Indium Dtpa In 111- Indium In-111 Pentetate Disodium solution prescribing information
INDICATIONS AND USAGE
Pentetate Indium Disodium In 111 is recommended for use in radionuclide cisternography.
DOSAGE AND ADMINISTRATION
Extreme care must be exercised to assure aseptic conditions in intrathecal injections.
The maximum recommended intrathecal dose in the average patient (70 kg) is 18.5 MBq, 500 µ Ci. The patient dose should be measured by a suitable radioactivity calibration system immediately prior to administration.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
CONTRAINDICATIONS
None known.
ADVERSE REACTIONS
Aseptic meningitis and pyrogenic reactions have been rarely (less than 0.4%) observed following cisternography with Pentetate lndium Disodium In 111.
One death has been reported to have occurred within 20 minutes following the administration of Pentetate Indium Disodium In 111 and appears to be drug related. In addition, two cases of septic meningitis have also been reported. There have also been reports of skin reactions and vomiting following administration of Pentetate lndium Disodium In 111. Relationship of the drug to these latter occurrences has not been established.
DESCRIPTION
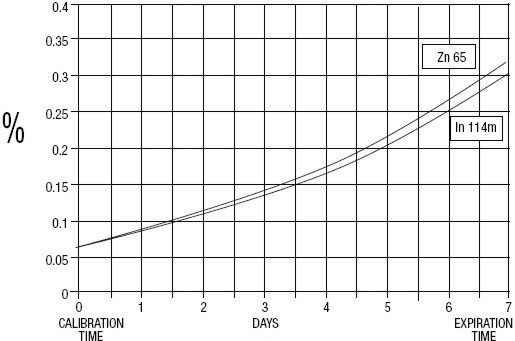
GE Healthcare (Medi-Physics, Inc.) Indium DTPA In 111 is a diagnostic drug for intrathecal use. It is available as a sterile, pyrogen-free, isotonic, aqueous solution, buffered to pH 7 to 8. At calibration time, each milliliter contains 37 MBq, 1 mCi of Pentetate Indium Disodium In 111 (no-carrier-added), 20 to 50 µ g of pentetic acid, and sodium bicarbonate for pH adjustment. The drug is to be discarded after single use.Radionuclidic purity at calibration time is at least 99.88% with less than 0.06% Indium In 114m and 0.06% Zinc Zn 65. The concentration of each radionuclidic contaminant changes with time. Graph 1 shows maximum concentration of each radionuclidic impurity as a function of time.
Graph 1 - Radionuclidic Impurities
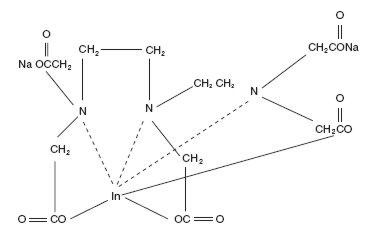
The chemical names are 1. Indate(2-)- 111 In -[ N,N -bis[2-[bis-(carboxymethyl)amino]ethyl]glycinato(5-)]-disodium; and 2. Disodium [ N,N -bis[2-(carboxymethyl)amino]glycinato(5-)]-indate (2-) 111 In.
Molecular formula: C 14 H 18 O 10 N 3 111 In Na 2
Molecular weight: 545.29
Structural formula:
PHYSICAL CHARACTERISTICS
Indium 111 decays by electron capture with a physical half-life of 67.9 hours.The energies of the photons that are useful for detection and imaging studies are listed in Table 1.
| Radiation | Mean %/Disintegration | Mean Energy (keV) |
|---|---|---|
| Gamma-2 | 90.2 | 171.3 |
| Gamma-3 | 94.0 | 245.4 |
EXTERNAL RADIATION
The specific gamma ray constant for Indium In 111 is 3.3 R/hr-mCi at 1 cm. The half-value thickness of lead (Pb) for Indium In 111 is 0.021 cm. To facilitate control of the radiation exposure from millicurie amounts of this radionuclide, a range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 2. For example, the use of a 0.8 cm thickness of Pb will attenuate the radiation emitted by a factor of about 1,000.
| Shield Thickness (Pb) cm | Coefficient of Attenuation |
|---|---|
| To correct for physical decay of this radionuclide, the fractions that remain at selected time intervals after the time of calibration are shown in Table 3. | |
| 0.021 | 0.5 |
| 0.19 | 10 -1 |
| 0.49 | 10 -2 |
| 0.80 | 10 -3 |
| 1.1 | 10 -4 |
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0 Calibration Time | 1.000 | 84 | 0.424 |
| 12 | 0.885 | 96 | 0.375 |
| 24 | 0.783 | 108 | 0.332 |
| 36 | 0.693 | 120 | 0.294 |
| 48 | 0.613 | 132 | 0.260 |
| 60 | 0.542 | 144 | 0.230 |
| 72 | 0.480 | 168 | 0.180 |
CLINICAL PHARMACOLOGY
After intrathecal administration, the radiopharmaceutical is absorbed from the subarachnoid space as described below, and the remainder flows superiorly to the basal cisterns within 2 to 4 hours and subsequently will be apparent in the Sylvian cisterns, the interhemispheric cisterns, and over the cerebral convexities. In normal individuals, the radiopharmaceutical will have ascended to the parasagittal region within 24 hours with simultaneous partial or complete clearance of activity from the basal cisterns and Sylvian regions. In contrast to air, the radiopharmaceutical does not normally enter the cerebral ventricles.
Although the primary absorption of cerebrospinal fluid (CSF) into the blood stream occurs at the arachnoid villi, there is some evidence that a significant fraction of CSF is also absorbed across both the cerebral and spinal leptomeninges. Lesser quantities may also be absorbed across the ventricular ependyma. It is also generally held that these alternate routes of CSF absorption may assume primary importance when the major routes of the flow are pathologically obstructed. Approximately 65% of the administered dose is excreted by the kidneys within 24 hours and this increases to 85% in 72 hours.
HOW SUPPLIED
Pentetate Indium Disodium In 111 (no-carrier-added) is supplied in single-dose glass vials, each containing 1.5 mL of solution with a concentration of 37 MBq, 1 mCi per mL and a total activity of 55.5 MBq, 1.5 mCi per vial at calibration time. Vials are packaged in individual lead shields with plastic outer containers.
NDC 17156-251-08
Storage
Store vial in its lead shield at a temperature of 5°-30°C (41°-86°F). Do not freeze.
Disposal
The residual materials may be discarded in ordinary trash provided the vials and syringes read no greater than background with an appropriate low-range survey meter. All identifying labels should be destroyed before discarding.
This radiopharmaceutical is licensed by Illinois Emergency Management Agency for distribution to persons licensed pursuant to 32 III. Adm. Code 330.260(a) and Part 335, Subpart E, 335.4010, or under equivalent licenses of an Agreement State or a Licensing State.