Get your patient on Komzifti (Ziftomenib)
Komzifti prescribing information
WARNING: DIFFERENTIATION SYNDROME
Differentiation syndrome, which can be fatal, has occurred with KOMZIFTI. Signs and symptoms may include fever, joint pain, hypotension, hypoxia, dyspnea, rapid weight gain or peripheral edema, pleural or pericardial effusions, pulmonary infiltrates, acute kidney injury, and rashes. If differentiation syndrome is suspected, interrupt KOMZIFTI and initiate oral or intravenous corticosteroids with hemodynamic and laboratory monitoring until symptom resolution; resume KOMZIFTI upon symptom improvement [see Dosage and Administration (2.5 ), Warnings and Precautions (5.1 ), and Adverse Reactions (6.1 )] .
INDICATIONS AND USAGE
KOMZIFTI is indicated for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) with a susceptible nucleophosmin 1 ( NPM1 ) mutation who have no satisfactory alternative treatment options [see Dosage and Administration (2.1 ), Clinical Pharmacology (12.1 ), and Clinical Studies (14 )] .
DOSAGE AND ADMINISTRATION
- Select patients for treatment with KOMZIFTI based on the presence of an NPM1 mutation. (2.1 )
- Recommended dosage: 600 mg orally once daily until disease progression or unacceptable toxicity. For patients without confirmed disease progression or unacceptable toxicity, treat for a minimum of 6 months to allow time for a clinical response. (2.2 )
- See Full Prescribing Information for administration instructions and dosage modifications. (2.3 , 2.4 , 2.5 )
Patient Selection
Select patients for the treatment of relapsed or refractory AML with KOMZIFTI based on the presence of an NPM1 mutation [see Clinical Studies (14 )] .
An FDA-approved test for the detection of NPM1 mutations is not currently available.
Recommended Dosage
The recommended dosage of KOMZIFTI is 600 mg taken orally once daily until disease progression or unacceptable toxicity. Do not start KOMZIFTI until the white blood cell (WBC) count is reduced to less than 25 x 10⁹/L. For patients without confirmed disease progression or unacceptable toxicity, treatment for a minimum of 6 months is recommended to allow time for a clinical response.
Administration Instructions
- Administer KOMZIFTI once daily on an empty stomach (at least 1 hour before or 2 hours after a meal) [see Clinical Pharmacology (12.3 )].
- Administer KOMZIFTI orally at about the same time each day.
- Swallow capsules whole. Do not open, break, or chew the capsules.
- If a dose of KOMZIFTI is missed or not taken at the usual time, administer the dose as soon as possible on the same day and at least 12 hours prior to the next scheduled dose. Return to the normal schedule the following day. Do not administer 2 doses within 12 hours.
Dosage Modifications for Concomitant Use of Acid-Reducing Agents
Avoid concomitant use of proton pump inhibitors (PPIs) with KOMZIFTI.
Avoid concomitant use of a histamine-2 (H2) receptor antagonist or a locally acting antacid with KOMZIFTI [see Drug Interactions (7.1 )] . If concomitant use cannot be avoided:
- Take KOMZIFTI 2 hours before or 10 hours after administration of an H2 receptor antagonist.
- Take KOMZIFTI 2 hours before or 2 hours after administration of a locally acting antacid.
Dosage Modifications for Adverse Reactions
Manage any abnormalities promptly [see Warnings and Precautions (5.1 , 5.2 ) and Adverse Reactions (6.1 )] .
Interrupt or reduce dose for adverse reactions as per Table 1 and Table 2 .
| Adverse Reaction a | Recommended Action |
|---|---|
a Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 5.0. | |
b Grade 1 is mild, Grade 2 is moderate, Grade 3 is severe, Grade 4 is life-threatening. | |
c See Table 2 for the reduced dose levels. | |
| Differentiation syndrome [see Warnings and Precautions (5.1 )] |
|
| Noninfectious leukocytosis (e.g., sudden or significant white blood cell (WBC) increase, including WBC doubling within the first 2 weeks of administration or an absolute increase of 10 x 10 9 /L) |
|
| Nonhematological adverse reactions Grade ≥ 3 b [see Adverse Reactions (6.1 )] |
|
•Permanently discontinue KOMZIFTI in patients unable to tolerate 200 mg orally once daily following second dose reduction. | |
| Recommended Dosage | |
| First dose reduction | 400 mg orally once daily |
| Second dose reduction | 200 mg orally once daily |
DOSAGE FORMS AND STRENGTHS
Capsules of ziftomenib are available in 200 mg strength. The capsules are white and are imprinted with “ZIF 200” in black ink.
USE IN SPECIFIC POPULATIONS
Lactation: Advise not to breastfeed. (8.2 )
Pregnancy
Risk Summary
Based on findings in animals and its mechanism of action [see Clinical Pharmacology (12.1 )] , KOMZIFTI can cause embryo-fetal harm when administered to a pregnant woman. There are no available data on KOMZIFTI use in pregnant women to evaluate for a drug-associated risk.
