Heparin Sodium - Heparin Sodium injection, Solution prescribing information
INDICATIONS AND USAGE
Heparin Sodium Injection is indicated for:
Anticoagulant therapy in prophylaxis and treatment of venous thrombosis and its extension;
Low-dose regimen for prevention of postoperative deep venous thrombosis and pulmonary embolism in patients undergoing major abdominothoracic surgery or who, for other reasons, are at risk of developing thromboembolic disease (see DOSAGE AND ADMINISTRATION );
Prophylaxis and treatment of pulmonary embolism;
Atrial fibrillation with embolization;
Treatment of acute and chronic consumptive coagulopathies (disseminated intravascular coagulation);
Prevention of clotting in arterial and cardiac surgery;
Prophylaxis and treatment of peripheral arterial embolism.
Heparin may also be employed as an anticoagulant in blood transfusions, extracorporeal circulation, and dialysis procedures.
DOSAGE AND ADMINISTRATION
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Confirm the choice of the correct Heparin Sodium Injection vial prior to administration of the drug to a patient (see WARNINGS , Fatal Medication Errors ). The 1 mL vial must not be confused with a ‘‘catheter lock flush’’ vial or other 1 mL vial of inappropriate strength. Confirm that you have selected the correct medication and strength prior to administration of the drug.
When heparin is added to an infusion solution for continuous intravenous administration, the container should be inverted at least six times to ensure adequate mixing and prevent pooling of the heparin in the solution.
Heparin sodium is not effective by oral administration and should be given by intermittent intravenous injection, intravenous infusion, or deep subcutaneous (intrafat, i.e., above the iliac crest or abdominal fat layer) injection. The intramuscular route of administration should be avoided because of the frequent occurrence of hematoma at the injection site.
The dosage of heparin sodium should be adjusted according to the patient’s coagulation test results. When heparin is given by continuous intravenous infusion, the coagulation time should be determined approximately every four hours in the early stages of treatment. When the drug is administered intermittently by intravenous injection, coagulation tests should be performed before each injection during the early stages of treatment and at appropriate intervals thereafter. Dosage is considered adequate when the activated partial thromboplastin time (APTT) is 1.5 to 2 times normal or when the whole blood clotting time is elevated approximately 2.5 to 3 times the control value. After deep subcutaneous (intrafat) injections, tests for adequacy of dosage are best performed on samples drawn four to six hours after the injection.
Periodic platelet counts, hematocrits and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration.
Converting to Oral Anticoagulant
When an oral anticoagulant of the coumarin or similar type is to be begun in patients already receiving heparin sodium, baseline and subsequent tests of prothrombin activity must be determined at a time when heparin activity is too low to affect the prothrombin time. This is about five hours after the last IV bolus and 24 hours after the last subcutaneous dose. If continuous IV heparin infusion is used, prothrombin time can usually be measured at any time.
In converting from heparin to an oral anticoagulant, the dose of the oral anticoagulant should be the usual initial amount and thereafter prothrombin time should be determined at the usual intervals. To ensure continuous anticoagulation, it is advisable to continue full heparin therapy for several days after the prothrombin time has reached the therapeutic range. Heparin therapy may then be discontinued without tapering.
