Get your patient on Nilandron - Nilutamide tablet (Nilutamide)
Nilandron - Nilutamide tablet prescribing information
Interstitial Pneumonitis
Interstitial pneumonitis has been reported in 2% of patients in controlled clinical trials in patients exposed to nilutamide. A small study in Japanese subjects showed that 8 of 47 patients (17%) developed interstitial pneumonitis. Reports of interstitial changes including pulmonary fibrosis that led to hospitalization and death have been reported rarely post-marketing. Symptoms included exertional dyspnea, cough, chest pain, and fever. X-rays showed interstitial or alveolo-interstitial changes, and pulmonary function tests revealed a restrictive pattern with decreased DLco. Most cases occurred within the first 3 months of treatment with NILANDRON, and most reversed with discontinuation of therapy. A routine chest X-ray should be performed prior to initiating treatment with NILANDRON. Baseline pulmonary function tests may be considered. Patients should be instructed to report any new or worsening shortness of breath that they experience while on NILANDRON. If symptoms occur, NILANDRON should be immediately discontinued until it can be determined if the symptoms are drug related.
INDICATIONS AND USAGE
Metastatic Prostate Cancer
NILANDRON tablets are indicated for use in combination with surgical castration for the treatment of metastatic prostate cancer (Stage D 2 ).
For maximum benefit, NILANDRON treatment must begin on the same day as or on the day after surgical castration.
DOSAGE AND ADMINISTRATION
The recommended dosage is 300 mg once a day for 30 days, followed thereafter by 150 mg once a day. NILANDRON tablets can be taken with or without food.
CONTRAINDICATIONS
NILANDRON tablets are contraindicated:
- in patients with severe hepatic impairment (baseline hepatic enzymes should be evaluated prior to treatment)
- in patients with severe respiratory insufficiency
- in patients with hypersensitivity to nilutamide or any component of this preparation.
ADVERSE REACTIONS
Clinical Trial Experience
The following adverse experiences were reported during a multicenter clinical trial comparing NILANDRON + surgical castration versus placebo + surgical castration. The most frequently reported (greater than 5%) adverse experiences during treatment with NILANDRON tablets in combination with surgical castration are listed below. For comparison, adverse experiences seen with surgical castration and placebo are also listed.
| Adverse Experience | NILANDRON + surgical castration (N=225) % All | Placebo + surgical castration (N=232) % All |
| Cardiovascular System | ||
| Hypertension | 5.3 | 2.6 |
| Digestive System | ||
| Nausea | 9.8 | 6.0 |
| Constipation | 7.1 | 3.9 |
| Endocrine System | ||
| Hot flushes | 28.4 | 22.4 |
| Metabolic and Nutritional System | ||
| Increased AST | 8.0 | 3.9 |
| Increased ALT | 7.6 | 4.3 |
| Nervous System | ||
| Dizziness | 7.1 | 3.4 |
| Respiratory System | ||
| Dyspnea | 6.2 | 7.3 |
| Special Senses | ||
| Impaired adaptation to dark | 12.9 | 1.3 |
| Abnormal vision | 6.7 | 1.7 |
| Urogenital System | ||
| Urinary tract infection | 8.0 | 9.1 |
The overall incidence of adverse experiences was 86% (194/225) for the NILANDRON group and 81% (188/232) for the placebo group.
The following adverse experiences were reported during a multicenter clinical trial comparing NILANDRON + leuprolide versus placebo + leuprolide. The most frequently reported (greater than 5%) adverse experiences during treatment with NILANDRON tablets in combination with leuprolide are listed below. For comparison, adverse experiences seen with leuprolide and placebo are also listed.