In animal reproduction studies, oral administration of ziftomenib to pregnant mice during the period of organogenesis resulted in adverse developmental outcomes, including embryo-fetal mortality, structural abnormalities, and altered fetal growth at maternal exposures approximately 0.3 times the human exposure (AUC) at the recommended dose (see Data ) . Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Ziftomenib was administered orally twice daily to pregnant mice at doses of 6, 20, and 60 mg/kg/day during organogenesis (gestation days 6-15). Decreased fetal body weight, embryo-fetal lethality, fetal external and skeletal malformations and variations (cleft palate, fused cervical arches, absent lumbar vertebrae fused and/or misaligned costal cartilage, absent or short rib, supernumerary site in the suture bone or split palatine of the skull, and/or fused, unossified, and/or incompletely ossified sternebrae) were noted at all doses. At the dose of 6 mg/kg/day in mice, the maternal exposure was approximately 0.3 times the human exposure at the recommended dose of 600 mg once daily.
Lactation
Risk Summary
There are no data on the presence of ziftomenib or its metabolites in human milk or the effects of ziftomenib or its metabolites on the breastfed child or milk production. Because of the potential for adverse reactions in the breastfed child, advise women not to breastfeed during treatment with KOMZIFTI and for 2 weeks after the last dose.
Females and Males of Reproductive Potential
Based on findings in animals and its mechanism of action, KOMZIFTI can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1 )] .
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to initiating KOMZIFTI.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with KOMZIFTI and for 6 months after the last dose.
Males
Advise males with female partners of reproductive potential to use effective contraception during treatment with KOMZIFTI and for 3 months after the last dose.
Infertility
Based on findings in animals, KOMZIFTI may impair fertility in females and males of reproductive potential. Findings in animals were not reversible after a 4-week recovery period [see Nonclinical Toxicology (13.1 )].
Pediatric Use
Safety and effectiveness of KOMZIFTI in pediatric patients have not been established.
Geriatric Use
Of the 112 patients with relapsed or refractory AML with an NPM1 mutation treated with KOMZIFTI, 70 (63%) patients were 65 years of age or older and 31 (28%) were 75 years or older.
No overall differences in effectiveness, safety, or pharmacokinetics of KOMZIFTI were observed between patients aged 65 years or older and younger patients [see Clinical Studies (14 ) and Clinical Pharmacology (12.3 )].
Renal Impairment
No dose adjustment is required for patients with mild or moderate (CL Cr 30 to 89 mL/min) renal impairment. The effects of severe (CL Cr less than 30 mL/min) renal impairment have not been studied [see Clinical Pharmacology (12.3 )].
Hepatic Impairment
No dose adjustment is required for patients with mild (bilirubin less than or equal to ULN and AST greater than ULN or bilirubin less than or equal to 1.5 x ULN) or moderate (bilirubin 1.5 to 3 x ULN) hepatic impairment. The effects of severe hepatic impairment (bilirubin greater than 3 x ULN) have not been studied [see Clinical Pharmacology (12.3 )] .
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
QTc Interval Prolongation: Monitor electrocardiograms and electrolytes. Correct hypokalemia and hypomagnesemia prior to treatment. Interrupt KOMZIFTI if the QTc interval is > 500 ms. (5.2 )
Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.3 , 8.1 , 8.3 )
Differentiation Syndrome
KOMZIFTI can cause fatal or life-threatening differentiation syndrome. Differentiation syndrome is associated with rapid proliferation and differentiation of myeloid cells. Symptoms of differentiation syndrome, including those seen in patients treated with KOMZIFTI, may include fever, hypoxia, joint pain, hypotension, dyspnea, rapid weight gain or peripheral edema, pleural or pericardial effusions, acute kidney injury, and rashes.
In the clinical trial, differentiation syndrome occurred in 29 (26%) of 112 patients with relapsed or refractory AML with an NPM1 mutation who were treated with KOMZIFTI at the recommended dosage. Differentiation syndrome was Grade 3 in 13% and fatal in two patients. In broader evaluation of all patients with any genetic form of AML treated with KOMZIFTI monotherapy in clinical trials, differentiation syndrome occurred in 25% of patients. Four fatal cases of differentiation syndrome occurred out of 39 patients with KMT2A -rearranged AML treated with KOMZIFTI. KOMZIFTI is not approved for use in patients with KMT2A -rearranged AML.
In the 112 patients with an NPM1 mutation, differentiation syndrome was observed with and without concomitant hyperleukocytosis, in as early as 3 days and up to 46 days after KOMZIFTI initiation. The median time to onset was 15 days. Two patients experienced more than one differentiation syndrome event. Treatment was interrupted and resumed in 15 (13%) patients, while it was permanently discontinued in 2 (2%) patients [see Adverse Reactions (6.1 )] .
