Get your patient on Metformin - Metformin Er 500 Mg tablet, Extended Release (Metformin Er 500 Mg)
Metformin - Metformin Er 500 Mg tablet, Extended Release prescribing information
INDICATIONS & USAGE
Metformin hydrochloride extended-release tablets, USP are indicated as an adjunct to diet and exercise to improve glycemic control in patients with type 2 diabetes mellitus.
DOSAGE & ADMINISTRATION
There is no fixed dosage regimen for the management of hyperglycemia in patients with type 2 diabetes with metformin hydrochloride extended-release tablets, USP or any other pharmacologic agent. Dosage of metformin hydrochloride extended-release tablets, USP must be individualized on the basis of both effectiveness and tolerance, while not exceeding the maximum recommended daily doses. The maximum recommended daily dose of metformin hydrochloride extended-release tablets, USP in adults is 2000 mg.
Metformin hydrochloride extended-release tablets, USP should generally be given once daily with the evening meal. Metformin hydrochloride extended-release tablets, USP should be started at a low dose, with gradual dose escalation, both to reduce gastrointestinal side effects and to permit identification of the minimum dose required for adequate glycemic control of the patient.
During treatment initiation and dose titration (see Recommended Dosing Schedule), fasting plasma glucose should be used to determine the therapeutic response to metformin hydrochloride extended-release tablets, USP and identify the minimum effective dose for the patient. Thereafter, glycosylated hemoglobin should be measured at intervals of approximately 3 months. The therapeutic goal should be to decrease both fasting plasma glucose and glycosylated hemoglobin levels to normal or near normal by using the lowest effective dose of metformin hydrochloride extended-release tablets, USP either when used as monotherapy or in combination with sulfonylurea or insulin.
Monitoring of blood glucose and glycosylated hemoglobin will also permit detection of primary failure, i.e., inadequate lowering of blood glucose at the maximum recommended dose of medication, and secondary failure, i.e., loss of an adequate blood glucose lowering response after an initial period of effectiveness.
Short-term administration of metformin hydrochloride extended-release tablets, USP may be sufficient during periods of transient loss of control in patients usually well-controlled on diet alone.
Metformin hydrochloride extended-release tablets, USP must be swallowed whole and never crushed or chewed. Occasionally, the inactive ingredients of metformin hydrochloride extended-release tablets, USP will be eliminated in the feces as a soft, hydrated mass. (See Patient Information printed below.)
Recommended Dosing Schedule
Adults In general, clinically significant responses are not seen at doses below 1500 mg per day. However, a lower recommended starting dose and gradually increased dosage is advised to minimize gastrointestinal symptoms.
The usual starting dose of metformin hydrochloride extended-release tablets, USP is 500 mg once daily with the evening meal. Dosage increases should be made in increments of 500 mg weekly, up to a maximum of 2000 mg once daily with the evening meal. If glycemic control is not achieved on metformin hydrochloride extended-release tablets, USP 2000 mg once daily, a trial of metformin hydrochloride extended-release tablets, USP 1000 mg twice daily should be considered. (See CLINICAL PHARMACOLOGY : Clinical Studies .)
In a randomized trial, patients currently treated with metformin hydrochloride tablets were switched to metformin hydrochloride extended-release tablets. Results of this trial suggest that patients receiving metformin hydrochloride tablets treatment may be safely switched to metformin hydrochloride extended-release tablets once daily at the same total daily dose, up to 2000 mg once daily. Following a switch from metformin hydrochloride tablets to metformin hydrochloride extended-release tablets, glycemic control should be closely monitored and dosage adjustments made accordingly (see CLINICAL PHARMACOLOGY : Clinical Studies ).
Pediatrics Safety and effectiveness of metformin hydrochloride extended-release tablets, USP in pediatric patients have not been established.
