Get your patient on Ketoprofen
Ketoprofen prescribing information
BOXED WARNING
Cardiovascular Thrombotic Events
• Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use.
• Ketoprofen capsules are contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Gastrointestinal Risk
• NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal (GI) events.
1 Throughout this package insert, the term NSAID refers to a non-aspirin non-steroidal anti-inflammatory drug.
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of ketoprofen capsules USP and other treatment options before deciding to use ketoprofen capsules USP. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals. Ketoprofen capsules USP are indicated for the management of the signs and symptoms of rheumatoid arthritis and osteoarthritis. Ketoprofen capsules USP are indicated for the management of pain. Ketoprofen capsules USP are also indicated for treatment of primary dysmenorrhea.
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of ketoprofen capsules and other treatment options before deciding to use ketoprofen capsules. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ). After observing the response to initial therapy with ketoprofen capsules, the dose and frequency should be adjusted to suit an individual patient's needs. Concomitant use of ketoprofen capsules and ketoprofen extended-release capsules is not recommended. If minor side effects appear, they may disappear at a lower dose which may still have an adequate therapeutic effect. If well tolerated but not optimally effective, the dosage may be increased. Individual patients may show a better response to 300 mg of ketoprofen capsules daily as compared to 200 mg, although in well-controlled clinical trials patients on 300 mg did not show greater mean effectiveness. They did, however, show an increased frequency of upper- and lower-GI distress and headaches. It is of interest that women also had an increased frequency of these adverse effects compared to men. When treating patients with 300 mg/day, the physician should observe sufficient increased clinical benefit to offset potential increased risk. In patients with mildly impaired renal function, the maximum recommended total daily dose of ketoprofen capsules is 150 mg. In patients with a more severe renal impairment (GFR less than 25 mL/min/1.73 m2 or end-stage renal impairment), the maximum total daily dose of ketoprofen capsules should not exceed 100 mg. In elderly patients, renal function may be reduced with apparently normal serum creatinine and/or BUN levels. Therefore, it is recommended that the initial dosage of ketoprofen capsules should be reduced for patients over 75 years of age (see Geriatric Use ). It is recommended that for patients with impaired liver function and serum albumin concentration less than 3.5 g/dL, the maximum initial total daily dose of ketoprofen capsules should be 100 mg. All patients with metabolic impairment, particularly those with both hypoalbuminemia and reduced renal function, may have increased levels of free (biologically active) ketoprofen and should be closely monitored. The dosage may be increased to the range recommended for the general population, if necessary, only after good individual tolerance has been ascertained. Because hypoalbuminemia and reduced renal function both increase the fraction of free drug (biologically active form), patients who have both conditions may be at greater risk of adverse effects. Therefore, it is recommended that such patients also be started on lower doses of ketoprofen capsules and closely monitored.
Rheumatoid Arthritis and Osteoarthritis The recommended starting dose of ketoprofen capsules in otherwise healthy patients is 75 mg three times or 50 mg four times a day. Smaller doses of ketoprofen capsules should be utilized initially in small individuals or in debilitated or elderly patients. The recommended maximum daily dose of ketoprofen capsules is 300 mg/day. Dosages higher than 300 mg/day of ketoprofen capsules are not recommended because they have not been studied. Concomitant use of ketoprofen capsules and ketoprofen extended-release capsules is not recommended. Relatively smaller people may need smaller doses. As with other non-steroidal anti-inflammatory drugs, the predominant adverse effects of ketoprofen are gastrointestinal. To attempt to minimize these effects, physicians may wish to prescribe that ketoprofen capsules be taken with antacids, food, or milk. Although food delays the absorption of ketoprofen capsules (see CLINICAL PHARMACOLOGY ), in most of the clinical trials ketoprofen was taken with food or milk. Physicians may want to make specific recommendations to patients about when they should take ketoprofen capsules in relation to food and/or what patients should do if they experience minor GI symptoms associated with ketoprofen capsules.