Prior to starting treatment with KOMZIFTI, reduce the WBC counts to less than 25 x 10⁹/L. If differentiation syndrome is suspected, interrupt KOMZIFTI, initiate oral or intravenous corticosteroids (e.g., dexamethasone 10 mg every 12 hours) for a minimum of 3 days with hemodynamic and laboratory monitoring. Resume treatment with KOMZIFTI at the same dose level when signs and symptoms improve and are Grade 2 or lower. Taper corticosteroids over a minimum of 3 days after adequate control or resolution of symptoms [see Dosage and Administration (2.5 )] . Symptoms of differentiation syndrome may recur with premature discontinuation of corticosteroid treatment.
QTc Interval Prolongation
KOMZIFTI can cause QT (QTc) interval prolongation [see Clinical Pharmacology (12.2 ) ] . In the clinical trial, QTc interval prolongation was reported as an adverse reaction in 12% of 112 patients treated with KOMZIFTI at the recommended dosage for relapsed or refractory AML with an NPM1 mutation. QTc interval prolongation was Grade 3 in 8% of patients. The heart-rate corrected QT interval (using Fridericia's method) (QTcF) was greater than 500 msec in 9% of patients, and the increase from baseline QTcF was greater than 60 msec in 12% of patients. KOMZIFTI dose reduction was required for 1% of patients due to QTc interval prolongation [see Adverse Reactions (6.1 )] . QTc prolongation occurred in 14% of the 42 patients less than 65 years of age and in 10% of the 70 patients 65 years of age or older.
Correct electrolyte abnormalities, including hypokalemia and hypomagnesemia, prior to treatment with KOMZIFTI. Perform an ECG prior to initiation of treatment with KOMZIFTI, and do not initiate KOMZIFTI in patients with QTcF > 480 msec. Perform an ECG at least once weekly for the first four weeks on treatment, and at least monthly thereafter. Interrupt KOMZIFTI if the QTc interval is > 500 ms or the change from baseline is > 60 ms (Grade 3) [see Dosage and Administration (2.5 )] . In patients with congenital long QTc syndrome, congestive heart failure, electrolyte abnormalities, or those who are taking medications known to prolong the QTc interval, more frequent ECG monitoring may be necessary. Concomitant use of KOMZIFTI with drugs known to prolong the QTc interval may increase the risk of QTc interval prolongation [see Drug Interactions (7.1 ), Clinical Pharmacology (12.2 )] .
Embryo-Fetal Toxicity
Based on findings in animals and its mechanism of action, KOMZIFTI can cause embryo-fetal harm when administered to a pregnant woman. In animal reproduction studies, oral administration of ziftomenib to pregnant mice during the period of organogenesis caused adverse developmental outcomes, including embryo-fetal mortality, structural abnormalities, and altered fetal growth at approximately 0.3 times the steady-state clinical exposure based on the area under the concentration-time curve (AUC) at the recommended human dose.
Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with KOMZIFTI and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with KOMZIFTI and for 3 months after the last dose [see Use in Specific Populations (8.1 , 8.3 )] .
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Relapsed or Refractory AML with an NPM1 Mutation
The safety of KOMZIFTI for the treatment of patients with relapsed or refractory AML with an NPM1 mutation was evaluated in KO-MEN-001 [see Clinical Studies (14 )] . Patients received KOMZIFTI 600 mg once daily (n=112). The median duration of exposure to KOMZIFTI was 2.2 months (range 0.1 to 21.5 months).
Fatal adverse reactions occurred in 4 (4%) patients who received KOMZIFTI, including 2 with differentiation syndrome, 1 with infection, and 1 with sudden death. Serious adverse reactions were reported in 79% of patients who received KOMZIFTI. Serious adverse reactions occurring in ≥ 5% of patients included infection without an identified pathogen (29%), febrile neutropenia (18%), bacterial infection (16%), differentiation syndrome (16%), and dyspnea (6%).
Dosage interruption of KOMZIFTI due to an adverse reaction occurred in 54% of patients. Adverse reactions that required dose interruption in ≥ 2% of patients included infection without an identified pathogen (15%), differentiation syndrome (13%), febrile neutropenia (5%), pyrexia (4%), electrocardiogram QT prolonged (4%), leukocytosis (4%), bacterial infection (3%), cardiac failure (2%), cholecystitis (2%), diarrhea (2%), pruritus (2%), and thrombosis (2%). Dose reduction of KOMZIFTI due to an adverse reaction occurred in 4% of patients. Permanent discontinuation of KOMZIFTI due to an adverse reaction occurred in 21% of patients. Adverse reactions that required permanent discontinuation of KOMZIFTI in ≥ 2% of patients were infection without an identified pathogen (8%), bacterial infection (4%), cardiac arrest (2%), and differentiation syndrome (2%).