Recommendations for Use in Renal Impairment
Assess renal function prior to initiation of metformin hydrochloride extended-release tablets and periodically thereafter. Metformin hydrochloride extended-release tablets are contraindicated in patients with an estimated glomerular filtration rate (eGFR) below 30 mL/minute/1.73 m2. Initiation of metformin hydrochloride extended-release tablets in patients with an eGFR between 30 to 45 mL/minute/1.73 m2 is not recommended. In patients taking metformin hydrochloride extended-release tablets whose eGFR later falls below 45 mL/min/1.73 m2, assess the benefit risk of continuing therapy. Discontinue metformin hydrochloride extended-release tablets if the patients eGFR later falls below 30 mL/minute/1.73 m2 (See WARNINGS and PRECAUTIONS ).
Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of liver disease, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart metformin hydrochloride extended-release tablets if renal function is stable.
Concomitant Metformin Hydrochloride Extended-Release Tablets, USP and Oral Sulfonylurea Therapy in Adult Patients
If patients have not responded to 4 weeks of the maximum dose of metformin hydrochloride extended-release tablets monotherapy, consideration should be given to gradual addition of an oral sulfonylurea while continuing metformin hydrochloride extended-release tablets, USP at the maximum dose, even if prior primary or secondary failure to a sulfonylurea has occurred. Clinical and pharmacokinetic drug- drug interaction data are currently available only for metformin plus glyburide (glibenclamide).
With concomitant metformin hydrochloride extended-release tablets and sulfonylurea therapy, the desired control of blood glucose may be obtained by adjusting the dose of each drug. In a clinical trial of patients with type 2 diabetes and prior failure on glyburide, patients started on metformin hydrochloride tablets 500 mg and glyburide 20 mg were titrated to 1000/20 mg, 1500/20 mg, 2000/20 mg or 2500/20 mg of metformin hydrochloride tablets and glyburide, respectively, to reach the goal of glycemic control as measured by FPG, HbA1c and plasma glucose response (see CLINICAL PHARMACOLOGY : Clinical Studies ). However, attempts should be made to identify the minimum effective dose of each drug to achieve this goal. With concomitant metformin hydrochloride extended-release tablets, USP and sulfonylurea therapy, the risk of hypoglycemia associated with sulfonylurea therapy continues and may be increased. Appropriate precautions should be taken. (See Package Insert of the respective sulfonylurea.)
If patients have not satisfactorily responded to 1 to 3 months of concomitant therapy with the maximum dose of metformin hydrochloride extended-release tabletsand the maximum dose of an oral sulfonylurea, consider therapeutic alternatives including switching to insulin with or without metformin hydrochloride extended-release tablets.
Concomitant Metformin Hydrochloride Extended-Release Tablets and Insulin Therapy in Adult Patients
The current insulin dose should be continued upon initiation of metformin hydrochloride extended-release tablets therapy. Metformin hydrochloride extended-release tablets therapy should be initiated at 500 mg once daily in patients on insulin therapy. For patients not responding adequately, the dose of metformin hydrochloride extended-release tablets should be increased by 500 mg after approximately 1 week and by 500 mg every week thereafter until adequate glycemic control is achieved. The maximum recommended daily dose is 2000 mg for metformin hydrochloride extended-release tablets. It is recommended that the insulin dose be decreased by 10% to 25% when fasting plasma glucose concentrations decrease to less than 120 mg/dL in patients receiving concomitant insulin and metformin hydrochloride extended-release tablets, USP. Further adjustment should be individualized based on glucose-lowering response.
Specific Patient Populations
Metformin hydrochloride extended-release tablets is not recommended for use in pregnancy. Metformin hydrochloride extended-release tablets, USP is not recommended in pediatric patients (below the age of 17 years).
The initial and maintenance dosing of metformin hydrochloride extended-release tablets should be conservative in patients with advanced age, due to the potential for decreased renal function in this population. Any dosage adjustment should be based on a careful assessment of renal function. Generally, elderly, debilitated, and malnourished patients should not be titrated to the maximum dose of metformin hydrochloride extended-release tablets.
