Get your patient on Haloperidol - Haloperidol solution, Concentrate (Haloperidol)
Haloperidol - Haloperidol solution, Concentrate prescribing information
WARNING
Increased Mortality in Elderly Patients With Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10 week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Haloperidol is not approved for the treatment of patients with dementia- related psychosis (see WARNINGS ).
INDICATIONS AND USAGE
Haloperidol oral solution is indicated for use in the management of manifestations of psychotic disorders.
Haloperidol oral solution is indicated for the control of tics and vocal utterances of Tourette's Disorder in children and adults.
Haloperidol oral solution is effective for the treatment of severe behavior problems in children of combative, explosive hyperexcitability (which cannot be accounted for by immediate provocation). Haloperidol is also effective in the short-term treatment of hyperactive children who show excessive motor activity with accompanying conduct disorders consisting of some or all of the following symptoms: impulsivity, difficulty sustaining attention, aggressivity, mood lability and poor frustration tolerance.
Haloperidol should be reserved for these two groups of children only after failure to respond to psychotherapy or medications other than antipsychotics.
DOSAGE AND ADMINISTRATION
There is considerable variation from patient to patient in the amount of medication required for treatment. As with all antipsychotic drugs, dosage should be individualized according to the needs and response of each patient. Dosage adjustments, either upward or downward, should be carried out as rapidly as practicable to achieve optimum therapeutic control.
To determine the initial dosage, consideration should be given to the patient's age, severity of illness, previous response to other antipsychotic drugs, and any concomitant medication or disease state. Children, debilitated or geriatric patients, as well as those with a history of adverse reactions to antipsychotic drugs, may require less haloperidol. The optimal response in such patients is usually obtained with more gradual dosage adjustments and at lower dosage levels, as recommended below.
Clinical experience suggests the following recommendations:
Initial Dosage Range
Adults
Moderate Symptomatology 0.5 mg to 2 mg b.i.d. or t.i.d. |
Severe Symptomatology 3 mg to 5 mg b.i.d. or t.i.d. |
To achieve prompt control, higher doses may be required in some cases.
Geriatric or Debilitated Patients 0.5 mg to 2 mg b.i.d. or t.i.d. |
Chronic or Resistant Patients 3 mg to 5 mg b.i.d. or t.i.d. |
Patients who remain severely disturbed or inadequately controlled may require dosage adjustment. Daily dosages up to 100 mg may be necessary in some cases to achieve an optimal response. Infrequently, haloperidol has been used in doses above 100 mg for severely resistant patients; however, the limited clinical usage has not demonstrated the safety of prolonged administration of such doses. |
Children
The following recommendations apply to children between the ages of 3 and 12 years (weight range 15 to 40 kg). Haloperidol is not intended for children under 3 years old. Therapy should begin at the lowest dose possible (0.5 mg per day). If required, the dose should be increased by an increment of 0.5 mg at 5 to 7 day intervals until the desired therapeutic effect is obtained (see chart below).
The total dose may be divided, to be given b.i.d. or t.i.d.
Psychotic Disorders 0.05 mg/kg/day to 0.15 mg/kg/day |
Non-Psychotic Behavior 0.05 mg/kg/day to 0.075 mg/kg/day Disorders and Tourette's Disorder |
Severely disturbed psychotic children may require higher doses. |
In severely disturbed, non-psychotic children or in hyperactive children with accompanying conduct disorders, who have failed to respond to psychotherapy or medications other than antipsychotics, it should be noted that since these behaviours may be short-lived, short-term administration of haloperidol may suffice. There is no evidence establishing a maximum effective dosage. There is little evidence that behaviour improvement is further enhanced in dosages beyond 6 mg per day. |
Maintenance Dosage
Upon achieving a satisfactory therapeutic response, dosage should then be gradually reduced to the lowest effective maintenance level.
Intramuscular Administration
Adults
Parenteral medication, administered intramuscularly in doses of 2 to 5 mg, is utilized for prompt control of acutely agitated patient with moderately severe to very severe symptoms. Depending on the response of the patient, subsequent doses may be given, administered as often as every hour, although 4 to 8 hour intervals may be satisfactory.
