Get your patient on Ipratropium Bromide - Ipratropium Bromide solution (Ipratropium Bromide)
Ipratropium Bromide - Ipratropium Bromide solution prescribing information
INDICATIONS AND USAGE
Ipratropium Bromide Inhalation Solution administered either alone or with other bronchodilators, especially beta adrenergics, is indicated as a bronchodilator for maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease, including chronic bronchitis and emphysema.
DOSAGE AND ADMINISTRATION
The usual dosage of ipratropium bromide inhalation solution is 500 mcg (1 Unit-Dose Vial) administered three to four times a day by oral nebulization, with doses 6 to 8 hours apart. Ipratropium bromide inhalation solution unit-dose vials contain 500 mcg ipratropium bromide, USP anhydrous in 2.5 mL normal saline. Ipratropium bromide inhalation solution can be mixed in the nebulizer with albuterol or metaproterenol if used within one hour. Drug stability and safety of Ipratropium Bromide Inhalation Solution when mixed with other drugs in a nebulizer have not been established.
CONTRAINDICATIONS
Ipratropium bromide is contraindicated in known or suspected cases of hypersensitivity to ipratropium bromide, or to atropine and its derivatives.
ADVERSE REACTIONS
Adverse reaction information concerning ipratropium bromide inhalation solution is derived from 12-week active-controlled clinical trials. Additional information is derived from foreign post-marketing experience and the published literature.
All adverse events, regardless of drug relationship, reported by three percent or more patients in the 12-week controlled clinical trials appear in the table below.
Additional adverse reactions reported in less than three percent of the patients treated with ipratropium bromide include tachycardia, palpitations, eye pain, urinary retention, urinary tract infection and urticaria. Cases of precipitation or worsening of narrow-angle glaucoma, mydriasis, and acute eye pain have been reported.
Lower respiratory adverse reactions (bronchitis, dyspnea and bronchospasm) were the most common events leading to discontinuation of ipratropium bromide therapy in the 12-week trials. Headache, mouth dryness and aggravation of COPD symptoms are more common when the total daily dose of ipratropium bromide equals or exceeds 2,000 mcg.
Allergic-type reactions such as skin-rash, angioedema of tongue, lips and face, urticaria, laryngospasm and anaphylactic reaction have been reported. Many of the patients had a history of allergies to other drugs and/or foods.
| PERCENT OF PATIENTS | |||||
|---|---|---|---|---|---|
| Ipratropium | Metaproterenol | Ipratropium/ Metaproterenol | Albuterol | Ipratropium/ Albuterol | |
| (500 mcg t.i.d.) | (15 mg t.i.d.) | (500 mcg t.i.d./ 15 mg t.i.d.) | (2.5 mg t.i.d.) | (500 mcg t.i.d./ 2.5 mg t.i.d.) | |
| n = 219 | n = 212 | n = 108 | n = 205 | n = 100 | |
Body as a Whole-General Disorders | |||||
Headache | 6.4 | 5.2 | 6.5 | 6.3 | 9.0 |
Pain | 4.1 | 3.3 | 0.9 | 2.9 | 5.0 |
Influenza-like symptoms | 3.7 | 4.7 | 6.5 | 0.5 | 1.0 |
Back Pain | 3.2 | 1.9 | 1.9 | 2.4 | 0.0 |
Chest Pain | 3.2 | 4.2 | 5.6 | 2.0 | 1.0 |
Cardiovascular Disorders | |||||
Hypertension/Hypertension Aggravated | 0.9 | 1.9 | 0.9 | 1.5 | 4.0 |
Central & Peripheral Nervous System | |||||
Dizziness | 2.3 | 3.3 | 1.9 | 3.9 | 4.0 |
Insomnia | 0.9 | 0.5 | 4.6 | 1.0 | 1.0 |
Tremor | 0.9 | 7.1 | 8.3 | 1.0 | 0.0 |
Nervousness | 0.5 | 4.7 | 6.5 | 1.0 | 1.0 |
Gastrointestinal System Disorders | |||||
Mouth Dryness | 3.2 | 0.0 | 1.9 | 2.0 | 3.0 |
Nausea | 4.1 | 3.8 | 1.9 | 2.9 | 2.0 |
Constipation | 0.9 | 0.0 | 3.7 | 1.0 | 1.0 |
Musculo-skeletal System Disorders | |||||
Arthritis | 0.9 | 1.4 | 0.9 | 0.5 | 3.0 |
Respiratory System Disorders (Lower) | |||||
Coughing | 4.6 | 8.0 | 6.5 | 5.4 | 6.0 |
Dyspnea | 9.6 | 13.2 | 16.7 | 12.7 | 9.0 |
Bronchitis | 14.6 | 24.5 | 15.7 | 16.6 | 20.0 |
Bronchospasm | 2.3 | 2.8 | 4.6 | 5.4 | 5.0 |
Sputum Increased | 1.4 | 1.4 | 4.6 | 3.4 | 0.0 |
Respiratory Disorder | 0.0 | 6.1 | 6.5 | 2.0 | 4.0 |
Respiratory System Disorders (Upper) | |||||
Upper Respiratory Tract Infection | 13.2 | 11.3 | 9.3 | 12.2 | 16.0 |
Pharyngitis | 3.7 | 4.2 | 5.6 | 2.9 | 4.0 |
Rhinitis | 2.3 | 4.2 | 1.9 | 2.4 | 0.0 |
Sinusitus | 2.3 | 2.8 | 0.9 | 5.4 | 4.0 |
Drug Interactions
Ipratropium bromide has been shown to be a safe and effective bronchodilator when used in conjunction with beta adrenergic bronchodilators. Ipratropium bromide has also been used with other pulmonary medications, including methylxanthines and corticosteroids, without adverse drug interactions.
