Get your patient on Kionex - Sodium Polystyrene Sulfonate suspension (Sodium Polystyrene Sulfonate)
Kionex - Sodium Polystyrene Sulfonate suspension prescribing information
INDICATIONS AND USAGE
Kionex Suspension is indicated for the treatment of hyperkalemia.
DOSAGE AND ADMINISTRATION
Administer Kionex Suspension at least 3 hours before or 3 hours after other oral medications. Patients with gastroparesis may require a 6 hour separation (see WARNINGS and PRECAUTIONS, Drug Interactions ).
The average daily adult dose is 15 g (60 mL) to 60 g (240 mL) of suspension. This is best provided by administering 15 g (60 mL) of Kionex Suspension one to four times daily. Each 60 mL of Kionex Suspension contains 1500 mg (65 mEq) of sodium. Since the in-vivo efficiency of sodium-potassium exchange resins is approximately 33%, about one-third of the resin’s actual sodium content is being delivered to the body.
In smaller children and infants, lower doses should be employed by using as a guide a rate of 1 mEq of potassium per gram of resin as the basis for calculation. Administer with patient in an upright position (see WARNINGS ).
Kionex Suspension may be introduced into the stomach through a plastic tube and, if desired, given with a diet appropriate for a patient in renal failure.
Kionex Suspension may also be given, although with less effective results, as an enema consisting (for adults) of 30 g (120 mL) to 50 g (200 mL) every six hours. The enema should be retained as long as possible and followed by a cleansing enema.
After an initial cleansing enema, a soft, large size (French 28) rubber tube is inserted into the rectum for a distance of about 20 cm, with the tip well into the sigmoid colon, and taped into place. The suspension is introduced at body temperature by gravity. The suspension is flushed with 50 or 100 mL of fluid, following which the tube is clamped and left in place. If back leakage occurs, the hips are elevated on pillows or a knee-chest position is taken temporarily. The suspension is kept in the sigmoid colon for several hours, if possible. Then the colon is irrigated with a sodium-free cleansing enema at body temperature in order to remove the resin. Two quarts of flushing solution may be necessary. The returns are drained constantly through a Y tube connection. Particular attention should be paid to this cleansing enema, because sorbitol is present in the vehicle.
The intensity and duration of therapy depend upon the severity and resistance of hyperkalemia. Kionex Suspension should not be heated for to do so may alter the exchange properties of the resin.
CONTRAINDICATIONS
Kionex Suspension is contraindicated in the following conditions: patients with hypokalemia, patients with a history of hypersensitivity to polystyrene sulfonate resins, obstructive bowel disease, oral or rectal administration in neonates (see PRECAUTIONS ).
ADVERSE REACTIONS
Kionex Suspension may cause some degree of gastric irritation. Anorexia, nausea, vomiting, and constipation may occur especially if high doses are given. Also, hypokalemia, hypocalcemia, hypomagnesemia and significant sodium retention, and their related clinical manifestations, may occur (see WARNINGS ). Occasionally diarrhea develops. Large doses in elderly individuals may cause fecal impaction (see PRECAUTIONS ). Rare instances of intestinal necrosis have been reported. Intestinal obstruction due to concretions of aluminum hydroxide, when used in combination with sodium polystyrene sulfonate, has been reported.
The following events have been reported from worldwide post marketing experience:
- Fecal impaction following rectal administration, particularly in children;
- Gastrointestinal concretions (bezoars) following oral administration;
- Ischemic colitis, gastrointestinal tract ulceration or necrosis which could lead to intestinal perforation; and
- Rare cases of acute bronchitis and/or bronchopneumonia associated with inhalation of particles of polystyrene sulfonate (see WARNINGS ).
To report SUSPECTED ADVERSE REACTIONS, contact ANI Pharmaceuticals, Inc. at 1-800-308-6755 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Kionex ® (Sodium Polystyrene Sulfonate Suspension USP) can be administered orally or in an enema. It is a raspberry flavored suspension containing 15 grams of cation-exchange resin (sodium polystyrene sulfonate USP) and 21.5 mL of sorbitol solution (equivalent to approximately 19.3 grams of sorbitol) per 60 mL of suspension. Also contains purified water, propylene glycol, xanthan gum, sodium saccharin, methylparaben, propylparaben, and flavor.
Sodium polystyrene sulfonate is a benzene, diethenyl-, polymer with ethenylbenzene, sulfonated, sodium salt and has the following structural formula:
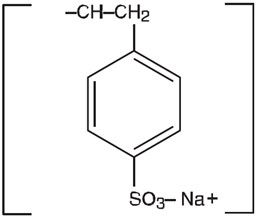
The sodium content of the suspension is 1500 mg (65 mEq) per 60 mL. It is a brown, slightly viscous suspension with an in-vitro exchange capacity of approximately 3.1 mEq ( in-vivo approximately 1 mEq) of potassium per 4 mL (1 gram) of suspension. It can be administered orally or in an enema.
CLINICAL PHARMACOLOGY
As the resin passes along the intestine or is retained in the colon after administration by enema, the sodium ions are partially released and are replaced by potassium ions. For the most part, this action occurs in the large intestine, which excretes potassium ions to a greater degree than does the small intestine. The efficiency of this process is limited and unpredictably variable. It commonly approximates the order of 33%, but the range is so large that definitive indices of electrolyte balance must be clearly monitored.
Metabolic data are unavailable.
HOW SUPPLIED
Kionex ® Suspension is a light brown, raspberry-flavored suspension supplied as follows: NDC 62559-356-01: Case containing 10 unit dose bottles of 60 mL (NDC 62559-356-60)
Dispense in a tight container, as defined in the USP. If repackaging into other containers, store in refrigerator and use within 14 days of packaging.
SHAKE WELL UNTIL FULLY SUSPENDED BEFORE USING.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Distributed by: ANI Pharmaceuticals, Inc. Baudette, MN 56623  Issued: 04/2024 LB4701-00
Issued: 04/2024 LB4701-00