Monitoring of renal function is necessary to aid in prevention of lactic acidosis, particularly in the elderly. (See WARNINGS .)
CONTRAINDICATIONS
Metformin hydrochloride extended-release tablets, USP are contraindicated in patients with:
1. Severe Renal Impairment (eGFR below 30mL/min/1.73m2) (see WARNINGS and PRECAUTIONS ).
2.Known hypersensitivity to metformin hydrochloride, USP.
3.Acute or chronic metabolic acidosis, including diabetic ketoacidosis, with or without coma. Diabetic ketoacidosis should be treated with insulin.
ADVERSE REACTIONS
In A US double blind clinical study of metformin hydrochloride tablets in patients with type 2 diabetes, a total of 141 patients received metformin hydrochloride tablets therapy (up to 2550 mg per day) and 145 patients received placebo. Adverse reactions reported in greater than 5% of the metformin hydrochloride tablets patients, and that were more common in metformin hydrochloride tablets than placebo treated patients, are listed in Table 6.
Table 6: Most Common Adverse Reactions (>5 Percent) in a Placebo-Controlled Clinical Study of Metformin Hydrochloride Tablets Monotherapy• | ||
Adverse Reaction | Metformin Hydrochloride Tablets (n=141) | Placebo (n=145) |
% of Patients | ||
| Diarrhea | 53.2 | 11.7 |
Nausea/Vomiting | 25.5 | 8.3 |
| Flatulence | 12.1 | 5.5 |
Asthenia | 9.2 | 5.5 |
| Indigestion | 7.1 | 4.1 |
Abdominal Discomfort | 6.4 | 4.8 |
Headache | 5.7 | 4.8 |
•Reactions that were more common in metformin hydrochloride tablets than placebo treated patients.
Diarrhea led to discontinuation of study medication in 6% of patients treated with metformin hydrochloride tablets. Additionally, the following adverse reactions were reported in ≥1 - ≤5% of metformin hydrochloride tablets patients and myalgia, lightheaded, dyspnea, nail disorder, rash, sweating increased, taste disorder, chest discomfort, chills, flu syndrome, flushing, and palpitation.
In worldwide clinical trials over 900 patients with type 2 diabetes have been treated with metformin hydrochloride extended-release tablets in placebo and active controlled studies. In placebo controlled trails, 781 patients were administered metformin hydrochloride extended-release tablets and 195 patients received placebo. Adverse reactions reported in greater than 5% of the metformin hydrochloride extended release tablets patients, and that were more common in metformin hydrochloride extended-release tablets than placebo treated patients are listed in Table 7.
Table 7: Most Common Adverse Reactions (>5 Percent) in Placebo-Controlled Studies of Metformin Hydrochloride Extended-Release Tablets• | ||
Metformin Hydrochloride Extended-Release Tablets(N=781) | Placebo (N=195) | |
Adverse Reactions | % of patients | |
| Diarrhea | 9.6 | 2.6 |
Nausea/Vomiting | 6.5 | 1.5 |
Diarrhea led to discontinuation of study medication in 0.6% of patients treated with metformin hydrochloride extended-release tablets. Additionally, the following adverse reactions were reported in ≥1 - ≤5% of metformin hydrochloride extended-release tablets patients and were more commonly reported with metformin hydrochloride extended-release tablets than placebo: abdominal pain, constipation, distention abdomen, dyspepsia/heartburn, flatulence, dizziness, headache, upper respiratory infection, taste disturbance.•Reactions that were more common in metformin hydrochloride extended-release tablets than placebo treated patients.