DESCRIPTION
The active ingredient, ipratropium bromide monohydrate, USP, is an anticholinergic bronchodilator chemically described as 8-azoniabicyclo [3.2.1]- octane, 3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-(1-methylethyl)-, bromide, monohydrate (endo, syn)-, (±)-; a synthetic quaternary ammonium compound, chemically related to atropine.
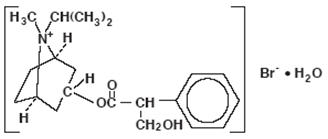
Ipratropium Bromide Monohydrate C 20 H 30 BrNO 3 •H 2 O Mol. Wt. 430.4
Ipratropium bromide is a white crystalline substance, freely soluble in water and lower alcohols. It is a quaternary ammonium compound and thus exists in an ionized state in aqueous solutions. It is relatively insoluble in non-polar media.
Ipratropium Bromide Inhalation Solution is administered by oral inhalation with the aid of a nebulizer. It contains ipratropium bromide, USP 0.02% (anhydrous basis) in a sterile, preservative-free, isotonic saline solution, pH-adjusted to 3.4 (3 to 4) with hydrochloric acid.
CLINICAL PHARMACOLOGY
Ipratropium bromide is an anticholinergic (parasympatholytic) agent that, based on animal studies, appears to inhibit vagally mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released from the vagus nerve.
Anticholinergics prevent the increases in intracellular concentration of cyclic guanosine monophosphate (cyclic GMP) that are caused by interaction of acetylcholine with the muscarinic receptor on bronchial smooth muscle.
The bronchodilation following inhalation of ipratropium bromide is primarily a local, site-specific effect, not a systemic one. Much of an administered dose is swallowed but not absorbed, as shown by fecal excretion studies. Following nebulization of a 2 mg dose, a mean 7% of the dose was absorbed into the systemic circulation either from the surface of the lung or from the gastrointestinal tract. The half-life of elimination is about 1.6 hours after intravenous administration. Ipratropium bromide is minimally (0 to 9% in vitro ) bound to plasma albumin and a 1 -acid glycoproteins. It is partially metabolized. Autoradiographic studies in rats have shown that ipratropium bromide does not penetrate the blood-brain barrier. Ipratropium bromide has not been studied in patients with hepatic or renal insufficiency. It should be used with caution in those patient populations.
In controlled 12-week studies in patients with bronchospasm associated with chronic obstructive pulmonary disease (chronic bronchitis and emphysema) significant improvements in pulmonary function (FEV 1 increases of 15% or more) occurred within 15 to 30 minutes, reached a peak in 1 to 2 hours, and persisted for periods of 4 to 5 hours in the majority of patients, with about 25% to 38% of the patients demonstrating increases of 15% or more for at least 7 to 8 hours. Continued effectiveness of ipratropium bromide inhalation solution was demonstrated throughout the 12-week period. In addition, significant increases in forced vital capacity (FVC) have been demonstrated. However, ipratropium bromide did not consistently produce significant improvement in subjective symptom scores nor in quality of life scores over the 12-week duration of study.
Additional controlled 12-week studies were conducted to evaluate the safety and effectiveness of ipratropium bromide inhalation solution administered concomitantly with the beta adrenergic bronchodilator solutions metaproterenol and albuterol compared with the administration of each of the beta agonists alone. Combined therapy produced significant additional improvement in FEV 1 and FVC. On combined therapy, the median duration of 15% improvement in FEV 1 was 5 to 7 hours, compared with 3 to 4 hours in patients receiving a beta agonist alone.
HOW SUPPLIED
Ipratropium Bromide Inhalation Solution Unit Dose Vial is supplied as a 0.02% clear, colorless solution containing 2.5 mL.
NDC 60687-394-83, 30 vials per carton / 1 vial per foil pouch
Each vial is made from a low density polyethylene (LDPE) resin.
Vials are supplied in a foil pouch.
Store between 59°F (15°C) and 86°F (30°C).
Protect from light.
Store unused vials in the foil pouch.