The most common (≥ 20%) adverse reactions, including laboratory abnormalities, were aspartate aminotransferase increased, infection without an identified pathogen, potassium decreased, albumin decreased, alanine aminotransferase increased, sodium decreased, creatinine increased, alkaline phosphatase increased, hemorrhage, diarrhea, nausea, fatigue, edema, bacterial infection, musculoskeletal pain, bilirubin increased, potassium increased, differentiation syndrome, pruritus, febrile neutropenia, and transaminases increased.
Table 3 summarizes the adverse reactions in KO-MEN-001.
†Includes the following fatal adverse reactions: infection (n=1) and differentiation syndrome (n=2). | ||
•No Grade 4 events were reported for this adverse reaction. | ||
a Includes abdominal abscess, abscess limb, anal abscess, anorectal infection, bronchitis, conjunctivitis, device related infection, device related sepsis, diverticulitis, emphysematous cystitis, enterocolitis infectious, gingivitis, hordeolum, infection, large intestine infection, nail infection, necrotizing fasciitis, penile infection, periodontitis, perirectal abscess, peritonitis, pneumonia, postoperative abscess, respiratory tract infection, rhinitis, sepsis, septic shock, sinusitis, skin infection, soft tissue infection, tooth infection, upper respiratory tract infection, urosepsis, vaginal infection, vascular device infection, wound infection. | ||
b Includes bacteremia, bacterial infection, bacterial pyelonephritis, breast cellulitis, cellulitis, clostridial infection, clostridium difficile colitis, clostridium difficile infection, enterobacter sepsis, enterococcal bacteremia, erysipelas, escherichia bacteremia, escherichia infection, escherichia sepsis, escherichia urinary tract infection, klebsiella bacteremia, klebsiella infection, klebsiella urinary tract infection, paronychia, pneumonia bacterial, pneumonia klebsiella, pseudomonal bacteremia, staphylococcal bacteremia. | ||
c Includes adenovirus infection, COVID-19, genital herpes, herpes simplex reactivation, herpes virus infection, herpes zoster, nasal herpes, oral herpes, pneumonia influenzas, pneumonia parainfluenza viral, respiratory syncytial virus infection, rhinovirus infection. | ||
d Includes angina bullosa hemorrhagic, bladder tamponade, catheter site hematoma, conjunctival hemorrhage, contusion, disseminated intravascular coagulation, ecchymosis, epistaxis, gastric hemorrhage, gastrointestinal hemorrhage, gingival bleeding, hematemesis, hematochezia, hematoma, hematuria, hemoptysis, hemorrhage, hemorrhoidal hemorrhage, hyperfibrinolysis, injection site hematoma, injection site hemorrhage, lower gastrointestinal hemorrhage, melaena, mouth hemorrhage, oral blood blister, petechiae, purpura, rectal hemorrhage, retinal hemorrhage, shock hemorrhagic, skin hemorrhage, subdural hematoma, tongue hemorrhage, traumatic hematoma, upper gastrointestinal hemorrhage, vaginal hemorrhage, vitreous hemorrhage. | ||
e Includes diarrhea, colitis, colitis erosive. | ||
f Includes nausea, vomiting. | ||
g Includes fatigue, asthenia, malaise. | ||
h Includes edema, edema peripheral, generalized edema, localized edema, peripheral swelling. | ||
i Includes arthralgia, back pain, bone pain, flank pain, musculoskeletal chest pain, musculoskeletal pain, myalgia, neck pain, pain in extremity. | ||
j Includes pruritus, nasal pruritus. | ||
k Includes white blood cell count increased, leukocytosis, hyperleukocytosis. | ||
l Includes transaminases increased, alanine aminotransferase increased, aspartate aminotransferase increased, gamma-glutamyl transferase increased, hypertransaminasemia. | ||
m Includes renal impairment: acute kidney injury, azotemia, blood creatinine increased, blood urea increased, chronic kidney disease, postrenal failure, renal failure. | ||
| Adverse Reaction | KOMZIFTI N=112 | |
| All Grades † (%) | Grade 3 or 4 (%) | |
| Infections and infestations | ||
| Infection without identified pathogen a | 52 | 38 |
| Bacterial infection b | 28 | 17 |
| Viral infection c | 16 | 5 |
| Vascular disorders | ||
| Hemorrhage d | 38 | 8 |
| Hypertension | 11 | 5 |
| Gastrointestinal disorders | ||
| Diarrhea e• | 36 | 5 |
| Nausea f• | 35 | 2 |
| General disorders and administration site conditions | ||
| Fatigue g | 34 | 8 |
| Edema h• | 30 | 3 |
| Musculoskeletal and connective tissue disorders | ||
| Musculoskeletal pain i | 28 | 4 |
| Neoplasms benign, malignant, and unspecified (incl cysts and polyps) | ||
| Differentiation syndrome | 26 | 13 |
| Skin and subcutaneous | ||
| Pruritus j• | 23 | 0 |
| Blood and lymphatic system disorder | ||
| Febrile neutropenia | 22 | 22 |
| Leukocytosis k• | 16 | 5 |
| Investigations | ||
| Transaminases increased l | 21 | 8 |
| Electrocardiogram QT prolonged • | 12 | 8 |
| Renal and urinary disorders | ||
| Renal impairment m | 19 | 6 |
| Respiratory, thoracic, and mediastinal disorders | ||
| Hypoxia | 9 | 5 |
Table 4 summarizes the laboratory abnormalities in KO-MEN-001.
