Isosorbide Mononitrate - Isosorbide Mononitrate tablet, Extended Release prescribing information
INDICATIONS AND USAGE
Isosorbide Mononitrate Extended-Release Tablets are indicated for the prevention of angina pectoris due to coronary artery disease. The onset of action of oral isosorbide mononitrate is not sufficiently rapid for this product to be useful in aborting an acute anginal episode.
DOSAGE AND ADMINISTRATION
The recommended starting dose of Isosorbide Mononitrate Extended-Release Tablets is 30 mg (given as a single 30 mg tablet or as 1/2 of a 60 mg tablet) or 60 mg (given as a single tablet) once daily. After several days, the dosage may be increased to 120 mg (given as a single 120 mg tablet or as two 60 mg tablets) once daily. Rarely, 240 mg may be required. The daily dose of Isosorbide Mononitrate Extended-Release Tablets should be taken in the morning on arising. Isosorbide Mononitrate Extended-Release Tablets should not be chewed or crushed and should be swallowed together with a half-glassful of fluid. Do not break the 30 mg tablet.
CONTRAINDICATIONS
Isosorbide Mononitrate Extended-Release Tablets are contraindicated in patients who have shown hypersensitivity or idiosyncratic reactions to other nitrates or nitrites.
ADVERSE REACTIONS
The table below shows the frequencies of the adverse events that occurred in >5% of the subjects in three placebo-controlled North American studies, in which patients in the active treatment arm received 30 mg, 60 mg, 120 mg, or 240 mg of Isosorbide Mononitrate Extended-Release Tablets once daily. In parentheses, the same table shows the frequencies with which these adverse events were associated with the discontinuation of treatment. Overall, 8% of the patients who received 30 mg, 60 mg, 120 mg, or 240 mg of isosorbide mononitrate in the three placebo-controlled North American studies discontinued treatment because of adverse events. Most of these discontinued because of headache. Dizziness was rarely associated with withdrawal from these studies. Since headache appears to be a dose-related adverse effect and tends to disappear with continued treatment, it is recommended that ISMN treatment be initiated at low doses for several days before being increased to desired levels.
FREQUENCY AND ADVERSE EVENTS (DISCONTINUED) a
Three Controlled North American Studies | |||||||
Dose | Placebo | 30 mg | 60 mg | 120 mg• | 240 mg• | ||
Patients | 96 | 60 | 102 | 65 | 65 | ||
Headache | 15% (0%) | 38% (5%) | 51% (8%) | 42% (5%) | 57% (8%) | ||
Dizziness | 4% (0%) | 8% (0%) | 11% (1%) | 9% (2%) | 9% (2%) | ||
a Some individuals for multiple reasons. • Patients were started on 60 mg and titrated to their final dose.
In addition, the three North American trials were pooled with 11 controlled trials conducted in Europe. Among the 14 controlled trials, a total of 711 patients were randomized to Isosorbide Mononitrate Extended-Release Tablets. When the pooled data were reviewed, headache and dizziness were the only adverse events that were reported by >5% of patients. Other adverse events, each reported by ≤5% of exposed patients, and in many cases of uncertain relation to drug treatment, were:
Autonomic Nervous System Disorders: Dry mouth, hot flushes.
Body as a Whole: Asthenia, back pain, chest pain, edema, fatigue, fever, flu-like symptoms, malaise, rigors.
Cardiovascular Disorders, General: Cardiac failure, hypertension, hypotension.
Central and Peripheral Nervous System Disorders: Dizziness, headache, hypoesthesia, migraine, neuritis, paresis, paresthesia, ptosis, tremor, vertigo.
Gastrointestinal System Disorders: Abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gastric ulcer, gastritis, glossitis, hemorrhagic gastric ulcer, hemorrhoids, loose stools, melena, nausea, vomiting.
Hearing and Vestibular Disorders: Earache, tinnitus, tympanic membrane perforation.
Heart Rate and Rhythm Disorders: Arrhythmia, arrhythmia atrial, atrial fibrillation, bradycardia, bundle branch block, extrasystole, palpitation, tachycardia, ventricular tachycardia.
Liver and Biliary System Disorders: SGOT increase, SGPT increase.
Metabolic and Nutritional Disorders: Hyperuricemia, hypokalemia.
Musculoskeletal System Disorders: Arthralgia, frozen shoulder, muscle weakness, musculoskeletal pain, myalgia, myositis, tendon disorder, torticollis.