DESCRIPTION
Metformin Hydrochloride Extended-Release Tablets, USP
Metformin hydrochloride extended-release tablets, USP are oral antihyperglycemic drugs used in the management of type 2 diabetes. Metformin hydrochloride, USP (N,N-dimethyl-monohydrochloride,Imidodicarbonimidic diamide) is not chemically or pharmacologically related to any other classes of oral antihyperglycemic agents. The structural formula is as shown:
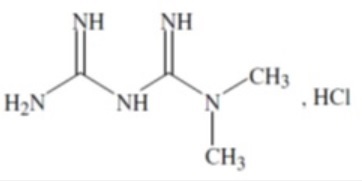
Metformin hydrochloride, USP is a white or almost white, crystalline powder with a molecular formula of C4H11N5•HCl and a molecular weight of 165.62. Metformin hydrochloride is freely soluble in water, slightly soluble in alcohol, practically insoluble in acetone and in Methylene chloride. The pKa of metformin is 12.4. The pH of a 1% aqueous solution of metformin hydrochloride is 6.35. Metformin hydrochloride extended-release tablets, USP contain 500 mg or 750 mg of metformin hydrochloride as the active ingredient. Metformin hydrochloride extended-release tablets, USP 500 mg and 750 mg tablets contain the inactive ingredients hypromellose, magnesium stearate, and polyvinyl pyrrolidone. Dissolution Method: Test 10 System Components and Performance - Metformin hydrochloride extended-release tablets, USP comprises a dual hydrophilic polymer matrix system. Metformin hydrochloride, USP is combined with a drug release controlling polymer to form an "inner" phase, which is then incorporated as discrete particles into an "external" phase of a second polymer. After administration, fluid from the gastrointestinal (GI) tract enters the tablet, causing the polymers to hydrate and swell. Drug is released slowly from the dosage form by a process of diffusion through the gel matrix that is essentially independent of pH. The hydrated polymer system is not rigid and is expected to be broken up by normal peristalsis in the GI tract. The biologically inert components of the tablet may occasionally remain intact during GI transit and will be eliminated in the feces as a soft, hydrated mass.
CLINICAL PHARMACOLOGY
Mechanism of Action
Metformin is an antihyperglycemic agent which improves glucose tolerance in patients with type 2 diabetes, lowering both basal and postprandial plasma glucose. Its pharmacologic mechanisms of action are different from other classes of oral antihyperglycemic agents. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. Unlike sulfonylureas, metformin does not produce hypoglycemia in either patients with type 2 diabetes or normal subjects (except in special circumstances, see PRECAUTIONS ) and does not cause hyperinsulinemia. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may actually decrease.
Pharmacokinetics
Absorption and Bioavailability
Following a single oral dose of Metformin Hydrochloride Extended-Release Tablets, C max is achieved with a median value of 7 hours and a range of 4 to 8 hours.Peak plasma levels are approximately 20% lower compared to the same dose of metformin hydrochloride tablets, however, the extent of absorption (as measured by AUC) is similar to metformin hydrochloride tablets. At steady state, the AUC and C max are less than dose proportional for metformin hydrochloride extended-release tablets within the range of 500 to 2000 mg administered once daily. Peak plasma levels are approximately 0.6, 1.1, 1.4, and 1.8 mcg/mL for 500, 1000, 1500, and 2000 mg once-daily doses, respectively. The extent of metformin absorption (as measured by AUC) from metformin hydrochloride extended-release tablets at a 2000 mg once-daily dose is similar to the same total daily dose administered as metformin hydrochloride tablets 1000 mg twice daily. After repeated administration of metformin hydrochloride extended-release tablets, metformin did not accumulate in plasma. Within-subject variability in C max and AUC of metformin from metformin hydrochloride extended-release tablets is comparable to that with metformin hydrochloride tablets. Although the extent of metformin absorption (as measured by AUC) from the metformin hydrochloride extended-release tablets increased by approximately 50% when given with food, there was no effect of food on C max and T max of metformin. Both high and low fat meals had the same effect on the pharmacokinetics of metformin hydrochloride extended-release tablets. Distribution The apparent volume of distribution (V/F) of metformin following single oral doses of metformin hydrochloride 850 mg averaged 654 ± 358 L. Metformin is negligibly bound to plasma proteins, in contrast to sulfonylureas, which are more than 90% protein bound. Metformin partitions into erythrocytes, most likely as a function of time. At usual clinical doses and dosing schedules of metformin hydrochloride tablets, steady state plasma concentrations of metformin are reached within 24 to 48 hours and are generally <1 mcg/mL. During controlled clinical trials of metformin hydrochloride tablets, maximum metformin plasma levels did not exceed 5 mcg/mL, even at maximum doses.