•The denominator used to calculate the rate varied from 108 to 110 based on the number of patients with a baseline value and at least one post-baseline value. | ||
| Laboratory Abnormality | KOMZIFTI | |
| Grades 1-4• (%) | Grade 3-4• (%) | |
| Aspartate aminotransferase increased | 53 | 4 |
| Potassium decreased | 52 | 22 |
| Albumin decreased | 51 | 5 |
| Alanine aminotransferase increased | 50 | 6 |
| Sodium decreased | 49 | 3 |
| Creatinine increased | 45 | 4 |
| Alkaline phosphatase increased | 41 | 5 |
| Bilirubin increased | 27 | 6 |
| Potassium increased | 26 | 6 |
DRUG INTERACTIONS
Strong or Moderate CYP3A4 Inhibitors: Monitor more frequently for adverse reactions. (7.1 )
Strong or Moderate CYP3A4 Inducers: Avoid concomitant use. (7.1 )
Proton Pump Inhibitors: Avoid concomitant use. (2.4 , 7.1 )
H2 Receptor Antagonists and Antacids: Avoid concomitant use. If concomitant use cannot be avoided, modify KOMZIFTI administration time. (2.4 , 7.1 )
Drugs that Prolong the QTc Interval: Avoid concomitant use. If concomitant use is unavoidable, monitor patients more frequently for QTc interval prolongations. (5.2 , 7.1 )
Effect of Other Drugs on KOMZIFTI
Strong or Moderate CYP3A4 Inhibitors
Monitor patients more frequently for KOMZIFTI-associated adverse reactions.
Ziftomenib is primarily metabolized by CYP3A. Concomitant use of KOMZIFTI with strong or moderate CYP3A4 inhibitors increases ziftomenib exposure [see Clinical Pharmacology (12.3 )], which may increase the risk of adverse reactions such as QT prolongation.
Strong or Moderate CYP3A4 Inducers
Avoid concomitant use of KOMZIFTI with strong or moderate CYP3A4 inducers.
Ziftomenib is primarily metabolized by CYP3A. Concomitant use of KOMZIFTI with strong or moderate CYP3A4 inducers may decrease ziftomenib exposure [see Clinical Pharmacology (12.3 )] , which may reduce the efficacy of KOMZIFTI.
Gastric Acid Reducing Agents
Concomitant administration of ziftomenib with proton pump inhibitors (PPIs) decreases ziftomenib exposure [see Clinical Pharmacology (12.3 )] , which may reduce the efficacy of KOMZIFTI.
Avoid concomitant use of KOMZIFTI with PPIs.
Avoid concomitant use of KOMZIFTI with H2 receptor antagonists (H2RAs) or locally acting antacids. If concomitant use cannot be avoided, modify KOMZIFTI administration time [see Dosage and Administration (2.4 )] .
Drugs that Prolong the QT Interval
Avoid concomitant use of KOMZIFTI with other product(s) with a known potential to prolong the QTc interval. If concomitant use cannot be avoided, obtain ECGs when initiating, during concomitant use, and as clinically indicated [see Warnings and Precautions (5.2 )] . Interrupt KOMZIFTI if the QTc interval is > 500 ms or the change from baseline is > 60 ms [see Dosage and Administration (2.5 )] .
Ziftomenib causes QTc interval prolongation [see Clinical Pharmacology (12.2 )] . Concomitant use of KOMZIFTI with other products that prolong the QTc interval may result in a greater increase in the QTc interval and adverse reactions associated with QTc interval prolongation, including Torsades de pointes, other serious arrhythmias, and sudden death [see Warnings and Precautions (5.2 )] .
DESCRIPTION
KOMZIFTI contains ziftomenib, a menin inhibitor.
The chemical name is ( S )-4-methyl-5-((4-((2-(methylamino)-6-(2,2,2-trifluoroethyl)thieno[2,3-d]pyrimidin-4-yl)amino)piperidin-1-yl)methyl)-1-(2-(4-(methylsulfonyl)piperazin-1-yl)propyl)-1 H -indole-2-carbonitrile.