Myo-, Endo-, Pericardial and Valve Disorders: Angina pectoris aggravated, heart murmur, heart sound abnormal, myocardial infarction, Q wave abnormality.
Platelet, Bleeding and Clotting Disorders: Purpura, thrombocytopenia.
Psychiatric Disorders: Anxiety, concentration impaired, confusion, decreased libido, depression, impotence, insomnia, nervousness, paroniria, somnolence.
Red Blood Cell Disorder: Hypochromic anemia.
Reproductive Disorders, Female: Atrophic vaginitis, breast pain.
Resistance Mechanism Disorders: Bacterial infection, moniliasis, viral infection.
Respiratory System Disorders: Bronchitis, bronchospasm, coughing, dyspnea, increased sputum, nasal congestion, pharyngitis, pneumonia, pulmonary infiltration, rales, rhinitis, sinusitis.
Skin and Appendages Disorders: Acne, hair texture abnormal, increased sweating, pruritus, rash, skin nodule.
Urinary System Disorders: Polyuria, renal calculus, urinary tract infection.
Vascular (Extracardiac) Disorders: Flushing, intermittent claudication, leg ulcer, varicose vein.
Vision Disorders: Conjunctivitis, photophobia, vision abnormal.
In addition, the following spontaneous adverse event has been reported during the marketing of isosorbide mononitrate: syncope.
To report SUSPECTED ADVERSE REACTIONS, contact Chartwell RX, LLC. at 1-845-232-1683 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Isosorbide mononitrate (ISMN), an organic nitrate and the major biologically active metabolite of isosorbide dinitrate (ISDN), is a vasodilator with effects on both arteries and veins.
Each tablet, for oral administration, contains either 30 mg, 60 mg or 120 mg of isosorbide mononitrate in an extended-release formulation. In addition, ISMN 30 mg tablets, USP contains the following inactive ingredients: colloidal silicon dioxide, compressible sugar, hydroxypropyl methylcellulose, lactose monohydrate, magnesium stearate. ISMN 60 mg tablets, USP contains the following inactive ingredients: colloidal silicon dioxide, compressible sugar, hydroxypropyl methylcellulose, lactose monohydrate, magnesium stearate, yellow iron oxide. ISMN 120 mg tablets, USP contains the following inactive ingredients: colloidal silicon dioxide, hydroxypropyl cellulose, hypromellose, lactose monohydrate, magnesium stearate and talc.
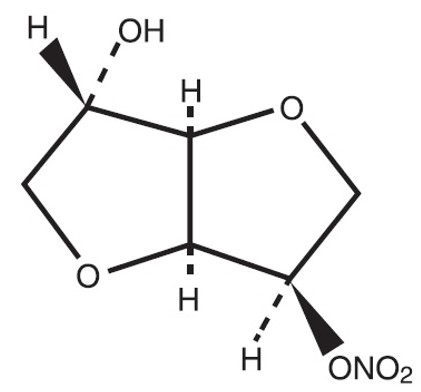
The molecular formula of ISMN is C 6 H 9 NO 6 and the molecular weight is 191.14. The chemical name for ISMN is: 1,4:3,6-dianhydro-,D-glucitol 5-nitrate; the compound has the following structural formula:
ISMN is a white, crystalline, odorless compound which is stable in air and in solution, has a melting point of about 90°C, and an optical rotation of +144° (2% in water, 20°C).
Isosorbide mononitrate is freely soluble in water, ethanol, methanol, chloroform, ethyl acetate, and dichloromethane. Isosorbide Mononitrate Extended-Release 120 mg Tablets, USP meets USP Dissolution Test 7.
FDA approved dissolution acceptance criteria for Isosorbide Mononitrate Extended-Release 30 mg and 60 mg Tablets, USP differ from that of the USP.
CLINICAL PHARMACOLOGY
Mechanism of Action
The Isosorbide Mononitrate Extended-Release Tablet is an oral extended-release formulation of ISMN, the major active metabolite of isosorbide dinitrate; most of the clinical activity of the dinitrate is attributable to the mononitrate.
The principal pharmacological action of ISMN and all organic nitrates in general is relaxation of vascular smooth muscle, producing dilatation of peripheral arteries and veins, especially the latter. Dilatation of the veins promotes peripheral pooling of blood, decreases venous return to the heart, thereby reducing left ventricular end-diastolic pressure and pulmonary capillary wedge pressure (preload). Arteriolar relaxation reduces systemic vascular resistance, systolic arterial pressure and mean arterial pressure (afterload). Dilatation of the coronary arteries also occurs. The relative importance of preload reduction, afterload reduction, and coronary dilatation remains undefined.
