Get your patient on Metronidazole
Metronidazole prescribing information
INDICATIONS AND USAGE
Metronidazole Gel is indicated for topical application in the treatment of inflammatory papules and pustules of rosacea.
DOSAGE AND ADMINISTRATION
Apply and rub in a thin film of metronidazole gel twice daily, morning and evening, to entire affected areas after washing.
Areas to be treated should be cleansed before application of metronidazole gel. Patients may use cosmetics after application of metronidazole gel.
CONTRAINDICATIONS
Metronidazole Gel is contraindicated in individuals with a history of hypersensitivity to metronidazole, parabens or other ingredients of the formulation.
ADVERSE REACTIONS
The following adverse experiences have been reported with the topical use of metronidazole: burning, skin irritation, dryness, transient redness, metallic taste, tingling or numbness of extremities and nausea.
T o report SUSPECTED ADVERSE REACTIONS, contact Viona Pharmaceuticals Inc.
at 1-888-304-5011 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
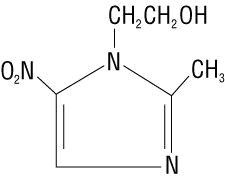
Metronidazole Gel USP, 0.75% contains metronidazole, USP, at a concentration of 7.5 mg per gram (0.75%) in a gel consisting of carbomer homopolymer , edetate disodium dihydrate, methylparaben, propylene glycol, propylparaben, purified water, and sodium hydroxide. Metronidazole is classified therapeutically as an antiprotozoal and antibacterial agent. Chemically, metronidazole is named 2-methyl-5-nitro-1 H -imidazole-1-ethanol and has the following structure:
CLINICAL PHARMACOLOGY
Bioavailability studies on the topical administration of 1 gram of metronidazole Gel (7.5 mg of metronidazole) to the face of 10 rosacea patients showed a maximum serum concentration of 66 nanograms per milliliter in one patient. This concentration is approximately 100 times less than concentrations afforded by a single 250 mg oral tablet. The serum metronidazole concentrations were below the detectable limits of the assay at the majority of time points in all patients. Three of the patients had no detectable serum concentrations of metronidazole at any time point. The mean dose of gel applied during clinical studies was 600 mg which represents 4.5 mg of metronidazole per application. Therefore, under normal usage levels, the formulation affords minimal serum concentrations of metronidazole. The mechanisms by which metronidazole gel acts in the treatment of rosacea are unknown, but appear to include an antiinflammatory effect.
HOW SUPPLIED
Metronidazole Gel USP, 0.75% is supplied in a 45 g tube
NDC 72578-209-04
Storage conditions:
Store at 20° C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to
86°F). [see USP Controlled Room Temperature].
Keep this and all drugs out of the reach of children.