Get your patient on Mydriacyl - Tropicamide solution/ Drops (Tropicamide)
Mydriacyl - Tropicamide solution/ Drops prescribing information
INDICATIONS AND USAGE
For mydriasis and cycloplegia for diagnostic procedures.
DOSAGE AND ADMINISTRATION
For refraction, instill one or two drops of 1% solution in the eye(s), repeated in five minutes. If patient is not seen within 20 to 30 minutes, an additional drop may be instilled to prolong mydriatic effect. For examination of fundus, instill one or two drops of 0.5% solution 15 or 20 minutes prior to examination. Individuals with heavily pigmented irides may require higher strength or more doses. Mydriasis will reverse spontaneously with time, typically in 4 to 8 hours. However, in some cases, complete recovery may take up to 24 hours.
CONTRAINDICATIONS
Contraindicated in persons showing hypersensitivity to any component of this preparation.
ADVERSE REACTIONS
Ocular: Transient stinging, blurred vision, photophobia and superficial punctate keratitis have been reported with the use of tropicamide. Increased intraocular pressure has been reported following the use of mydriatics.
Non-Ocular: Dryness of the mouth, tachycardia, headache, allergic reactions, nausea, vomiting, pallor, central nervous system disturbances and muscle rigidity have been reported with the use of tropicamide. Psychotic reactions, behavioral disturbances, and vasomotor or cardiorespiratory collapse in children have been reported with the use of anticholinergic drugs.
Drug Interactions
Tropicamide may interfere with the antihypertensive action of carbachol, pilocarpine, or ophthalmic cholinesterase inhibitors.
DESCRIPTION
MYDRIACYL ® (tropicamide ophthalmic solution, USP) is an anticholinergic prepared as a sterile topical ophthalmic solution in two strengths. The active ingredient is represented by the chemical structure:
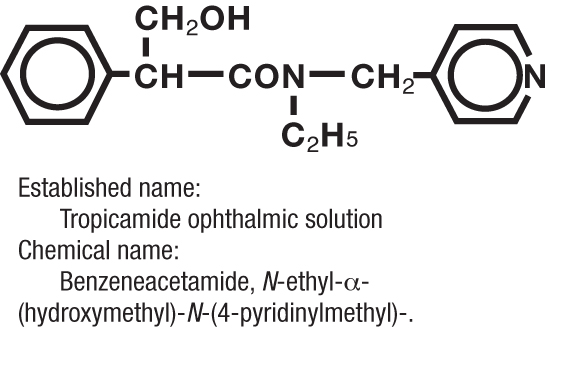
Each mL of MYDRIACYL ® (tropicamide ophthalmic solution, USP) contains: Active: tropicamide 0.5% or 1%. Preservative: benzalkonium chloride 0.01%. Inactives: sodium chloride, edetate disodium, hydrochloric acid and/or sodium hydroxide (to adjust pH), purified water; pH range 4.0-5.8.
CLINICAL PHARMACOLOGY
This anticholinergic preparation blocks the responses of the sphincter muscle of the iris and the ciliary muscle to cholinergic stimulation, dilating the pupil (mydriasis). The stronger preparation (1%) also paralyzes accommodation. This preparation acts in 15-30 minutes, and the duration of activity is approximately 3-8 hours. Complete recovery from mydriasis in some individuals may require 24 hours. The weaker strength may be useful in producing mydriasis with only slight cycloplegia. Heavily pigmented irides may require more doses than lightly pigmented irides.
HOW SUPPLIED
3 mL and 15 mL in plastic DROP-TAINER® dispensers.
0.5% 15 mL: NDC 0998-0354-15
1% 3 mL: NDC 0065-0355-03
15 mL: NDC 0998-0355-15
STORAGE: Store at 8°C to 27°C (46°F-80°F). Do not refrigerate or store at high temperatures. Keep container tightly closed.
Rx Only