Pharmacodynamics
Dosing regimens for most chronically used drugs are designed to provide plasma concentrations that are continuously greater than a minimally effective concentration. This strategy is inappropriate for organic nitrates. Several well-controlled clinical trials have used exercise testing to assess the antianginal efficacy of continuously delivered nitrates. In the large majority of these trials, active agents were indistinguishable from placebo after 24 hours (or less) of continuous therapy. Attempts to overcome tolerance by dose escalation, even to doses far in excess of those used acutely, have consistently failed. Only after nitrates have been absent from the body for several hours has their antianginal efficacy been restored. Isosorbide Mononitrate Extended-Release Tablets, during long-term use over 42 days dosed at 120 mg once daily, continued to improve exercise performance at 4 hours and at 12 hours after dosing but its effects (although better than placebo) are less than or at best equal to the effects of the first dose of 60 mg.
Pharmacokinetics and Metabolism
After oral administration of ISMN as a solution or immediate-release tablets, maximum plasma concentrations of ISMN are achieved in 30 to 60 minutes, with an absolute bioavailability of approximately 100%. After intravenous administration, ISMN is distributed into total body water in about 9 minutes with a volume of distribution of approximately 0.6-0.7 L/kg. Isosorbide mononitrate is approximately 5% bound to human plasma proteins and is distributed into blood cells and saliva. Isosorbide mononitrate is primarily metabolized by the liver, but unlike oral isosorbide dinitrate, it is not subject to first-pass metabolism. Isosorbide mononitrate is cleared by denitration to isosorbide and glucuronidation as the mononitrate, with 96% of the administered dose excreted in the urine within 5 days and only about 1% eliminated in the feces. At least six different compounds have been detected in urine, with about 2% of the dose excreted as the unchanged drug and at least five metabolites. The metabolites are not pharmacologically active. Renal clearance accounts for only about 4% of total body clearance. The mean plasma elimination half-life of ISMN is approximately 5 hours.
The disposition of ISMN in patients with various degrees of renal insufficiency, liver cirrhosis, or cardiac dysfunction was evaluated and found to be similar to that observed in healthy subjects. The elimination half-life of ISMN was not prolonged, and there was no drug accumulation in patients with chronic renal failure after multiple oral dosing.
The pharmacokinetics and/or bioavailability of Isosorbide Mononitrate Extended-Release Tablets have been studied in both normal volunteers and patients following single- and multiple-dose administration. Data from these studies suggest that the pharmacokinetics of ISMN administered as Isosorbide Mononitrate Extended-Release Tablets are similar between normal healthy volunteers and patients with angina pectoris. In single- and multiple-dose studies, the pharmacokinetics of ISMN were dose proportional between 30 mg and 240 mg.
In a multiple-dose study, the effect of age on the pharmacokinetic profile of Isosorbide Mononitrate Extended-Release Tablets 60 mg and 120 mg (2 x 60 mg) was evaluated in subjects ≥45 years. The results of that study indicate that there are no significant differences in any of the pharmacokinetic variables of ISMN between elderly (≥65 years) and younger individuals (45 – 64 years) for the isosorbide mononitrate extended-release 60 mg dose. The administration of isosorbide mononitrate extended-release 120 mg (2 x 60 mg tablets every 24 hours for 7 days) produced a dose-proportional increase in C max and AUC, without changes in T max or the terminal half-life. The older group (65-74 years) showed 30% lower apparent oral clearance (Cl/F) following the higher dose, i.e., 120 mg, compared to the younger group (45-64 years); Cl/F was not different between the two groups following the 60 mg regimen. While Cl/F was independent of dose in the younger group, the older group showed slightly lower Cl/F following the 120 mg regimen compared to the 60 mg regimen. Differences between the two age groups, however, were not statistically significant. In the same study, females showed a slight (15%) reduction in clearance when the dose was increased. Females showed higher AUCs and C max compared to males, but these differences were accounted for by differences in body weight between the two groups. When the data were analyzed using age as a variable, the results indicated that there were no significant differences in any of the pharmacokinetic variables of ISMN between older (≥65 years) and younger individuals (45-64 years). The results of this study, however, should be viewed with caution due to the small number of subjects in each age subgroup and consequently the lack of sufficient statistical power.