Metabolism and Elimination
Intravenous single-dose studies in normal subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) nor biliary excretion. Renal clearance (see Table 1 ) is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Specific Population
Patients with Type 2 Diabetes
In the presence of normal renal function, there are no differences between single- or multiple-dose pharmacokinetics of metformin between patients with type 2 diabetes and normal subjects (see Table 1) nor is there any accumulation of metformin in either group at usual clinical doses.
The pharmacokinetics of metformin hydrochloride extended-release tablets in patients with type 2 diabetes is comparable to those in healthy normal adults.
Renal Impairment
In patients with decreased renal function (based on measured creatinine clearance), the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased in proportion to the decrease in creatinine clearance (see Table 1: also see also see CONTRAINDICATIONS , WARNINGS , PRECAUTIONS , and DOSAGE AND ADMINISTRATION ).
Hepatic Impairment
No parmacokinetic studies of metformin have been conducted in patients with hepatic insufficiency (see PRECAUTIONS ).
Geriatrics
Limited data from controlled pharmacokinetic studies of metformin hydrochloride tablets in healthy elderly subjects suggest that total plasma clearance of metformin is decreased, the half-life is prolonged, and Cmax is increased, compared to healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function (see Table 1; also see WARNINGS , PRECAUTIONS , and DOSAGE AND ADMINISTRATION ). Metformin hydrochloride extended-release tablets treatment should not be initiated in patients ≥80 years of age unless measurement of creatinine clearance demonstrates that renal function is not reduced (see WARNINGS and DOSAGE AND ADMINISTRATION ).
| Table 1: Select Mean (± S.D.) Metformin Pharmacokinetic Parameters Following Single or Multiple Oral Doses of Metformin Hydrochloride Tablets | |||
| Subject Groups: Metformin Hydrochloride Tablet dose a (number of subjects) | Cmax b (mcg/mL) | Tmax c (hrs) | Renal Clearance (mL/min) |
| Healthy, nondiabetic adults: | |||
| 500 mg single dose (24) | 1.03 (±0.33) | 2.75 (±0.81) | 600 (±132) |
| 850 mg single dose (74)d | 1.60 (±0.38) | 2.64 (±0.82) | 552 (±139) |
| 850 mg three times daily for 19 doses e (9) | 2.01 (±0.42) | 1.79 (±0.94) | 642 (±173) |
| Adults with type 2 diabetes: | |||
| 850 mg single dose (23) | 1.48 (±0.5) | 3.32 (±1.08) | 491 (±138) |
| 850 mg three times daily for 19 dosese (9) | 1.90 (±0.62) | 2.01 (±1.22) | 550 (±160) |
| Elderly f , healthy nondiabetic adults: | |||
| 850 mg single dose (12) | 2.45 (±0.70) | 2.71 (±1.05) | 412 (±98) |
| Renal-impaired adults: | |||
| 850 mg single dose | |||
| Mild (CL cr g 61-90 mL/min) (5) | 1.86 (±0.52) | 3.20 (±0.45) | 384 (±122) |
| Moderate (CL cr 31-60 mL/min) (4) | 4.12 (±1.83) | 3.75 (±0.50) | 108 (±57) |
| Severe (CL cr 10-30 mL/min) (6) | 3.93 (±0.92) | 4.01 (±1.10) | 130 (±90) |
a All doses given fasting except the first 18 doses of the multiple dose studies
b Peak plasma concentration
c Time to peak plasma concentration
d Combined results (average means) of five studies: mean age 32 years (range 23-59 years)
e Kinetic study done following dose 19, given fasting
f Elderly subjects, mean age 71 years (range 65-81 years)
g CLcr = creatinine clearance normalized to body surface area of 1.73 m 2
Gender Metformin pharmacokinetic parameters did not differ significantly between normal subjects and patients with type 2 diabetes when analyzed according to gender (males = 19, females = 16). Race
No studies of metformin pharmacokinetic parameters according to race have been performed.
