Lidocaine - Lidocaine Hydrochloride jelly prescribing information
INDICATIONS AND USAGE
Lidocaine HCl 2% Jelly is indicated for prevention and control of pain in procedures involving the male and female urethra, for topical treatment of painful urethritis, and as an anesthetic lubricant for endotracheal intubation (oral and nasal).
DOSAGE AND ADMINISTRATION
When Lidocaine HCl 2% Jelly is used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind.
The dosage varies and depends upon the area to be anesthetized, vascularity of the tissues, individual tolerance, and the technique of anesthesia. The lowest dosage needed to provide effective anesthesia should be administered. Dosages should be reduced for children and for elderly and debilitated patients. Although the incidence of adverse effects with Lidocaine HCl 2% Jelly is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
For Surface Anesthesia of the Male Adult Urethra: When using Lidocaine 2% Jelly 30 mL tubes, sterilize the plastic cone for 5 minutes in boiling water, cool, and attach to the tube. The cone may be gas sterilized or cold sterilized, as preferred. Slowly instill approximately 15 mL (300 mg of lidocaine HCl) into the urethra or until the patient has a feeling of tension. A penile clamp is then applied for several minutes at the corona. An additional dose of not more than 15 mL (300mg) can be instilled for adequate anesthesia.
Prior to sounding or cystoscopy, a penile clamp should be applied for 5 to 10 minutes to obtain adequate anesthesia. A total dose of 30 mL (600 mg) is usually required to fill and dilate the male urethra. Prior to catheterization, smaller volumes of 5 to 10 mL (100 to 200 mg) are usually adequate for lubrication.
For Surface Anesthesia of the Female Adult Urethra: When using Lidocaine 2% Jelly 30 mL tubes, sterilize the plastic cone for 5 minutes in boiling water, cool, and attach to the tube. The cone may be gas sterilized or cold sterilized, as preferred. Slowly instill 3 to 5 mL (60 to 100 mg of lidocaine HCl) of the jelly into the urethra. If desired, some jelly may be deposited on a cotton swab and introduced into the urethra. In order to obtain adequate anesthesia, several minutes should be allowed prior to performing urological procedures.
Lubrication for Endotracheal Intubation: Apply a moderate amount of jelly to the external surface of the endotracheal tube shortly before use. Care should be taken to avoid introducing the product into the lumen of the tube. Do not use the jelly to lubricate endotracheal stylettes (see WARNINGS and ADVERSE REACTIONS ) concerning rare reports of inner lumen occlusion. It is also recommended that use of endotracheal tubes with dried jelly on the external surface be avoided for lack of lubricating effect.
CONTRAINDICATIONS
Lidocaine is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type or to other components of Lidocaine HCl 2% Jelly.
ADVERSE REACTIONS
Adverse experiences following the administration of lidocaine are similar in nature to those observed in other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Central Nervous System: CNS manifestations are excitatory and/or depressant and may be characterized by light headedness: nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression, and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest.
Drowsiness following the administration of lidocaine is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption.
Cardiovascular System: Cardiovascular manifestations are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse which may lead to cardiac arrest.
Allergic: Allergic reactions are characterized by cutaneous lesions, urticaria, edema, or anaphylactoid reactions. Allergic reactions may occur as a result of sensitivity either to the local anesthetic agent or other components in the formulation. Allergic reactions as a result of sensitivity to lidocaine are extremely rare and, if they occur, should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
To report SUSPECTED ADVERSE REACTIONS, contact Sentiss at 1-855-473-6847 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
| Class | Examples |
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastics Agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | Phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
Carcinogenesis - Long-term studies in animals have not been performed to evaluate the carcinogenic potential of lidocaine.
Mutagenesis - The mutagenic potential of lidocaine has been tested in the Ames Salmonella reverse mutation assay, and an in vitro chromosome aberrations assay in human lymphocytes and in an in vivo mouse micronucleus assay. There was no indication of any mutagenic effect in these studies.
Impairment of Fertility - The effect of lidocaine on fertility was examined in the rat model. Administration of 30 mg/kg, s.c. (180 mg/m 2 ) to the mating pair did not produce alterations in fertility or general reproductive performance of rats. There are no studies that examine the effect of lidocaine on sperm parameters. There was no evidence of altered fertility.
DESCRIPTION
Lidocaine HCl 2% Jelly is a sterile aqueous product that contains a local anesthetic agent and is administered topically (see INDICATIONS AND USAGE for specific uses).
Lidocaine HCl 2% Jelly contains lidocaine HCl which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the following structural formula:
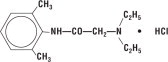
Its molecular formula is C 14 H 22 N 2 O • HCl and its molecular weight is 270.80.
Lidocaine HCl 2% Jelly also contains hypromellose, and the resulting mixture maximizes contact with mucosa and provides lubrication for instrumentation. The unused portion should be discarded after initial use.
Composition of Lidocaine HCl 2% Jelly 30 mL and 5 mL tubes: Each mL contains 20 mg of lidocaine HCl. The formulation also contains methylparaben, propylparaben, hypromellose, and sodium hydroxide and/or hydrochloric acid to adjust pH to 6.0 to 7.0.
CLINICAL PHARMACOLOGY
Mechanism of Action: Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Onset of Action: The onset of action is 3 to 5 minutes. It is ineffective when applied to intact skin.
Hemodynamics: Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system.
Pharmacokinetics and Metabolism: Lidocaine may be absorbed following topical administration to mucous membranes, its rate and extent of absorption depending upon concentration and total dose administered, the specific site of application, and duration of exposure. In general, the rate of absorption of local anesthetic agents following topical application occurs most rapidly after intratracheal administration. Lidocaine is also well-absorbed from the gastrointestinal tract, but little intact drug may appear in the circulation because of biotransformation in the liver.
Lidocaine is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged.
The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL, 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Studies of lidocaine metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2.0 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged twofold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
HOW SUPPLIED
Lidocaine HCl 2% Jelly is supplied in the listed dosage forms.
| NDC 72485-611-30 | 30 mL aluminum tube single pack |
| NDC 72485-611-10 | 5 mL aluminum tube single pack |
| NDC 72485-611-31 | 5 mL aluminum tube ten pack |
A detachable applicator cone and a key for expressing the contents are included in the 30 mL carton.
Storage: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Distributed by:
Armas Pharmaceuticals, Inc.
Freehold, NJ 07728 USA
Manufactured by:
Ophtapharm AG Riethofstrasse 1, Hettlingen, 8442, Switzerland (CHE) for Sentiss
Made in Switzerland 83027101 Rev. 04/24
Mechanism of Action: Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Onset of Action: The onset of action is 3 to 5 minutes. It is ineffective when applied to intact skin.
Hemodynamics: Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system.