Management of Pain and Dysmenorrhea The usual dose of ketoprofen capsules recommended for mild-to-moderate pain and dysmenorrhea is 25 to 50 mg every 6 to 8 hours as necessary. A smaller dose should be utilized initially in small individuals, in debilitated or elderly patients, or in patients with renal or liver disease (see PRECAUTIONS ). A larger dose may be tried if the patient’s response to a previous dose was less than satisfactory, but doses above 75 mg have not been shown to give added analgesia. Daily doses above 300 mg are not recommended because they have not been adequately studied. Because of its typical non-steroidal anti-inflammatory drug-side-effect profile, including as its principal adverse effect GI side effects (see WARNINGS and ADVERSE REACTIONS ), higher doses of ketoprofen capsules should be used with caution and patients receiving them observed carefully.
Special Populations
Elderly
Clearance and unbound fraction The plasma and renal clearance of ketoprofen is reduced in the elderly (mean age, 73 years) compared to a younger normal population (mean age, 27 years). Hence, ketoprofen peak concentration and AUC increase with increasing age. In addition, there is a corresponding increase in unbound fraction with increasing age. Data from one trial suggest that the increase is greater in women than in men. It has not been determined whether age-related changes in absorption among the elderly contribute to the changes in bioavailability of ketoprofen. In a study conducted with young and elderly men and women, results for subjects older than 75 years of age showed that free drug AUC increased by 40% and Cmax increased by 60% as compared with estimates of the same parameters in young subjects (those younger than 35 years of age;. Also in the elderly, the ratio of intrinsic clearance/availability decreased by 35% and plasma half-life was prolonged by 26%. This reduction is thought to be due to a decrease in hepatic extraction associated with aging. Renally Impaired Studies of the effects of renal-function impairment have been small. They indicate a decrease in clearance in patients with impaired renal function. In 23 patients with renal impairment, free ketoprofen peak concentration was not significantly elevated, but free ketoprofen clearance was reduced from 15 L/kg/h for normal subjects to 7 L/kg/h in patients with mildly impaired renal function, and to 4 L/kg/h in patients with moderately to severely impaired renal function. The elimination t1/2 was prolonged from 1.6 hours in normal subjects to approximately 3 hours in patients with mild renal impairment, and to approximately 5 to 9 hours in patients with moderately to severely impaired renal function. Hepatically Impaired For patients with alcoholic cirrhosis, no significant changes in the kinetic disposition of immediate-release ketoprofen capsules were observed relative to age-matched normal subjects: the plasma clearance of drug was 0.07 L/kg/h in 26 hepatically impaired patients. The elimination half-life was comparable to that observed for normal subjects. However, the unbound (biologically active) fraction was approximately doubled, probably due to hypoalbuminemia and high variability which was observed in the pharmacokinetics for cirrhotic patients. Therefore, these patients should be carefully monitored and daily doses of ketoprofen kept at the minimum providing the desired therapeutic effect.
CONTRAINDICATIONS
Ketoprofen capsules are contraindicated in patients who have shown hypersensitivity to ketoprofen. Ketoprofen capsules USP should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic reactions to ketoprofen have been reported in such patients. Ketoprofen capsules are contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
ADVERSE REACTIONS
The incidence of common adverse reactions (above 1%) was obtained from a population of 835 ketoprofen-treated patients in double-blind trials lasting from 4 to 54 weeks and in 622 patients treated with ketoprofen extended-release capsules in trials lasting from 4 to 16 weeks. Minor gastrointestinal side effects predominated; upper gastrointestinal symptoms were more common than lower gastrointestinal symptoms. In crossover trials in 321 patients with rheumatoid arthritis or osteoarthritis, there was no difference in either upper or lower gastrointestinal symptoms between patients treated with 200 mg of ketoprofen extended-release capsules once a day or 75 mg of ketoprofen immediate-release capsules TID (255 mg/day). Peptic ulcer or GI bleeding occurred in controlled clinical trials in less than 1% of 1,076 patients; however, in open label continuation studies in 1,292 patients, the rate was greater than 2%. The incidence of peptic ulceration in patients on NSAIDs is dependent on many risk factors including age, sex, smoking, alcohol use, diet, stress, concomitant drugs such as aspirin and corticosteroids, as well as the dose and duration of treatment with NSAIDs (see WARNINGS ). Gastrointestinal reactions were followed in frequency by central nervous system side effects, such as headache, dizziness, or drowsiness. The incidence of some adverse reactions appears to be dose-related (see DOSAGE AND ADMINISTRATION ). Rare adverse reactions (incidence less than 1%) were collected from one or more of the following sources: foreign reports to manufacturers and regulatory agencies, publications, U.S. clinical trials, and/or U.S. postmarketing spontaneous reports. Reactions are listed below under body system, then by incidence or number of cases in decreasing incidence.