Clinical Studies
Metformin Hydrochloride Extended-Release Tablets
A 24-week, double-blind, placebo-controlled study of metformin hydrochloride extended-release tablets, taken once daily with the evening meal, was conducted in patients with type 2 diabetes who had failed to achieve glycemic control with diet and exercise (HbA1c 7 to 10%, FPG 126 to 270 mg/dL). Patients entering the study had a mean baseline HbA1c of 8% and a mean baseline FPG of 176 mg/dL. After 12 weeks treatment, mean HbA 1c had increased from baseline by 0.1% and mean FPG decreased from baseline by 2 mg/dL in the placebo group, compared with a decrease in mean HbA 1c of 0.6% and a decrease in mean FPG of 23 mg/dL in patients treated with metformin hydrochloride extended-release tablets 1000 mg once daily. Subsequently, the treatment dose was increased to 1500 mg once daily if HbA 1c was ≥7% but <8% (patients with HbA 1c ≥8% were discontinued from the study). At the final visit (24-week), mean HbA 1c had increased 0.2% from baseline in placebo patients and decreased 0.6% with metformin hydrochloride extended-release tablets.
A 16-week, double-blind, placebo-controlled, dose-response study of metformin hydrochloride extended-release tablets, taken once daily with the evening meal or twice daily with meals, was conducted in patients with type 2 diabetes who had failed to achieve glycemic control with diet and exercise (HbA 1c 7 to 11%, FPG 126 to 280 mg/dL). Changes in glycemic control and body weight are shown in Table 2 .
| Table 2:Sum mary of Mean Ch a nges f rom Baseline• in Hb A1c, Fasting Plasma Glucose, and Body Weight at Final Visit (16-week study) | ||||||
| Metformin Hydrochloride Extended–Release Tablets | Placebo | |||||
| 500 mg once Daily | 1000 mg once Daily | 1500 mg once Daily | 2000 mg once Daily | 1000 mg Twice Daily | ||
| H e m og l o bi n A1c (%) Baseline Change at FINAL VISIT p-value a | ( n= 115) 8.2 -0.4 <0.001 | ( n= 115) 8.4 -0.6 <0.001 | ( n= 111) 8.3 -0.9 <0.001 | ( n= 125) 8.4 -0.8 <0.001 | ( n= 112) 8.4 -1.1 <0.001 | ( n= 111) 8.4 0.1 - |
| FP G (mg/dL) Baseline Change at FINAL VISIT p-value a | ( n= 126) 182.7 -15.2 <0.001 | ( n= 118) 183.7 -19.3 <0.001 | ( n= 120) 178.9 -28.5 <0.001 | ( n= 132) 181 -29.9 <0.001 | ( n= 122) 181.6 -33.6 <0.001 | ( n= 113) 179.6 7.6 - |
| Bo dy Weight (lbs) Baseline Change at FINAL VISIT p-value a | ( n= 125) 192.9 -1.3 NS•• | ( n= 119) 191.8 -1.3 NS•• | ( n= 117) 188.3 -0.7 NS•• | ( n= 131) 195.4 -1.5 NS•• | ( n= 119) 192.5 -2.2 NS•• | ( n= 113) 194.3 -1.8 - |
•All patients on diet therapy at Baseline
a All comparisons versus Placebo
••Not statistically significant
Compared with placebo, improvement in glycemic control was seen at all dose levels of metformin hydrochloride extended-release tablets and treatment was not associated with any significant change in weight (see DOSAGE AND ADMINISTRATION ) for dosing recommendations for metformin hydrochloride extended-release tablets.