| Adverse Experience | NILANDRON + leuprolide (N=209) % All | Placebo + leuprolide (N=202) % All |
| Body as a Whole | ||
| Pain | 26.8 | 27.7 |
| Headache | 13.9 | 10.4 |
| Asthenia | 19.1 | 20.8 |
| Back pain | 11.5 | 16.8 |
| Abdominal pain | 10.0 | 5.4 |
| Chest pain | 7.2 | 4.5 |
| Flu syndrome | 7.2 | 3.0 |
| Fever | 5.3 | 6.4 |
| Cardiovascular System | ||
| Hypertension | 9.1 | 9.9 |
| Digestive System | ||
| Nausea | 23.9 | 8.4 |
| Constipation | 19.6 | 16.8 |
| Anorexia | 11.0 | 6.4 |
| Dyspepsia | 6.7 | 4.5 |
| Vomiting | 5.7 | 4.0 |
| Endocrine System | ||
| Hot flushes | 66.5 | 59.4 |
| Impotence | 11.0 | 12.9 |
| Libido decreased | 11.0 | 4.5 |
| Hemic and Lymphatic System | ||
| Anemia | 7.2 | 6.4 |
| Metabolic and Nutritional System | ||
| Increased AST | 12.9 | 13.9 |
| Peripheral edema | 12.4 | 17.3 |
| Increased ALT | 9.1 | 8.9 |
| Musculoskeletal System | ||
| Bone Pain | 6.2 | 5.0 |
| Nervous System | ||
| Insomnia | 16.3 | 15.8 |
| Dizziness | 10.0 | 11.4 |
| Depression | 8.6 | 7.4 |
| Hypesthesia | 5.3 | 2.0 |
| Respiratory System | ||
| Dyspnea | 10.5 | 7.4 |
| Upper respiratory infection | 8.1 | 10.9 |
| Pneumonia | 5.3 | 3.5 |
| Skin and Appendages | ||
| Sweating | 6.2 | 3.0 |
| Body hair loss | 5.7 | 0.5 |
| Dry skin | 5.3 | 2.5 |
| Rash | 5.3 | 4.0 |
| Special Senses | ||
| Impaired adaptation to dark | 56.9 | 5.4 |
| Chromatopsia | 8.6 | 0.0 |
| Impaired adaptation to light | 7.7 | 1.0 |
| Abnormal vision | 6.2 | 4.5 |
| Urogenital System | ||
| Testicular atrophy | 16.3 | 12.4 |
| Gynecomastia | 10.5 | 11.9 |
| Urinary tract infection | 8.6 | 21.3 |
| Hematuria | 8.1 | 7.9 |
| Urinary tract disorder | 7.2 | 10.4 |
| Nocturia | 6.7 | 6.4 |
The overall incidence of adverse experiences is 99.5% (208/209) for the NILANDRON group and 98.5% (199/202) for the placebo group.
Some frequently occurring adverse experiences, for example hot flushes, impotence, and decreased libido, are known to be associated with low serum androgen levels and known to occur with medical or surgical castration alone. Notable was the higher incidence of visual disturbances (variously described as impaired adaptation to darkness, abnormal vision, and colored vision), which led to treatment discontinuation in 1% to 2% of patients.
Interstitial pneumonitis occurred in one (<1%) patient receiving NILANDRON in combination with surgical castration and in seven patients (3%) receiving NILANDRON in combination with leuprolide and one patient receiving placebo in combination with leuprolide. Overall, it has been reported in 2% of patients receiving NILANDRON. This included a report of interstitial pneumonitis in 8 of 47 patients (17%) in a small study performed in Japan.
In addition, the following adverse experiences were reported in 2 to 5% of patients treated with NILANDRON in combination with leuprolide or orchiectomy.
Body as a Whole: Malaise (2%).
Cardiovascular System: Angina (2%), heart failure (3%), syncope (2%).
Digestive System: Diarrhea (2%), gastrointestinal disorder (2%), gastrointestinal hemorrhage (2%), melena (2%).
Metabolic and Nutritional System: Alcohol intolerance (5%), edema (2%), weight loss (2%).
Musculoskeletal System: Arthritis (2%).
Nervous System: Dry mouth (2%), nervousness (2%), paresthesia (3%).
Respiratory System: Cough increased (2%), interstitial lung disease (2%), lung disorder (4%), rhinitis (2%).
Skin and Appendages: Pruritus (2%).
Special Senses: Cataract (2%), photophobia (2%).
Laboratory Values: Haptoglobin increased (2%), leukopenia (3%), alkaline phosphatase increased (3%), BUN increased (2%), creatinine increased (2%), hyperglycemia (4%).
To report SUSPECTED ADVERSE REACTIONS, contact Advanz Pharma (US) Corp. at 1-877-370-1142 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
In vitro, nilutamide has been shown to inhibit the activity of liver cytochrome P-450 isoenzymes and, therefore, may reduce the metabolism of compounds requiring these systems.