Therapeutic Anticoagulant Effect with Full-Dose Heparin
Although dosage must be adjusted for the individual patient according to the results of suitable laboratory tests, the following dosage schedules may be used as guidelines:
METHOD OF ADMINISTRATION | FREQUENCY | RECOMMENDED DOSE (based on 150 lb [68 kg] patient) |
Deep Subcutaneous (Intrafat) Injection A different site should be used for each injection to prevent the development of massive hematoma | Initial Dose | 5,000 units by IV injection, followed by 10,000 to 20,000 units of a concentrated solution, subcutaneously |
Every 8 hours or Every 12 hours | 8,000 to 10,000 units of a concentrated solution 15,000 to 20,000 units of a concentrated solution | |
Intermittent Intravenous Injection | Initial Dose | 10,000 units, either undiluted or in 50 to 100 mL of 0.9% Sodium Chloride Injection, USP |
Every 4 to 6 hours | 5,000 to 10,000 units, either undiluted or in 50 to 100 mL of 0.9% Sodium Chloride Injection, USP | |
Intravenous Infusion | Initial Dose | 5,000 units by IV injection |
Continuous | 20,000 to 40,000 units/ 24 hours in 1,000 mL of 0.9% Sodium Chloride Injection, USP (or in any compatible solution) for infusion |
Pediatric Use
Use preservative-free HEPARIN SODIUM INJECTION in neonates and infants (see PRECAUTIONS, Pediatric Use ).
There are no adequate and well-controlled studies on heparin use in pediatric patients. Pediatric dosing recommendations are based on clinical experience. In general, the following dosage schedule may be used as a guideline in pediatric patients:
Initial Dose
75 to 100 units/kg (IV bolus over 10 minutes)
Maintenance Dose
Infants: 25 to 30 units/kg/hour; Infants < 2 months have the highest requirements (average 28 units/kg/hour)
Children > 1 year of age: 18 to 20 units/kg/hour;
Older children may require less heparin, similar to weight-adjusted adult dosage
Monitoring
Adjust heparin to maintain aPTT of 60 to 85 seconds, assuming this reflects an anti-Factor Xa level of 0.35 to 0.70.
Geriatric Use
Patients over 60 years of age may require lower doses of heparin.
Surgery of the Heart and Blood Vessels
Patients undergoing total body perfusion for open-heart surgery should receive an initial dose of not less than 150 units of heparin sodium per kilogram of body weight. Frequently, a dose of 300 units of heparin sodium per kilogram of body weight is used for procedures estimated to last less than 60 minutes, or 400 units per kilogram for those estimated to last longer than 60 minutes.
Low-Dose Prophylaxis of Postoperative Thromboembolism
A number of well-controlled clinical trials have demonstrated that low-dose heparin prophylaxis, given just prior to and after surgery, will reduce the incidence of postoperative deep vein thrombosis in the legs (as measured by the I-125 fibrinogen technique and venography) and of clinical pulmonary embolism. The most widely used dosage has been 5,000 units 2 hours before surgery and 5,000 units every 8 to 12 hours thereafter for seven days or until the patient is fully ambulatory, whichever is longer. The heparin is given by deep subcutaneous injection in the arm or abdomen with a fine needle (25 to 26 gauge) to minimize tissue trauma. A concentrated solution of heparin sodium is recommended. Such prophylaxis should be reserved for patients over the age of 40 who are undergoing major surgery. Patients with bleeding disorders and those having neurosurgery, spinal anesthesia, eye surgery or potentially sanguineous operations should be excluded, as well as patients receiving oral anticoagulants or platelet-active drugs (see WARNINGS ). The value of such prophylaxis in hip surgery has not been established. The possibility of increased bleeding during surgery or postoperatively should be borne in mind. If such bleeding occurs, discontinuance of heparin and neutralization with protamine sulfate are advisable. If clinical evidence of thromboembolism develops despite low-dose prophylaxis, full therapeutic doses of anticoagulants should be given unless contraindicated. All patients should be screened prior to heparinization to rule out bleeding disorders, and monitoring should be performed with appropriate coagulation tests just prior to surgery. Coagulation test values should be normal or only slightly elevated. There is usually no need for daily monitoring of the effect of low-dose heparin in patients with normal coagulation parameters.
Extracorporeal Dialysis
Follow equipment manufacturers’ operating directions carefully.
Blood Transfusion
Addition of 400 to 600 USP units per 100 mL of whole blood is usually employed to prevent coagulation. Usually, 7,500 USP units of heparin sodium are added to 100 mL of 0.9% Sodium Chloride Injection, USP (or 75,000 USP units/1,000 mL of 0.9% Sodium Chloride Injection, USP) and mixed; from this sterile solution, 6 to 8 mL are added per 100 mL of whole blood.