A 24-week, double-blind, randomized study of metformin hydrochloride extended-release tablets, taken once daily with the evening meal, and metformin hydrochloride tablets, taken twice daily (with breakfast and evening meal), was conducted in patients with type 2 diabetes who had been treated with metformin hydrochloride 500 mg twice daily for at least 8 weeks prior to study entry.
The metformin hydrochloride dose had not necessarily been titrated to achieve a specific level of glycemic control prior to study entry. Patients qualified for the study if HbA 1c was ≤8.5% and FPG was ≤200 mg/dL. Changes in glycemic control and body weight are shown in Table 3 .
| Table3: Summary of Mean Changes from Baseline• in HbA1c, Fasting Plasma Glucose, and Body Weight at Week 12 and at Final Visit (24-week study) | |||
|---|---|---|---|
| Metformin Hydrochloride Tablets 500 mg Twice Daily | Metformin Hydrochloride Extended-Release Tablets | ||
| 1000 mg Once Daily | 1500 mg Once Daily | ||
| Hemoglobin A1c (%) | (n=67) | (n=72) | (n=66) |
| Baseline | 7.06 | 6.99 | 7.02 |
| Change at 12 Weeks | 0.14 | 0.23 | 0.04 |
| (95% CI) | (-0.03, 0.31) | (0.10, 0.36) | (-0.08, 0.15) |
| Change at FINAL VISIT | 0.14 a | 0.27 | 0.13 |
| (95% CI) | (-0.04, 0.31) | (0.11, 0.43) | (-0.02, 0.28) |
| FPG (mg/dL) | (n=69) | (n=72) | (n=70) |
| Baseline | 127.2 | 131 | 131.4 |
| Change at 12 Weeks | 12.9 | 9.5 | 3.7 |
| (95% CI) | (6.5, 19.4) | (4.4, 14.6) | (-0.4, 7.8) |
| Change at FINAL VISIT | 14 | 11.5 | 7.6 |
| (95% CI) | (7, 21) | (4.4, 18.6) | (1.0, 14.2) |
| Body Weight (lbs) | (n=71) | (n=74) | (n=71) |
| Baseline | 210.3 | 202.8 | 192.7 |
| Change at 12 Weeks | 0.4 | 0.9 | 0.7 |
| (95% CI) | (-0.4, 1.5) | (0.0, 2) | (-0.4, 1.8) |
| Change at FINAL VISIT | 0.9 | 1.1 | 0.9 |
| (95% CI) | (-0.4, 2.2) | (-0.2, 2.4) | (-0.4, 2) |
• All patients on metformin hydrochloride tablets 500 mg twice daily at Baseline
a n=68
After 12 weeks of treatment, there was an increase in mean HbA 1c in all groups; in the metformin hydrochloride extended-release tablets 1000 mg group, the increase from baseline of 0.23% was statistically significant (see DOSAGE AND ADMINISTRATION ).
Changes in lipid parameters in the previously described placebo-controlled dose-response study of metformin hydrochloride extended-release tablets are shown in Table 4.
