8. Turn Patient onto Side
- When an unconscious person wakes up, he or she may throw up (vomit).
- Turn the unconscious patient on their side to prevent choking (see Figure 11 ).
Figure 11
Step 9. Make Sure Patient Receives Immediate Medical Attention After Use
- Call for emergency medical help right after GVOKE® PFS has been injected.
- Even if GVOKE PFS helps the patient to wake up, you should still call for emergency medical help right away.
- The patient’s healthcare provider should also be notified whenever a severe drop in blood sugar (hypoglycemic reactions) happens. Hypoglycemia may happen again after receiving an injection from GVOKE PFS. The patient’s diabetes medicine may need to be changed.
- Feed the patient as soon as he or she wakes up and is able to swallow. Give the patient a fast-acting source of sugar (such as a regular soft drink or fruit juice) and a long-acting source of sugar (such as crackers and cheese or a meat sandwich). If the patient does not wake up within 15 minutes, give another dose of glucagon if a second GVOKE PFS is available and notify emergency medical services right away.
5. STORING GVOKE® PFS
Storage Information
- Store in sealed original foil pouch until time of use.
- Store at room temperature, 68° to 77°F (20° to 25°C).
- Do not refrigerate or freeze.
6. DISPOSING OF GVOKE® PFS
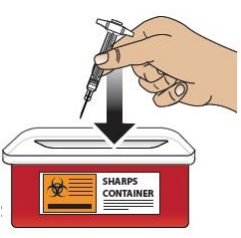
Dispose of GVOKE PFS in an FDA Cleared Sharps Disposal Container
To prevent injury caused from contact with the used needle, put the used syringe in a safe place until it can be disposed of into a FDA cleared sharps container right away after use (see Figure 12 ). Do not throw away (dispose of) loose needles and syringes in your household trash.
If you do not have a FDA cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic
- can be closed with a tight-fitting puncture-resistant lid, without sharps being able to come out
- upright and stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container.
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA’s website at: http://www.fda.gov/safesharpsdisposal .
Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Always keep the sharps container out of the reach of children. If needed, make sure to get a refill of GVOKE PFS.
Figure 12
7. ADDITIONAL INFORMATION
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by: Xeris Pharmaceuticals, Inc. Chicago, IL 60607
Revised 04/2023
©2023 by Xeris Pharmaceuticals, Inc.