The chemical structure is:
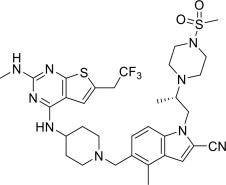
The molecular formula is C 33 H 42 F 3 N 9 O 2 S 2 and the molecular weight is 717.88 g/mol.
Ziftomenib is a white to off-white powder and is highly soluble in aqueous solution at pH 1.2 and practically insoluble at pH ≥4.
KOMZIFTI (ziftomenib) is available as a 200 mg capsule for oral administration.
Each KOMZIFTI capsule contains 200 mg ziftomenib and the following inactive ingredients: croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and sodium lauryl sulfate. The capsule shells, imprinted with black ink, contain the following inactive ingredients: hypromellose and titanium dioxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
Ziftomenib is a menin inhibitor that blocks the interaction of menin and lysine [K]-specific methyltransferase 2A (KMT2A).
Acute leukemias can be driven by mutations in NPM1 that recruit the wild-type menin-KMT2A complex to promoters of leukemogenic genes. Susceptible NPM1 mutations are defined as those that result in loss of the nucleolar localization signal and the insertion of a new nuclear export signal leading to cytoplasmic accumulation of mutant NPM1 protein that disrupts normal cell function and drives leukemogenesis through altered gene expression.
Pharmacologic disruption of the menin-KMT2A protein-protein interaction by ziftomenib blocks oncogenic activity of mutant NPM1, which induces differentiation of leukemic cells as evidenced by increased expression of differentiation markers. In nonclinical studies, ziftomenib demonstrated in vitro and in vivo antitumor activity in models of NPM1 -mutant leukemia.
Pharmacodynamics
Ziftomenib exposure-response relationships have not been fully characterized and the time course of pharmacodynamic response is unknown.
Cardiac Electrophysiology
The effect of KOMZIFTI on the QTc interval was evaluated at 600 mg (the approved recommended dosage) in patients with relapsed or refractory acute myeloid leukemia in KO-MEN-001 (n=133).
The increase in the QTc interval was concentration-dependent with the largest mean increase in QTc interval predicted to be 7.7 ms (upper confidence interval = 12.6 ms) after administration of ziftomenib 600 mg once daily. There were 9 subjects (7%) with QTcF > 500 ms and an increase in QTc > 60 ms over baseline [see Warnings and Precautions (5.2 )] .
Pharmacokinetics
Ziftomenib pharmacokinetics were characterized in patients with relapsed or refractory AML. Pharmacokinetic parameters are presented as mean (% CV), unless otherwise specified.
Abbreviations: C max = maximum plasma concentration; AUC = area under the time concentration curve; T max = time to peak concentration | ||
a Steady-state (Cycle 2 Day 1). | ||
b Approximately 1,000 calories, 60% fat, 25% carbohydrates, 15% protein. | ||
| Parameter | Dosage | |
| 600 mg once daily (with strong CYP3A4 inhibitors) | 600 mg once daily (without CYP3A4 inhibitors) | |
| General Information | ||
| Exposure a | ||
| C max (ng/mL) | 617 (33) | 288 (41) |
| AUC (ng•h/mL) | 13,185 (36) | 5,512 (48) |
| Dose Proportionality | Dose proportional over 200 mg to 600 mg once daily doses (0.33 to 1.0 times the recommended dose of 600 mg once daily) | |
| Accumulation | ~ 3-fold | |
| Absorption | ||
| T max [Median (range)] (hours) | 4.1 (0.5, 8.15) | |
| Absolute Bioavailability | 12.9% | |
| Effect of Food | ||
| High-Fat Meal b | AUC and C max increased 4-fold | |
| Distribution | ||
| Apparent Volume of Distribution (L) | 30100 (98%) | |
| Protein Binding | 99% (albumin) | |
| Elimination | ||
| Half-Life (hours) | 189.8 (23) | 93.6 (21) |
| Apparent Clearance (L/h) | 41.5 (29) | 101.2 (27) |
| Metabolism | ||
| Primary Pathway | CYP3A | |
| Excretion | ||
| Feces | 86% (73% as unchanged drug) | |
| Urine | 0.5% (0.03% as unchanged drug) | |
Specific Populations
No clinically significant differences in the pharmacokinetics of ziftomenib were observed based on age (18 to 86 years), sex, race, ethnicity, mild (bilirubin less than or equal to ULN and AST greater than ULN or bilirubin less than or equal to 1.5 x ULN) or moderate (bilirubin 1.5 to 3 x ULN) hepatic impairment, or mild to moderate (CL Cr 30 to 89 mL/min) renal impairment.
The effects of severe hepatic impairment (bilirubin greater than 3 x ULN) or severe renal impairment (CL Cr less than 30 mL/min) on ziftomenib pharmacokinetics have not been studied.
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Gastric Acid Reducing Agents: Administration of PPIs reduced ziftomenib AUC 0-inf by 53% and C max by 70%.