The following table summarizes key pharmacokinetic parameters of ISMN after single- and multiple-dose administration of ISMN as an oral solution or Isosorbide Mononitrate Extended-Release Tablets:
SINGLE-DOSE STUDIES | MULTIPLE- DOSE STUDIES | |||
PARAMETER | ISMN 60 mg | ISMN Extended- Release Tablets 60 mg | ISMN Extended- Release Tablets 60 mg | ISMN Extended- Release Tablets 120 mg |
C max (ng/mL) | 1242 to 1534 | 424 to 541 | 575 to 572 | 1151 to 1180 |
T max (hr) | 0.6 to 0.7 | 3.1 to 4.5 | 2.9 to 4.2 | 3.1 to 3.2 |
AUC (ng . hr/ mL) | 8189 to 8313 | 5990 to 7452 | 6625 to 7555 | 14241 to 16800 |
t 1/2 (hr) | 4.8 to 5.1 | 6.3 to 6.6 | 6.2 to 6.3 | 6.2 to 6.4 |
CI/F (mL/ min) | 120 to 122 | 151 to 187 | 132 to 151 | 119 to 140 |
Food Effects
The influence of food on the bioavailability of ISMN after single-dose administration of Isosorbide Mononitrate Extended-Release Tablets 60 mg was evaluated in three different studies involving either a "light" breakfast or a high-calorie, high-fat breakfast. Results of these studies indicate that concomitant food intake may decrease the rate (increase in T max ) but not the extent (AUC) of absorption of ISMN.
Clinical Trials
Controlled trials with Isosorbide Mononitrate Extended-Release Tablets have demonstrated antianginal activity following acute and chronic dosing. Administration of Isosorbide Mononitrate Extended-Release Tablets once daily, taken early in the morning on arising, provided at least 12 hours of antianginal activity.
In a placebo-controlled parallel study, 30, 60, 120 and 240 mg of Isosorbide Mononitrate Extended-Release Tablets were administered once daily for up to 6 weeks. Prior to randomization, all patients completed a 1- to 3-week single-blind placebo phase to demonstrate nitrate responsiveness and total exercise treadmill time reproducibility. Exercise tolerance tests using the Bruce Protocol were conducted prior to and at 4 and 12 hours after the morning dose on days 1, 7, 14, 28 and 42 of the double-blind period. Isosorbide Mononitrate Extended-Release Tablets 30 and 60 mg (only doses evaluated acutely) demonstrated a significant increase from baseline in total treadmill time relative to placebo at 4 and 12 hours after the administration of the first dose. At day 42, the 120 and 240 mg dose of Isosorbide Mononitrate Extended-Release Tablets demonstrated a significant increase in total treadmill time at 4 and 12 hours post dosing, but by day 42, the 30 and 60 mg doses no longer were differentiable from placebo. Throughout chronic dosing, rebound was not observed in any isosorbide mononitrate extended-release treatment group.
Pooled data from two other trials, comparing Isosorbide Mononitrate Extended-Release Tablets 60 mg once daily, ISDN 30 mg QID, and placebo QID in patients with chronic stable angina using a randomized, double-blind, three-way crossover design found statistically significant increases in exercise tolerance times for Isosorbide Mononitrate Extended-Release Tablets compared to placebo at hours 4, 8 and 12 and to ISDN at hour 4. The increases in exercise tolerance on day 14, although statistically significant compared to placebo, were about half of that seen on day 1 of the trial.
HOW SUPPLIED
Isosorbide Mononitrate Extended-Release Tablets, USP 30 mg are White, biconvex oval shaped tablets, scored and embossed "30" on one side.
Bottles of 90 NDC 62135-586-90
Isosorbide Mononitrate Extended-Release Tablets, USP 60 mg are Light yellow, biconvex oval shaped tablets, scored on both sides and embossed ‘DX 31’ on one side.
Bottles of 90 NDC 62135-587-90
Isosorbide Mononitrate Extended-Release Tablets, USP 120 mg are white, biconvex, oval-shaped tablets, embossed with '120' on one side.
Bottles of 90 NDC 62135-588-90
Store at 20° to 25°C (68° to 77°F) (see USP Controlled Room Temperature). Protect from excessive moisture.
Manufactured by: Dexcel Ltd. 1 Dexcel St. Or-Akiva, 3060000, Israel.
Manufactured for: Chartwell RX, LLC. Congers, NY 10920
Revised : 04/2023
L71449