Controlled trials to establish the safety and effectiveness of intramuscular administration in children have not been conducted.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Switchover Procedure
(From Intramuscular Administration)
The oral form should supplant the injectable as soon as practicable. In the absence of bioavailability studies establishing bioequivalence between these two dosage forms the following guidelines for dosage are suggested. For an initial approximation of the total daily dose required, the parenteral dose administered in the preceding 24 hours may be used. Since this dose is only an initial estimate, it is recommended that careful monitoring of clinical signs and symptoms, including clinical efficacy, sedation, and adverse effects, be carried out periodically for the first several days following the initiation of switchover. In this way, dosage adjustments, either upward or downward, can be quickly accomplished. Depending on the patient's clinical status, the first oral dose should be given within 12 to 24 hours following the last parenteral dose.
ADVERSE REACTIONS
CNS Effects
Extrapyramidal Symptoms (EPS)
EPS during the administration of haloperidol have been reported frequently, often during the first few days of treatment. EPS can be categorized generally as Parkinson-like symptoms, akathisia, or dystonia (including opisthotonos and oculogyric crisis). While all can occur at relatively low doses, they occur more frequently and with greater severity at higher doses. The symptoms may be controlled with dose reductions or administration of antiparkinson drugs such as benztropine mesylate USP or trihexyphenidyl hydrochloride USP. It should be noted that persistent EPS have been reported; the drug may have to be discontinued in such cases.
Withdrawal Emergent Neurological Signs
Generally, patients receiving short term therapy experience no problems with abrupt discontinuation of antipsychotic drugs. However, some patients on maintenance treatment experience transient dyskinetic signs after abrupt withdrawal. In certain of these cases the dyskinetic movements are indistinguishable from the syndrome described below under "Tardive Dyskinesia" except for duration. It is not known whether gradual withdrawal of antipsychotic drugs will reduce the rate of occurrence of withdrawal emergent neurological signs but until further evidence becomes available, it seems reasonable to gradually withdraw use of haloperidol.
Tardive Dyskinesia
As with all antipsychotic agents haloperidol has been associated with persistent dyskinesias. Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary dyskinetic movements, may appear in some patients on long-term therapy or may occur after drug therapy has been discontinued. The risk appears to be greater in elderly patients on high-dose therapy, especially females. The symptoms are persistent and in some patients appear irreversible. The syndrome is characterized by rhythmical involuntary movements of tongue, face, mouth or jaw (e.g., protrusion of tongue, puffing of cheeks, puckering of mouth, chewing movements). Sometimes these may be accompanied by involuntary movements of extremities and the trunk.
There is no known effective treatment for tardive dyskinesia; antiparkinson agents usually do not alleviate the symptoms of this syndrome. It is suggested that all antipsychotic agents be discontinued if these symptoms appear. Should it be necessary to reinstitute treatment, or increase the dosage of the agent, or switch to a different antipsychotic agent, this syndrome may be masked.
It has been reported that fine vermicular movement of the tongue may be an early sign of tardive dyskinesia and if the medication is stopped at that time the full syndrome may not develop.
Tardive Dystonia
Tardive dystonia, not associated with the above syndrome, has also been reported. Tardive dystonia is characterized by delayed onset of choreic or dystonic movements, is often persistent, and has the potential of becoming irreversible.
Dystonia
Class effect
Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Other CNS Effects
Insomnia, restlessness, anxiety, euphoria, agitation, drowsiness, depression, lethargy, headache, confusion, vertigo, grand mal seizures, exacerbation of psychotic symptoms including hallucinations, and catatonic-like behavioural states which may be responsive to drug withdrawal and/or treatment with anticholinergic drugs.
Body as a Whole
Neuroleptic malignant syndrome (NMS), hyperpyrexia and heat stroke have been reported with haloperidol (see WARNINGS for further information concerning NMS).
Cardiovascular Effects
Tachycardia, hypotension, hypertension and ECG changes including prolongation of the Q-T interval and ECG pattern changes compatible with the polymorphous configuration of torsade de pointes.
