Get your patient on Hydroxyzine Hydrochloride - Hydroxyzine Hydrochloride tablet, Film Coated (Hydroxyzine Hydrochloride)
Hydroxyzine Hydrochloride - Hydroxyzine Hydrochloride tablet, Film Coated prescribing information
INDICATIONS AND USAGE
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested.
Useful in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses and in histamine-mediated pruritus.
As a sedative when used as a premedication and following general anesthesia, hydroxyzine may potentiate meperidine and barbiturates, so their use in pre-anesthetic adjunctive therapy should be modified on an individual basis. Atropine and other belladonna alkaloids are not affected by the drug. Hydroxyzine is not known to interfere with the action of digitalis in any way and it may be used concurrently with this agent.
The effectiveness of hydroxyzine as an antianxiety agent for long term use, that is more than 4 months, has not been assessed by systematic clinical studies. The physician should reassess periodically the usefulness of the drug for the individual patient.
DOSAGE AND ADMINISTRATION
For symptomatic relief of anxiety and tension associated with psychoneurosis and as an adjunct in organic disease states in which anxiety is manifested: adults, 50 to 100 mg q.i.d.; children under 6 years, 50 mg daily in divided doses; children over 6 years, 50 to 100 mg daily in divided doses.
For use in the management of pruritus due to allergic conditions such as chronic urticaria and atopic and contact dermatoses and in histamine-mediated pruritus: adults, 25 mg t.i.d. or q.i.d.; children under 6 years, 50 mg daily in divided doses; children over 6 years, 50 to 100 mg daily in divided doses.
As a sedative when used as a premedication and following general anesthesia: 50 to 100 mg for adults and 0.6 mg/kg of body weight in children.
When treatment is initiated by the intramuscular route of administration, subsequent doses may be administered orally.
As with all potent medication, the dosage should be adjusted according to the patient's response to therapy.
CONTRAINDICATIONS
Oral hydroxyzine hydrochloride is contraindicated in patients with known hypersensitivity to hydroxyzine hydrochloride products, and in patients with known hypersensitivity to cetirizine hydrochloride or levocetirizine hydrochloride.
Hydroxyzine is contraindicated in patients with a prolonged QT interval.
Hydroxyzine, when administered to the pregnant mouse, rat, and rabbit induced fetal abnormalities in the rat and mouse at doses substantially above the human therapeutic range. Clinical data in human beings are inadequate to establish safety in early pregnancy. Until such data are available, hydroxyzine is contraindicated in early pregnancy.
Hydroxyzine is contraindicated for patients who have shown a previous hypersensitivity to any component of this medication.
ADVERSE REACTIONS
Side effects reported with the administration of hydroxyzine hydrochloride are usually mild and transitory in nature.
Anticholinergic: Dry mouth.
Central Nervous System: Drowsiness is usually transitory and may disappear in a few days of continued therapy or upon reduction of dose. Involuntary motor activity including rare instances of tremor and convulsions have been reported, usually with doses considerably higher than those recommended. Clinically significant respiratory depression has not been reported at recommended doses.
Cardiac System: QT prolongation, Torsade de Pointes.
In postmarketing experience, the following additional undesirable effects have been reported:
Body as a Whole: Allergic reaction.
Nervous System: Headache.
Psychiatric: Hallucination.
Skin and Appendages: Oral hydroxyzine hydrochloride is associated with Acute Generalized Exanthematous Pustulosis (AGEP) and fixed drug eruptions in postmarketing reports.
Pruritus, rash, urticaria.
DESCRIPTION
Hydroxyzine hydrochloride, USP has the chemical name of 2-[2-[4-( p -Chloro-α-phenylbenzyl)-1-piperazinyl]ethoxy]ethanol dihydrochloride.
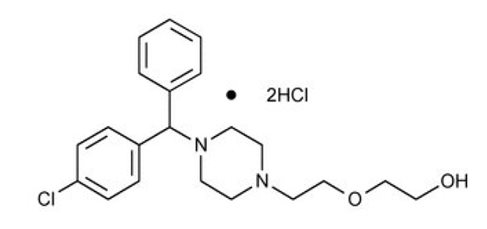
C 21 H 27 CIN 2 O 2 ·2HCl
M.W. 447.83
Hydroxyzine hydrochloride, USP occurs as a white, odorless powder which is very soluble in water.
Each tablet for oral administration contains 10 mg, 25 mg or 50 mg hydroxyzine hydrochloride, USP. Inactive ingredients include carnauba wax, colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate, microcrystalline cellulose, D&C Yellow #10 Aluminum Lake (25 mg and 50 mg), FD&C Blue #2 Aluminum Lake (25 mg), FD&C Red #40 Aluminum Lake (50 mg), FD&C Yellow #6 Aluminum Lake (10 mg and 50 mg), hypromellose, polyethylene glycol 3350, polyvinyl alcohol, talc, titanium dioxide, triacetin and yellow iron oxide (10 mg).
CLINICAL PHARMACOLOGY
Hydroxyzine hydrochloride is unrelated chemically to the phenothiazines, reserpine, meprobamate or the benzodiazepines. Hydroxyzine is not a cortical depressant, but its action may be due to a suppression of activity in certain key regions of the subcortical area of the central nervous system.
Primary skeletal muscle relaxation has been demonstrated experimentally. Bronchodilator activity, and antihistaminic and analgesic effects have been demonstrated experimentally and confirmed clinically. An antiemetic effect, both by the apomorphine test and the veriloid test, has been demonstrated.
Pharmacological and clinical studies indicate that hydroxyzine in therapeutic dosage does not increase gastric secretion or acidity and in most cases has mild antisecretory activity.
Hydroxyzine is rapidly absorbed from the gastrointestinal tract and hydroxyzine’s clinical effects are usually noted within 15 to 30 minutes after oral administration.
HOW SUPPLIED
Hydroxyzine Hydrochloride Tablets USP, 10 mg are available as 9/32", Orange round biconvex film-coated tablets, debossed “Є” above “159” on one side and plain on the other side, packaged in bottles of 500’s, NDC 24658-781-05 and in bottles of 1000’s, NDC 24658-781-10.
Hydroxyzine Hydrochloride Tablets USP, 25 mg are available as 10/32", Green round biconvex film-coated tablets debossed “Є” above “160” on one side and plain on the other side, packaged in bottles of 1000’s, NDC 24658-782-10.
Hydroxyzine Hydrochloride Tablets USP, 50 mg are available as 11/32", Yellow round biconvex film-coated tablets, debossed “Є” above “161” on one side and plain on the other side, packaged in bottles of 1000’s, NDC 24658-783-10.
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure, as required.
Store at 20°-25°C (68°-77°F) [see USP Controlled Room Temperature].
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Distributed by:
PuraCap Laboratories, LLC
DBA Blu Pharmaceuticals
Greenvale, NY 11548
Manufactured in USA
Rev. 09-2024-00
MF159BLUREV09/24
OS0007