Get your patient on Ketoconazole - Ketoconazole cream (Ketoconazole)
Ketoconazole - Ketoconazole cream prescribing information
INDICATIONS AND USAGE
Ketoconazole cream, 2% is indicated for the topical treatment of tinea corporis, tinea cruris and tinea pedis caused by Trichophyton rubrum, T. mentagrophytes and Epidermophyton floccosum; in the treatment of tinea (pityriasis) versicolor caused by Malassezia furfur (Pityrosporum orbiculare); in the treatment of cutaneous candidiasis caused by Candida spp. and in the treatment of seborrheic dermatitis.
DOSAGE AND ADMINISTRATION
Cutaneous candidiasis, tinea corporis, tinea cruris, tinea pedis, and tinea (pityriasis) versicolor
It is recommended that ketoconazole cream, 2% be applied once daily to cover the affected and immediate surrounding area. Clinical improvement may be seen fairly soon after treatment is begun; however, candidal infections and tinea cruris and corporis should be treated for two weeks in order to reduce the possibility of recurrence.
Patients with tinea versicolor usually require two weeks of treatment. Patients with tinea pedis require six weeks of treatment.
Seborrheic dermatitis
Ketoconazole cream, 2% should be applied to the affected area twice daily for four weeks or until clinical clearing.
If a patient shows no clinical improvement after the treatment period, the diagnosis should be redetermined.
CONTRAINDICATIONS
Ketoconazole cream, 2% is contraindicated in persons who have shown hypersensitivity to the active or excipient ingredients of this formulation.
ADVERSE REACTIONS
During clinical trials 45 (5.0%) of 905 patients treated with ketoconazole cream, 2% and 5 (2.4%) of 208 patients treated with placebo reported side effects consisting mainly of severe irritation, pruritus and stinging. One of the patients treated with ketoconazole cream developed a painful allergic reaction.
In worldwide postmarketing experience, rare reports of contact dermatitis have been associated with ketoconazole cream or one of its excipients, namely propylene glycol.
To report SUSPECTED ADVERSE REACTIONS, contact Northstar RxLLC at 1-800-206-7821 and/or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Ketoconazole cream, 2% contains the broad-spectrum synthetic antifungal agent, ketoconazole 2%, formulated in an aqueous cream vehicle consisting of butylated hydroxyanisole (BHA), cetyl alcohol, isopropyl myristate, polysorbate 60, polysorbate 80, propylene glycol, purified water, sorbitan monostearate and stearyl alcohol.
Ketoconazole is cis -1-acetyl-4-[4-[[2-(2,4-dichlorophenyl)-2-(1 H -imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl] piperazine and has the following structural formula:
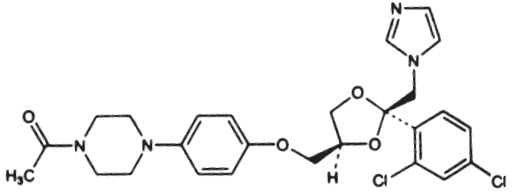
Molecular Formula: C 26 H 28 Cl 2 N 4 O 4
Molecular Weight: 531.43
CLINICAL PHARMACOLOGY
When ketoconazole cream, 2% was applied dermally to intact or abraded skin of beagle dogs for 28 consecutive days at a dose of 80 mg, there were no detectable plasma levels using an assay method having a lower detection limit of 2 ng/mL.
After a single topical application to the chest, back and arms of normal volunteers, systemic absorption of ketoconazole was not detected at the 5 ng/mL level in blood over a 72-hour period.
Two dermal irritancy studies, a human sensitization test, a phototoxicity study and a photoallergy study conducted in 38 male and 62 female volunteers showed no contact sensitization of the delayed hypersensitivity type, no irritation, no phototoxicity and no photoallergenic potential due to ketoconazole cream, 2%.
Microbiology
Ketoconazole is a broad spectrum synthetic antifungal agent which inhibits the in vitro growth of the following common dermatophytes and yeasts by altering the permeability of the cell membrane: dermatophytes: Trichophyton rubrum, T. mentagrophytes, T. tonsurans, Microsporum canis, M. audouini, M. gypseum and Epidermophyton floccosum; yeasts: Candida albicans, Malassezia ovale (Pityrosporum ovale) and C. tropicalis; and the organism responsible for tinea versicolor, Malassezia furfur (Pityrosporum orbiculare). Only those organisms listed in the INDICATIONS AND USAGE section have been proven to be clinically affected. Development of resistance to ketoconazole has not been reported.
Mode of Action
In vitro studies suggest that ketoconazole impairs the synthesis of ergosterol, which is a vital component of fungal cell membranes. It is postulated that the therapeutic effect of ketoconazole in seborrheic dermatitis is due to the reduction of M. ovale, but this has not been proven.
HOW SUPPLIED
Ketoconazole cream, 2% is supplied in 15 g (NDC 16714-955-01), 30 g (NDC 16714-955-02), and 60 g (NDC 16714-955-03) tubes.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].