Laboratory Samples
Addition of 70 to 150 units of heparin sodium per 10 to 20 mL sample of whole blood is usually employed to prevent coagulation of the sample. Leukocyte counts should be performed on heparinized blood within two hours after addition of the heparin. Heparinized blood should not be used for isoagglutinin, complement, or erythrocyte fragility tests or platelet counts.
CONTRAINDICATIONS
Heparin sodium should NOT be used in patients with the following conditions:
Severe thrombocytopenia;
When suitable blood coagulation tests, e.g., the whole blood clotting time, partial thromboplastin time, etc., cannot be performed at appropriate intervals (this contraindication refers to full-dose heparin; there is usually no need to monitor coagulation parameters in patients receiving low-dose heparin);
An uncontrollable active bleeding state (see WARNINGS ), except when this is due to disseminated intravascular coagulation.
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Hemorrhage
Hemorrhage is the chief complication that may result from heparin therapy (see WARNINGS ). An overly prolonged clotting time or minor bleeding during therapy can usually be controlled by withdrawing the drug (see OVERDOSAGE ). It should be appreciated that gastrointestinal or urinary tract bleeding during anticoagulant therapy may indicate the presence of an underlying occult lesion. Bleeding can occur at any site but certain specific hemorrhagic complications may be difficult to detect:
(a) Adrenal hemorrhage, with resultant acute adrenal insufficiency, has occurred during anticoagulant therapy. Therefore, such treatment should be discontinued in patients who develop signs and symptoms of acute adrenal hemorrhage and insufficiency. Initiation of corrective therapy should not depend on laboratory confirmation of the diagnosis, since any delay in an acute situation may result in the patient’s death.
(b) Ovarian (corpus luteum) hemorrhage developed in a number of women of reproductive age receiving short- or long-term anticoagulant therapy. This complication, if unrecognized, may be fatal.
(c) Retroperitoneal hemorrhage.
Thrombocytopenia, Heparin-induced Thrombocytopenia (HIT) and Heparin-induced Thrombocytopenia and Thrombosis (HITT) and Delayed Onset of HIT and HITT
See WARNINGS .
Local Irritation
Local irritation, erythema, mild pain, hematoma or ulceration may follow deep subcutaneous (intrafat) injection of heparin sodium. These complications are much more common after intramuscular use, and such use is not recommended.
Hypersensitivity
Generalized hypersensitivity reactions have been reported, with chills, fever and urticaria as the most usual manifestations, and asthma, rhinitis, lacrimation, headache, nausea and vomiting, and anaphylactoid reactions, including shock, occurring more rarely. Itching and burning, especially on the plantar side of the feet, may occur (see WARNINGS and PRECAUTIONS ).
Certain episodes of painful, ischemic and cyanosed limbs have in the past been attributed to allergic vasospastic reactions. Whether these are in fact identical to the thrombocytopenia-associated complications, remains to be determined.
Miscellaneous
Osteoporosis following long-term administration of high doses of heparin, cutaneous necrosis after systemic administration, suppression of aldosterone synthesis, delayed transient alopecia, priapism, and rebound hyperlipemia on discontinuation of heparin sodium have also been reported.
Significant elevations of aminotransferase (SGOT [S-AST] and SGPT [S-ALT]) levels have occurred in a high percentage of patients (and healthy subjects) who have received heparin.
DESCRIPTION
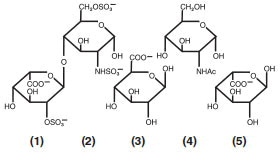
Heparin is a heterogeneous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans, having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are: (1) α-L-iduronic acid 2-sulfate, (2) 2-deoxy-2- sulfamino-α-D-glucose 6-sulfate, (3) β-D-glucuronic acid, (4) 2-acetamido-2-deoxy-α-D-glucose and (5) α-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2)> (1)> (4)> (3)> (5), and are joined by glycosidic linkages, forming polymers of varying sizes. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium, the acidic protons of the sulfate units are partially replaced by sodium ions.