Incidence Greater Than 1% (Probable Causal Relationship) Digestive: Dyspepsia (11%), nausea•, abdominal pain•, diarrhea•, constipation•, flatulence•, anorexia, vomiting, stomatitis. Nervous System: Headache•, dizziness, CNS inhibition (i.e., pooled reports of somnolence, malaise, depression, etc.) or excitation (i.e., insomnia, nervousness, dreams, etc.)•. Special Senses: Tinnitus, visual disturbance. Skin and Appendages: Rash. Urogenital: Impairment of renal function (edema, increased BUN)•, signs or symptoms of urinary-tract irritation. • Adverse events occurring in 3 to 9% of patients.
Incidence Less Than 1% (Probable Causal Relationship) Body as a Whole: Chills, facial edema, infection, pain, allergic reaction, anaphylaxis. Cardiovascular: Hypertension, palpitation, tachycardia, congestive heart failure, peripheral vascular disease, vasodilation. Digestive: Appetite increased, dry mouth, eructation, gastritis, rectal hemorrhage, melena, fecal occult blood, salivation, peptic ulcer, gastrointestinal perforation, hematemesis, intestinal ulceration, hepatic dysfunction, hepatitis, cholestatic hepatitis, jaundice. Hemic: Hypocoagulability, agranulocytosis, anemia, hemolysis, purpura, thrombocytopenia. Metabolic and Nutritional: Thirst, weight gain, weight loss, hyponatremia. Musculoskeletal: Myalgia. Nervous System: Amnesia, confusion, impotence, migraine, paresthesia, vertigo. Respiratory: Dyspnea, hemoptysis, epistaxis, pharyngitis, rhinitis, bronchospasm, laryngeal edema. Skin and Appendages: Alopecia, eczema, pruritus, purpuric rash, sweating, urticaria, bullous rash, exfoliative dermatitis, photosensitivity, skin discoloration, onycholysis, toxic epidermal necrolysis, erythema multiforme, Stevens-Johnson syndrome.and fixed drug eruption (FDE) Special Senses: Conjunctivitis, conjunctivitis sicca, eye pain, hearing impairment, retinal hemorrhage and pigmentation change, taste perversion. Urogenital: Menometrorrhagia, hematuria, renal failure, interstitial nephritis, nephrotic syndrome.
To report SUSPECTED ADVERSE REACTIONS, contact Oncora Pharma at 1-662-993-9625 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Incidence Less Than 1% (Causal Relationship Unknown)
The following rare adverse reactions, whose causal relationship to ketoprofen is uncertain, are being listed to serve as alerting information to the physician. Body as a Whole: Septicemia, shock. Cardiovascular: Arrhythmias, myocardial infarction. Digestive: Buccal necrosis, ulcerative colitis, microvesicular steatosis, pancreatitis. Endocrine: Diabetes mellitus (aggravated). Nervous System: Dysphoria, hallucination, libido disturbance, nightmares, personality disorder, aseptic meningitis. Urogenital: Acute tubulopathy, gynecomastia
Drug Interactions
The following drug interactions were studied with ketoprofen doses of 200 mg/day. The possibility of increased interaction should be kept in mind when ketoprofen capsule doses greater than 50 mg as a single dose or 200 mg of ketoprofen per day are used concomitantly with highly bound drugs. ACE-inhibitors Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors.
Antacids Concomitant administration of magnesium hydroxide and aluminum hydroxide does not interfere with the rate or extent of the absorption of ketoprofen administered as ketoprofen capsules.
Aspirin Ketoprofen does not alter aspirin absorption; however, in a study of 12 normal subjects, concurrent administration of aspirin decreased ketoprofen protein binding and increased ketoprofen plasma clearance from 0.07 L/kg/h without aspirin to 0.11 L/kg/h with aspirin. The clinical significance of these changes is not known; however, as with other NSAIDs, concomitant administration of ketoprofen and aspirin is not generally recommended because of the potential of increased adverse effects.
