Kit For The Preparation Of Technetium Tc99m Mertiatide - Betiatide injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE
Technetium Tc 99m mertiatide is a renal imaging agent for use in the diagnosis of congenital and acquired abnormalities, renal failure, urinary tract obstruction, and calculi in adults and pediatric patients. (See Pediatric Use .) It is a diagnostic aid in providing renal function, split function, renal angiograms, and renogram curves for whole kidney and renal cortex.
DOSAGE AND ADMINISTRATION
The suggested dose range employed in the average adult patient (70kg) for renal function and imaging studies is 185 MBq (5 mCi) to 370 MBq (10 mCi). In pediatric patients the recommended dose range is 2.6 MBq/kg (70 μCi/kg) to 5.2 MBq/kg (140 μCi/kg) with a minimum dose of 37 MBq (1 mCi).
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to administration.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
Aseptic procedures and a shielded syringe should be employed in withdrawing doses for administration to patients. The user should wear waterproof gloves during the administration procedure.
CONTRAINDICATIONS
None known.
ADVERSE REACTIONS
The following adverse reactions have been reported: nausea, vomiting, wheezing, dyspnea, itching, rash, tachycardia, hypertension, shaking chills, fever, and seizure.
DESCRIPTION
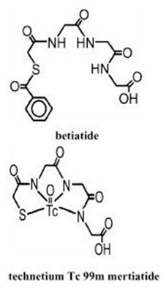
Kit for the Preparation of Technetium Tc99m Mertiatide is used for the preparation of technetium Tc 99m mertiatide, a diagnostic radiopharmaceutical. It is supplied as a sterile, nonpyrogenic, lyophilized powder. Each vial contains betiatide (N-[N-[N-[(benzoylthio) acetyl]glycyl]glycyl]-glycine). After reconstitution with sterile sodium pertechnetate Tc 99m injection, the technetium Tc 99m mertiatide (disodium[N-[N-[N-(mercaptoacetyl) glycyl]glycyl] glycinato (2-) - N,N′,N″,S′]oxotechnetate (2-)) which is formed is suitable for intravenous administration.
Each 10 milliliter vial contains 1 milligram betiatide, 0.05 milligram (minimum) stannous chloride dihydrate (SnCl 2 ∙2H 2 O) and 0.2 milligram (maximum) total tin expressed as stannous chloride dihydrate (SnCl 2 ∙2H 2 O), 40 milligrams sodium tartrate dihydrate (Na 2 C 4 H 2 O 6 ∙2H 2 O), and 20 milligrams lactose monohydrate. Prior to lyophilization, sodium hydroxide or hydrochloric acid may be added for pH adjustment. The pH of the reconstituted drug is between 5.0 and 6.0. No bacteriostatic preservative is present. The contents are sealed under argon. Betiatide is light sensitive and must be protected from light. Betiatide and technetium Tc 99m mertiatide have the following structural formulas:
CLINICAL PHARMACOLOGY
Following intravenous injection of technetium Tc 99m mertiatide, the appearance, concentration, and excretion of the tracer in the kidney can be monitored to assess renal function. Although technetium Tc 99m mertiatide is highly plasma protein bound following intravenous injection, the protein binding is reversible and the tracer is rapidly excreted by the kidneys via active tubular secretion and glomerular filtration. Following intravenous injection of technetium Tc 99m mertiatide in normal volunteers, 89% of the tracer was plasma protein bound. In healthy subjects with normal renal function (mean serum creatinine 1.2 mg/dL) technetium Tc 99m mertiatide was rapidly cleared from the blood. The plasma clearance was approximately 0.3 liters/minute and the amount of technetium Tc 99m mertiatide excreted in the urine in three hours was nearly 90% of the dose. In a study performed in three patients with renal impairment (serum creatinine greater than 6.3 mg/dL), there was decreased blood clearance and a decrease in the amount excreted in the urine over three hours. In these patients, 78% of the tracer was plasma protein bound after intravenous injection. The mean plasma clearance of technetium Tc 99m mertiatide was 0.03 liters/minute and 21.3% was excreted in three hours on average. In both healthy subjects and patients with renal impairment, the plasma concentration-time profile showed a biexponential decline.
HOW SUPPLIED
Kit for the Preparation of Technetium Tc 99m Mertiatide is supplied as a lyophilized powder packaged in vials. Each reaction vial contains 1 mg betiatide, 0.05 mg (minimum) stannous chloride dihydrate (SnCl 2 ∙2H 2 O), 0.2 mg (maximum) total tin expressed as stannous chloride dihydrate (SnCl 2 ∙2H 2 O), 40 mg sodium tartrate dihydrate (Na 2 C 4 H 2 O 6 ∙2H 2 O), and 20 mg lactose monohydrate.
The pH of the reconstituted drug is between 5.0 and 6.0. No bacteriostatic preservative is present.
Packages containing 5 reaction vials (NDC 45567-0655-1) are available.
STORAGE
Kit for the Preparation of Technetium Tc 99m Mertiatide should be stored at controlled room temperature 20°C to 25°C (68-77°F) and protected from light until use. The reconstituted vial should be stored at room temperature (15°C to 30°C) and must be used within six hours of preparation.