Hematologic Effects
Reports have appeared citing the occurrence of mild and usually transient leukopenia and leukocytosis, minimal decreases in red blood cell counts, anemia, or a tendency toward lymphomonocytosis. Agranulocytosis has rarely been reported to have occurred with the use of haloperidol, and then only in association with other medication.
Liver Effects
Impaired liver function and/or jaundice have been reported.
Dermatologic Reactions
Maculopapular and acneiform skin reactions and isolated cases of photosensitivity and loss of hair.
Endocrine Disorders
Lactation, breast engorgement, mastalgia, menstrual irregularities, gynecomastia, impotence, increased libido, hyperglycemia, hypoglycemia and hyponatremia.
Gastrointestinal Effects
Anorexia, constipation, diarrhea, hypersalivation, dyspepsia, nausea and vomiting.
Autonomic Reactions
Dry mouth, blurred vision, urinary retention, diaphoresis and priapism.
Respiratory Effects
Laryngospasm, bronchospasm and increased depth of respiration.
Special Senses
Cataracts, retinopathy and visual disturbances.
Other
Cases of sudden and unexpected death have been reported in association with the administration of haloperidol. The nature of the evidence makes it impossible to determine definitively what role, if any, haloperidol played in the outcome of the reported cases. The possibility that haloperidol caused death cannot, of course, be excluded, but it is to be kept in mind that sudden and unexpected death may occur in psychotic patients when they go untreated or when they are treated with other antipsychotic drugs.
Postmarketing Events
Hyperammonemia has been reported in a 5 1⁄2 year old child with citrullinemia, an inherited disorder of ammonia excretion, following treatment with haloperidol.
To report SUSPECTED ADVERSE REACTIONS, contact Advagen Pharma Ltd, at 866-488-0312 or FDA at 1-800- FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Haloperidol is the first of the butyrophenone series of major tranquilizers. The chemical designation is 4-[4-(p-chlorophenyl)-4-hydroxy-piperidino]-4'-fluorobutyrophenone and it has the following structural formula:
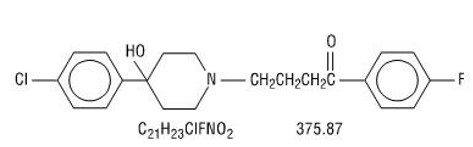
Haloperidol oral solution USP (concentrate) contains 2 mg per mL haloperidol (as the lactate).
Inactive ingredients: Lactic acid, methylparaben, propylparaben, propylene glycol, and purified water.
CLINICAL PHARMACOLOGY
The precise mechanism of action has not been clearly established.
HOW SUPPLIED
Haloperidol oral solution USP (concentrate), 2 mg per mL (as the lactate) is a clear, colorless solution, free from any visible foreign and particulate matter, free of precipitation and hazy mass is available in the following oral dosage forms:
NDC 72888-173-47: 15 mL Bottle with an accompanying dropper calibrated at 0.5 mg and 1.0 mg.
NDC 72888-173-48: 4 fl oz (120 mL) Bottle: with an accompanying dropper calibrated at 1.0 mg, 1.5 mg, 2.0 mg, 3.0 mg, 4.0 mg and 5.0 mg
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Protect from freezing. Protect from light.
Dispense in a tight, light-resistant container as described in the USP.
Distributed by: Advagen Pharma Ltd., East Windsor, NJ 08520, USA
Manufactured by: Rubicon Research Limited Satara 415004, India.
Rev. 12/2025
Haloperidol - Haloperidol solution, Concentrate PubMed™ news
- Journal Article • 2026 MayOzonation and UV-C photolysis for the removal of antipsychotics in real hospital wastewater: kinetics, removal efficiency, and insights into transformation products.
- Journal Article • 2026 MayNeuronal deletion of PDE7A averts morphine-induced behavioral plasticity and impairs downstream AKT signaling.
- Journal Article • 2026 MayUltrastructure of dopaminergic varicosities revealed by cryo-correlative light and electron microscopy.
- Journal Article • 2026 AprReward Circuit Adaptations After Chronic Antipsychotic Treatment Confer Addiction Vulnerability.
- Journal Article • 2026 AprExploring Current Emergency Medical Services Approaches to Manage Agitated Older Adult Patients: An Analysis of Statewide Protocols.