Diuretics NSAIDs can reduce the natriuretic effect of furosemide and thiazides in some patients. Hydrochlorothiazide, given concomitantly with ketoprofen, produces a reduction in urinary potassium and chloride excretion compared to hydrochlorothiazide alone. Patients taking diuretics are at a greater risk of developing renal failure secondary to a decrease in renal blood flow caused by prostaglandin inhibition (see PRECAUTIONS ). During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see WARNINGS , Renal Effects ), as well as to assure diuretic efficacy.
Digoxin In a study in 12 patients with congestive heart failure where ketoprofen and digoxin were concomitantly administered, ketoprofen did not alter the serum levels of digoxin.
Lithium NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Methotrexate Ketoprofen, like other NSAIDs, may cause changes in the elimination of methotrexate leading to elevated serum levels of the drug and increased toxicity. NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
Probenecid Probenecid increases both free and bound ketoprofen by reducing the plasma clearance of ketoprofen to about one-third, as well as decreasing its protein binding. Therefore, the combination of ketoprofen and probenecid is not recommended.
Warfarin The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone. In a short-term controlled study in 14 normal volunteers, ketoprofen did not significantly interfere with the effect of warfarin on prothrombin time. Bleeding from a number of sites may be a complication of warfarin treatment and GI bleeding a complication of ketoprofen treatment. Because prostaglandins play an important role in hemostasis and ketoprofen has an effect on platelet function as well (see Drug/Laboratory Test Interactions , Effect on Blood Coagulation), concurrent therapy with ketoprofen and warfarin requires close monitoring of patients on both drugs.
DESCRIPTION
Ketoprofen is a non-steroidal anti-inflammatory drug. The chemical name for ketoprofen is 2-(3-benzoylphenyl)-propionic acid with the following structural formula:
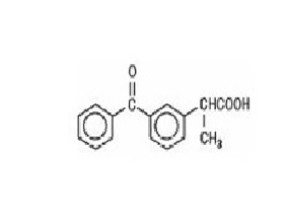
C 16 H 14 O 3 M.W. 254.29
It has a pKa of 5.94 in methanol: water (3:1) and an n-octanol: water partition coefficient of 0.97 (buffer pH 7.4). Ketoprofen is a white or off-white, odorless, nonhygroscopic, fine to granular powder, melting at about 95°C. It is freely soluble in ethanol, chloroform, acetone, ether and soluble in benzene and strong alkali, but practically insoluble in water at 20°C.
Ketoprofen Capsules USP 50 mg of Ketoprofen USP for oral administration.
Inactive Ingredients Drug Product Colloidal silicon dioxide, lactose monohydrate, magnesium stearate, povidone, sodium starch glycolate.
Capsule Shell Constituents 25 mg: D&C yellow 10, edible printing ink, FD&C blue 1, FD&C yellow 6, gelatin, sodium lauryl sulfate, and titanium dioxide. 50 mg: D&C yellow 10, edible printing ink, FD&C green 3, gelatin, sodium lauryl sulfate, and titanium dioxide. 75 mg: Edible printing ink, gelatin, sodium lauryl sulfate and titanium dioxide.
CLINICAL PHARMACOLOGY
Ketoprofen is a non-steroidal anti-inflammatory drug with analgesic and antipyretic properties. The anti-inflammatory, analgesic and antipyretic properties of ketoprofen have been demonstrated in classical animal and in vitro test systems. In anti-inflammatory models ketoprofen has been shown to have inhibitory effects on prostaglandin and leukotriene synthesis, to have antibradykinin activity, as well as to have lysosomal membrane-stabilizing action. However, its mode of action, like that of other non-steroidal anti-inflammatory drugs, is not fully understood.
Pharmacodynamics
Ketoprofen is a racemate with only the S enantiomer possessing pharmacological activity. The enantiomers have similar concentration time curves and do not appear to interact with one another. An analgesic effect-concentration relationship for ketoprofen was established in an oral surgery pain study with immediate-release ketoprofen capsules. The effect-site rate constant (k e0 ) was estimated to be 0.9 hour (95% confidence limits: 0 to 2.1), and the concentration (C e50 ) of ketoprofen that produced one-half the maximum PID (pain intensity difference) was 0.3 mcg/mL (95% confidence limits: 0.1 to 0.5). Thirty-three (33) to 68% of patients had an onset of action (as measured by reporting some pain relief) within 30 minutes following a single oral dose in postoperative pain and dysmenorrhea studies. Pain relief (as measured by remedication) persisted for up to 6 hours in 26 to 72% of patients in these studies.