Strong, Moderate, or Weak CYP3A4 Inhibitors: Itraconazole, voriconazole, and posaconazole (strong CYP3A4 inhibitors) are estimated to increase ziftomenib AUC by up to 3-fold and C max by up to 2-fold.
Erythromycin, fluconazole, and isavuconazole (moderate CYP3A4 inhibitors) are estimated to increase ziftomenib AUC by up to 2-fold and C max by up to 2-fold.
Cimetidine (weak CYP3A inhibitor) is estimated to increase ziftomenib AUC and C max by up to 1.4-fold.
Strong, Moderate, or Weak CYP3A4 Inducers: Rifampin (strong CYP3A4 inducer) is estimated to decrease ziftomenib AUC by up to 80% and C max by up to 70%.
Efavirenz (moderate CYP3A4 inducer) is estimated to decrease ziftomenib AUC and C max by up to 70%.
Dexamethasone (weak CYP3A4 inducer) is estimated to decrease ziftomenib AUC and C max by up to 40%.
CYP3A4 Substrates: Ziftomenib is predicted to increase the AUC and C max of midazolam (CYP3A4 substrate) up to 1.9- and 1.4-fold, respectively, when ziftomenib is administered alone.
Other Drugs: Ziftomenib is not predicted to have clinically significant effects on the exposure of repaglinide (CYP2C8 substrate), ( S )-warfarin (CYP2C9 substrate), omeprazole (CYP2C19 substrate), and raltegravir (UGT1A1 substrate).
In Vitro Studies
Cytochrome P450 (CYP450) Enzymes: Ziftomenib does not inhibit CYP1A2 or CYP2D6. Ziftomenib induces CYP1A2 but not CYP2B6 or CYP3A4.
UDP-Glucuronosyltransferase (UGT): Ziftomenib inhibits UGT1A9 and UGT2B17.
Transporter Systems: Ziftomenib is a substrate of P-gp and BCRP, but not BSEP or MRP. Ziftomenib does not inhibit P-gp, BCRP, OAT1, OAT3, OCT2, MATE1, MATE2-K, OATP1B1, OATP1B3, BSEP, or MRP2.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with ziftomenib. In a repeat dose toxicity study in mice treated with ziftomenib for 13 weeks, lymphoma was observed in multiple organs in one animal.
Ziftomenib was not mutagenic in an in vitro bacterial reverse mutation assay and was not genotoxic in an in vitro micronucleus assay or in an in vivo mouse bone marrow micronucleus assay.
Fertility studies in animals have not been conducted with ziftomenib. In a repeat dose, 13-week toxicity study in dogs treated with twice daily oral administration of ziftomenib at 6, 20, or 60 mg/kg/day, changes were reported in male and female reproductive organs, including decreased mean testis weight, increased vacuolation of the seminiferous tubular epithelial cells, marked decrease in sperm in the epididymal lumen at ≥ 20 mg/kg/day, and fewer developing follicles in the ovaries at ≥ 60 mg/kg/day. At the dose of 20 mg/kg/day in dogs, exposures (AUC) were as low as 0.3 times the human exposure (AUC) at the recommended dose. At the dose of 60 mg/kg/day, exposures were as low as 1.2 times the human exposure at the recommended dose. Reversibility was not assessed at 20 mg/kg/day and findings were not reversible at 60 mg/kg/day after the 4-week recovery period.
Animal Toxicology and/or Pharmacology
In 13-week repeat-dose toxicity studies in mice and dogs, incidences of hyperplasia in a variety of organs occurred in several animals from both species. The findings in the mouse study were observed at ≥ 12 mg/kg/day at exposures as low as 0.3 times the AUC at the recommended dose, and the findings in the dog study were observed at ≥ 6 mg/kg/day at exposures as low as 0.05 times the AUC at the recommended dose.
CLINICAL STUDIES
The efficacy of KOMZIFTI was evaluated in an open-label, single-arm, multicenter clinical trial (Study KO-MEN-001, NCT04067336; KOMET-001) in 112 adult patients with relapsed or refractory AML with an NPM1 mutation identified using next-generation sequencing or polymerase chain reaction. Patients with NPM1 mutations, including Type A, B, and D mutations and other NPM1 mutations likely to result in cytoplasmic localization of the NPM1 protein, were enrolled. Other eligibility criteria included creatinine clearance ≥ 30 mL/min, total bilirubin ≤ 1.5 x the upper limit of normal (ULN), aminotransferases ≤ 2 x ULN, QTcF ≤ 480 msec, and Eastern Cooperative Oncology Group performance status score of 0-2. KOMZIFTI was given orally at a dose of 600 mg once daily until disease progression or unacceptable toxicity. Patients were permitted to resume treatment with KOMZIFTI following hematopoietic stem cell transplant (HSCT). Four (3.6%) of the 112 patients underwent stem cell transplantation following KOMZIFTI treatment.