| T able 4:Summary of Mean Percent Changes from Baseline• in Major Lipid Variables at Final Visit (16-week study) | ||||||
| Metformin Hydrochloride Extended–Release Tablets | Placebo | |||||
| 500 mg once Daily | 1000 mg once Daily | 1500 mg once Daily | 2000 mg once Daily | 1000 mg Twice Daily | ||
| T o tal Cholesterol (mg/dL) Baseline Mean % Change at FINAL VISIT | ( n= 120) 210.3 1% | ( n= 113) 218.1 1.7% | ( n= 110) 214.6 0.7% | ( n= 126) 204.4 -1.6% | ( n= 117) 208.2 -2.6% | ( n= 110) 208.6 -2.6% |
| T o tal Triglyceides (mg/dL) Baseline Mean % Change at FINAL VISIT | ( n= 120) 220.2 14.5% | ( n= 113) 211.9 9.4% | ( n= 110) 198 15.1% | ( n= 126) 194.2 14.9% | ( n= 117) 179 9.4% | ( n= 110) 211.7 10.9% |
| LDL Cholesterol (mg/dL) Baseline Mean % Change at FINAL VISIT | ( n= 119) 131 -1.4% | ( n= 113) 134.9 -1.6% | ( n= 109) 135.8 -3.5% | ( n= 126) 125.8 -3.3% | ( n= 117) 131.4 -5.5% | ( n= 107) 131.9 -3.2% |
| HDL Cholesterol (mg/dL) Baseline Mean % Change at FINAL VISIT | ( n= 120) 40.8 6.2% | ( n= 108) 41.6 8.6% | ( n= 108) 40.6 5.5% | ( n= 125) 40.2 6.1% | ( n= 117) 42.4 7.1% | ( n= 108) 39.4 5.8% |
•All patients on diet therapy at Baseline
Changes in lipid parameters in the previously described study of metformin hydrochloride tablets and metformin hydrochloride extended-release tablets are shown in Table 5.
| Table 5: Summary of Mean Percent Changes from Baseline• in Major Lipid Variables at Final Visit (24 week study) | |||
| Metformin Hydrochloride Tablets 500 mg Twice Daily | Metformin Hydrochloride Extended-Release Tablets | ||
| 1000 mg Once Daily | 1500 mg Once Daily | ||
| Total Cholesterol (mg/dL) | (n=68) | (n=70) | (n=66) |
| Baseline | 199 | 201.9 | 201.6 |
| Mean % Change at FINAL VISIT | 0.1% | 1.3% | 0.1% |
| Total Triglycerides (mg/dL) | (n=68) | (n=70) | (n=66) |
| Baseline | 178 | 169.2 | 206.8 |
| Mean % Change at FINAL VISIT | 6.3% | 25.3% | 33.4% |
| LDL-Cholesterol (mg/dL) | (n=68) | (n=70) | (n=66) |
| Baseline | 122.1 | 126.2 | 115.7 |
| Mean % Change at FINAL VISIT | -1.3% | -3.3% | -3.7% |
| HDL-Cholesterol (mg/dL) | (n=68) | (n=70) | (n=65) |
| Baseline | 41.9 | 41.7 | 44.6 |
| Mean % Change at FINALVISIT | 4.8% | 1% | -2.1% |
•All patients on metformin hydrochloride tablets 500 mg twice daily at baseline
HOW SUPPLIED
Metformin Hydrochloride Extended-Release Tablets, USP
500 mg Bottles of 30 NDC 70010-491-03
500 mg Bottles of 90 NDC 70010-491-09
500 mg Bottles of 100 NDC 70010-491-01
500 mg Bottles of 500 NDC 70010-491-05
500 mg Bottles of 1000 NDC 70010-491-10
750 mg Bottles of 30 NDC 70010-492-03
750 mg Bottles of 90 NDC 70010-492-09
750 mg Bottles of 100 NDC 70010-492-01
750 mg Bottles of 500 NDC 70010-492-05
750 mg Bottles of 1000 NDC 70010-492-10
Metformin Hydrochloride Extended-Release Tablets, USP 500 mg tablets are white to off-white uncoated, modified capsule shaped tablets debossed with “G7” on one side and plain on the other side.
Metformin Hydrochloride Extended-Release Tablets, USP 750 mg tablets are white to off-white uncoated, modified capsule shaped tablets debossed with “G8” on one side and plain on the other side. Storage
Store at 20° to 25° C (68° to 77° F); excursions permitted within 15° to 30° C (59° to 86° F). [See USP Controlled Room Temperature.]
Dispense in light-resistant containers. Dispense with Patient Information available at: https://granulespharma.com/product/metformin-er-tab-500-750/ Manufactured by:
Granules India Limited
Hyderabad-500 081, India Manufactured for:
Granules Pharmaceuticals Inc.
Chantilly, VA 20151. Revision date: May 2023