Pharmacokinetics
General
The systemic availability (FS) when the oral formulation is compared with IV administration is approximately 90% in humans. For 75 to 200 mg single doses, the area under the curve has been shown to be dose proportional. Ketoprofen is > 99% bound to plasma proteins, mainly to albumin.
Absorption Ketoprofen is rapidly and well-absorbed, with peak plasma levels occurring within 0.5 to 2 hours.
When ketoprofen is administered with food, its total bioavailability (AUC) is not altered; however, the rate of absorption is slowed. Food intake reduces C max by approximately one-half and increases the mean time to peak concentration (t max ) from 1.2 hours for fasting subjects (range, 0.5 to 3 hours) to 2.0 hours for fed subjects (range, 0.75 to 3 hours). The fluctuation of plasma peaks may also be influenced by circadian changes in the absorption process. Concomitant administration of magnesium hydroxide and aluminum hydroxide does not interfere with absorption of ketoprofen from ketoprofen capsules. Multiple Dosing Steady-state concentrations of ketoprofen are attained within 24 hours after commencing treatment with immediate-release ketoprofen capsules. In studies with healthy male volunteers, trough levels at 24 hours following administration of immediate-release ketoprofen 50 mg capsules QID for 12 hours were 0.07 mg/L and 0.13 mg/L at 24 hours following administration of immediate-release ketoprofen 75 mg capsules TID for 12 hours. Thus, relative to the peak plasma concentration, the accumulation of ketoprofen after multiple doses of immediate-release ketoprofen capsules is minimal. The figure below shows a reduction in peak height and area after the second 50 mg dose. This is probably due to a combination of food effects, circadian effects, and plasma sampling times. It is unclear to what extent each factor contributes to the loss of peak height and area. The shaded area represents ± 1 standard deviation (S.D.) around the mean for immediate-release ketoprofen capsules.
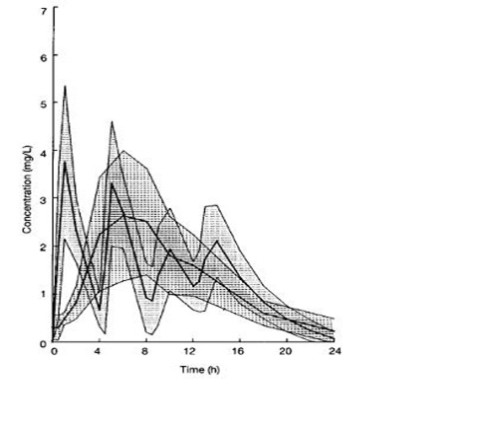
KETOPROFEN PLASMA CONCENTRATIONS IN SUBJECTS RECEIVING KETOPROFEN CAPSULES 50 MG EVERY 4 HOURS FOR 16 HOURS

Metabolism The metabolic fate of ketoprofen is glucuronide conjugation to form an unstable acyl-glucuronide. The glucuronic acid moiety can be converted back to the parent compound. Thus, the metabolite serves as a potential reservoir for parent drug, and this may be important in persons with renal insufficiency, whereby the conjugate may accumulate in the serum and undergo deconjugation back to the parent drug (see SpecialPopulations , Renally Impaired). The conjugates are reported to appear only in trace amounts in plasma in healthy adults, but are higher in elderly subjects-presumably because of reduced renal clearance. It has been demonstrated that in elderly subjects following multiple doses (50 mg every 6 h), the ratio of conjugated to parent ketoprofen AUC was 30% and 3%, respectively, for the S & R enantiomers. There are no known active metabolites of ketoprofen. Ketoprofen has been shown not to induce drug-metabolizing enzymes.
Elimination The plasma clearance of ketoprofen is approximately 0.08 L/kg/h with a Vd of 0.1 L/kg after IV administration. The elimination half-life of ketoprofen has been reported to be 2.05 ± 0.58 h (Mean ± S.D.) following IV administration from 2 to 4 hours following administration of ketoprofen capsules. In cases of slow drug absorption, the elimination rate is dependent on the absorption rate and thus t1/2 relative to an IV dose appears prolonged. In a 24 hour period, approximately 80% of an administered dose of ketoprofen is excreted in the urine, primarily as the glucuronide metabolite.