Heparin Sodium Injection, USP is a sterile solution of heparin sodium derived from porcine intestinal mucosa, standardized for anticoagulant activity, in water for injection. It is to be administered by intravenous or deep subcutaneous routes. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram.
Structure of Heparin Sodium (representative subunits):
Heparin Sodium Injection, USP (porcine), preserved with parabens, is available as follows :
Each mL of the 1,000 units per mL preparation contains: 1,000 USP Heparin units (porcine); 9 mg sodium chloride; 1.5 mg methylparaben; 0.15 mg propylparaben; Water for Injection q.s. Made isotonic with sodium chloride. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (5.0 to 7.5).
Each mL of the 5,000 units per mL preparation contains: 5,000 USP Heparin units (porcine); 5 mg sodium chloride; 1.5 mg methylparaben; 0.15 mg propylparaben; Water for Injection q.s. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (5.0 to 7.5).
CLINICAL PHARMACOLOGY
Heparin inhibits reactions that lead to the clotting of blood and the formation of fibrin clots both in vitro and in vivo .Heparin acts at multiple sites in the normal coagulation system. Small amounts of heparin in combination with antithrombin III (heparin cofactor) can inhibit thrombosis by inactivating activated Factor X and inhibiting the conversion of prothrombin to thrombin. Once active thrombosis has developed, larger amounts of heparin can inhibit further coagulation by inactivating thrombin and preventing the conversion of fibrinogen to fibrin. Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilizing factor.
Bleeding time is usually unaffected by heparin. Clotting time is prolonged by full therapeutic doses of heparin; in most cases, it is not measurably affected by low doses of heparin.
Patients over 60 years of age, following similar doses of heparin, may have higher plasma levels of heparin and longer activated partial thromboplastin times (APTTs) compared with patients under 60 years of age.
Peak plasma levels of heparin are achieved two to four hours following subcutaneous administration, although there are considerable individual variations. Loglinear plots of heparin plasma concentrations with time, for a wide range of dose levels, are linear, which suggests the absence of zero order processes. Liver and the reticuloendothelial system are the sites of biotransformation. The biphasic elimination curve, a rapidly declining alpha phase (t 1⁄2 = 10 minutes) and after the age of 40 a slower beta phase, indicates uptake in organs. The absence of a relationship between anticoagulant half-life and concentration half-life may reflect factors such as protein binding of heparin.
Heparin does not have fibrinolytic activity; therefore, it will not lyse existing clots.
HOW SUPPLIED
Heparin Sodium Injection, USP (porcine) contains parabens and is available as follows:
Product Code | Unit of Sale | Strength | Each |
NP504011 | NDC 63323-540-57 Unit of 25 | 10,000 USP units per 10 mL (1,000 USP units per mL) | NDC 63323-540-57 10 mL multiple dose, flip-top vial |
NP926201• | NDC 63323-262-55 Unit of 25 | 5,000 USP units per mL | NDC 63323-262-55 1 mL fill in a 3 mL multiple dose, flip-top vial |
NP504015 | NDC 63323-540-67 Unit of 25 | 10,000 USP units per 10 mL (1,000 USP units per mL) | NDC 63323-540-07 10 mL multiple dose, flip-top vial |
NP926206 | NDC 63323-262-26 Unit of 25 | 5,000 USP units per mL | NDC 63323-262-09 1 mL fill in a 2 mL multiple dose, flip-top vial |
•Packaged in a plastic vial.
Use only if solution is clear and seal intact.
Do not use if solution is discolored or contains a precipitate.
This container closure is not made from natural rubber latex.