The baseline demographic and disease characteristics for the 112 treated patients are shown in Table 6 .
| Demographics and Disease Characteristics | KOMZIFTI (600 mg once daily) N=112 |
|---|---|
| Demographics | |
| Median Age (years) (Range) | 69 (22, 86) |
| Age Categories, n (%) | |
| <65 years | 42 (38) |
| ≥65 years | 70 (63) |
| Sex, n (%) | |
| Male | 49 (44) |
| Female | 63 (56) |
| Race, n (%) | |
| White | 88 (79) |
| Black or African American | 2 (2) |
| Asian | 4 (4) |
| Other | 2 (2) |
| Unknown | 16 (14) |
| Ethnicity, n (%) | |
| Hispanic or Latino | 3 (3) |
| Not Hispanic or Latino | 87 (78) |
| Unknown | 22 (20) |
| Disease Characteristics | |
| Type of AML, n (%) | |
| De novo AML | 95 (85) |
| Secondary AML | 17 (15) |
| Disease status, n (%) | |
| Primary Refractory | 7 (6) |
| Refractory Relapse | 37 (33) |
| Untreated Relapse | 68 (61) |
| Median number of prior lines of therapy (range) | 2 (1, 7) |
| Prior stem cell transplantation, n (%) | 26 (23) |
Efficacy was established based on the rate of complete remission (CR) plus CR with partial hematological recovery (CRh), the duration of CR+CRh, and the rate of conversion from transfusion dependence to transfusion independence. The median follow-up was 4.2 months (range, 0.1 to 41.2 months).
The efficacy results are shown in Table 7 .
| Endpoint | KOMZIFTI (600 mg once daily) N=112 |
|---|---|
CI: confidence interval; the 95% CI rate was calculated using the exact method based on binomial distribution; NE: not estimable; +: censor. | |
a CR is defined as bone marrow blasts <5%, absolute neutrophil count (ANC) > 1.0 x 10 9 /L, and platelet count > 100 x 10 9 /L. | |
b CRh is defined as bone marrow blasts <5%, (ANC) > 0.5 x 10⁹/L; platelet count > 50 x 10 9 /L. | |
c Duration of CR+CRh (DOCR + CRh) is defined as the time from first CR/CRh to the first documented relapse or death, whichever occurs first. | |
d Duration of CR (DOCR) is defined as the time from first CR to the first documented relapse or death, whichever occurs first. | |
e Duration of CRh (DOCRh) is defined as the time from first CRh to the first documented relapse or death, whichever occurs first. | |
| CR a +CRh b , n (%) | 24 (21.4) |
| 95% CI | (14.2, 30.2) |
| Median DOCR+CRh c (months) | 5.0 |
| 95% CI | (1.9, 8.1) |
| CR a , n (%) | 19 (17.0) |
| 95% CI | (10.5, 25.2) |
| Median DOCR d (months) | 5.0 |
| 95% CI | (2.8, 8.1) |
| CRh b , n (%) | 5 (4.5) |
| 95% CI | (1.5, 10.1) |
| Observed DOCRh e (months) | 0.0 + , 1.5 + , 1.5, 1.6, 11.4 |
For patients who achieved a CR or CRh, the median time to first response was 2.7 months (range, 0.9 to 15 months). Of the 24 patients who achieved a response of CR or CRh, 21 (88%) patients did so within 6 months of initiating KOMZIFTI.
Among the 66 patients who were dependent on red blood cell (RBC) and/or platelet transfusions at baseline, 14 (21.2%) became independent of RBC and platelet transfusions during any 56-day post-baseline period. Of the 46 patients who were independent of both RBC and platelet transfusions at baseline, 12 (26.1%) patients remained transfusion independent during any 56-day post-baseline period.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
KOMZIFTI 200 mg capsules are supplied as white capsules printed with “ZIF 200” in black ink.
KOMZIFTI capsules are available in white, induction-sealed, square, high-density polyethylene bottles of 90 capsules with child-resistant closure (NDC 84696-200-90).
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Ziftomenib is a menin inhibitor that blocks the interaction of menin and lysine [K]-specific methyltransferase 2A (KMT2A).
Acute leukemias can be driven by mutations in NPM1 that recruit the wild-type menin-KMT2A complex to promoters of leukemogenic genes. Susceptible NPM1 mutations are defined as those that result in loss of the nucleolar localization signal and the insertion of a new nuclear export signal leading to cytoplasmic accumulation of mutant NPM1 protein that disrupts normal cell function and drives leukemogenesis through altered gene expression.
Pharmacologic disruption of the menin-KMT2A protein-protein interaction by ziftomenib blocks oncogenic activity of mutant NPM1, which induces differentiation of leukemic cells as evidenced by increased expression of differentiation markers. In nonclinical studies, ziftomenib demonstrated in vitro and in vivo antitumor activity in models of NPM1 -mutant leukemia.