Enterohepatic recirculation of the drug has been postulated, although biliary levels have never been measured to confirm this.
Special Populations
Elderly
Clearance and unbound fraction The plasma and renal clearance of ketoprofen is reduced in the elderly (mean age, 73 years) compared to a younger normal population (mean age, 27 years). Hence, ketoprofen peak concentration and AUC increase with increasing age. In addition, there is a corresponding increase in unbound fraction with increasing age. Data from one trial suggest that the increase is greater in women than in men. It has not been determined whether age-related changes in absorption among the elderly contribute to the changes in bioavailability of ketoprofen. In a study conducted with young and elderly men and women, results for subjects older than 75 years of age showed that free drug AUC increased by 40% and Cmax increased by 60% as compared with estimates of the same parameters in young subjects (those younger than 35 years of age;. Also in the elderly, the ratio of intrinsic clearance/availability decreased by 35% and plasma half-life was prolonged by 26%. This reduction is thought to be due to a decrease in hepatic extraction associated with aging. Renally Impaired Studies of the effects of renal-function impairment have been small. They indicate a decrease in clearance in patients with impaired renal function. In 23 patients with renal impairment, free ketoprofen peak concentration was not significantly elevated, but free ketoprofen clearance was reduced from 15 L/kg/h for normal subjects to 7 L/kg/h in patients with mildly impaired renal function, and to 4 L/kg/h in patients with moderately to severely impaired renal function. The elimination t1/2 was prolonged from 1.6 hours in normal subjects to approximately 3 hours in patients with mild renal impairment, and to approximately 5 to 9 hours in patients with moderately to severely impaired renal function. Hepatically Impaired For patients with alcoholic cirrhosis, no significant changes in the kinetic disposition of immediate-release ketoprofen capsules were observed relative to age-matched normal subjects: the plasma clearance of drug was 0.07 L/kg/h in 26 hepatically impaired patients. The elimination half-life was comparable to that observed for normal subjects. However, the unbound (biologically active) fraction was approximately doubled, probably due to hypoalbuminemia and high variability which was observed in the pharmacokinetics for cirrhotic patients. Therefore, these patients should be carefully monitored and daily doses of ketoprofen kept at the minimum providing the desired therapeutic effect.
Clinical Trials
Rheumatoid Arthritis and Osteoarthritis
The efficacy of ketoprofen has been demonstrated in patients with rheumatoid arthritis and osteoarthritis. In other trials, ketoprofen demonstrated effectiveness comparable to aspirin, ibuprofen, naproxen, piroxicam, diclofenac, and indomethacin. In some of these studies there were more dropouts due to gastrointestinal side effects among patients on ketoprofen than among patients on other NSAIDs. In studies with patients with rheumatoid arthritis, ketoprofen was administered in combination with gold salts, antimalarials, low-dose methotrexate, d-penicillamine, and/or corticosteroids with results comparable to those seen with control non-steroidal drugs.
Management of Pain The effectiveness of immediate-release ketoprofen capsules as a general-purpose analgesic has been studied in standard pain models which have shown the effectiveness of doses of 25 to 150 mg. Doses of 25 mg were superior to placebo. Doses larger than 25 mg generally could not be shown to be significantly more effective, but there was a tendency toward faster onset and greater duration of action with 50 mg, and, in the case of dysmenorrhea, a significantly greater effect overall with 75 mg. Doses greater than 50 to 75 mg did not have increased analgesic effect. Studies in postoperative pain have shown that ketoprofen in doses of 25 to 100 mg was comparable to 650 mg of acetaminophen with 60 mg of codeine, or 650 mg of acetaminophen with 10 mg of oxycodone. Ketoprofen tended to be somewhat slower in onset; peak pain relief was about the same and the duration of the effect tended to be 1 to 2 hours longer, particularly with the higher doses of ketoprofen.
HOW SUPPLIED
Ketoprofen capsules USP are available as follows:
50 mg: Light green color cap and body, size “1” imprinted with “KETO 50” on cap with an edible blue ink and containing white to off-white crystalline powder supplied as: Bottles of 100 NDC 85477-111-01
Bottles of 100 NDC 85477-111-01
Keep tightly closed.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Manufactured for: Oncora Pharma
Dallas, TX 75228 Revised: 09/2021