Consequently, drugs with a low therapeutic margin, such as vitamin K antagonists, phenytoin, and theophylline, could have a delayed elimination and increases in their serum half-life leading to a toxic level. The dosage of these drugs or others with a similar metabolism may need to be modified if they are administered concomitantly with nilutamide. For example, when vitamin K antagonists are administered concomitantly with nilutamide, prothrombin time should be carefully monitored and, if necessary, the dosage of vitamin K antagonists should be reduced.
DESCRIPTION
NILANDRON ® tablets contain nilutamide, a nonsteroidal, orally active antiandrogen having the chemical name 5,5-dimethyl-3-[4-nitro-3-(trifluoromethyl)phenyl]-2,4-imidazolidinedione with the following structural formula:
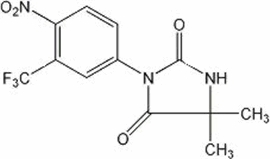
Nilutamide is a microcrystalline, white to practically white powder with a molecular weight of 317.25. Its molecular formula is C 12 H 10 F 3 N 3 O 4 .
It is freely soluble in ethyl acetate, acetone, chloroform, ethyl alcohol, dichloromethane, and methanol. It is slightly soluble in water [<0.1% W/V at 25°C (77°F)]. It melts between 153°C and 156°C (307.4°F and 312.8°F).
Each NILANDRON tablet contains 150 mg of nilutamide. Other ingredients in NILANDRON tablets are corn starch, lactose, povidone, docusate sodium, magnesium stearate, and talc.
CLINICAL PHARMACOLOGY
Mechanism of Action
Prostate cancer is known to be androgen sensitive and responds to androgen ablation. In animal studies, nilutamide has demonstrated antiandrogenic activity without other hormonal (estrogen, progesterone, mineralocorticoid, and glucocorticoid) effects. In vitro, nilutamide blocks the effects of testosterone at the androgen receptor level. In vivo, nilutamide interacts with the androgen receptor and prevents the normal androgenic response.
Pharmacokinetics
Absorption:
Analysis of blood, urine, and feces samples following a single oral 150-mg dose of [ 14 C]-nilutamide in patients with metastatic prostate cancer showed that the drug is rapidly and completely absorbed and that it yields high and persistent plasma concentrations.
Distribution:
After absorption of the drug, there is a detectable distribution phase. There is moderate binding of the drug to plasma proteins and low binding to erythrocytes. The binding is nonsaturable except in the case of alpha-1-glycoprotein, which makes a minor contribution to the total concentration of proteins in the plasma. The results of binding studies do not indicate any effects that would cause nonlinear pharmacokinetics.
Metabolism:
The results of a human metabolism study using 14 C-radiolabelled tablets show that nilutamide is extensively metabolized and less than 2% of the drug is excreted unchanged in urine after 5 days. Five metabolites have been isolated from human urine. Two metabolites display an asymmetric center, due to oxidation of a methyl group, resulting in the formation of D- and L-isomers. One of the metabolites was shown, in vitro, to possess 25 to 50% of the pharmacological activity of the parent drug, and the D-isomer of the active metabolite showed equal or greater potency compared to the L-isomer. However, the pharmacokinetics and the pharmacodynamics of the metabolites have not been fully investigated.
Elimination:
The majority (62%) of orally administered [ 14 C]-nilutamide is eliminated in the urine during the first 120 hours after a single 150-mg dose. Fecal elimination is negligible, ranging from 1.4% to 7% of the dose after 4 to 5 days. Excretion of radioactivity in urine likely continues beyond 5 days. The mean elimination half-life of nilutamide determined in studies in which subjects received a single dose of 100–300 mg ranged from 38.0 to 59.1 hours with most values between 41 and 49 hours. The elimination of at least one metabolite is generally longer than that of unchanged nilutamide (59–126 hours). During multiple dosing of 150 mg nilutamide (given as 3 × 50 mg) twice a day, steady state was reached within 2 to 4 weeks for most patients, and mean steady state AUC 0–12 was 110% higher than the AUC 0–∞ obtained from the first 150 mg dose. These data and in vitro metabolism data suggest that, upon multiple dosing, metabolic enzyme inhibition may occur for this drug.
Clinical Studies
Nilutamide through its antiandrogenic activity can complement surgical castration, which suppresses only testicular androgens. The effects of the combined therapy were studied in patients with previously untreated metastatic prostate cancer.
In a double-blind, randomized, multicenter study that enrolled 457 patients (225 treated with orchiectomy and NILANDRON, 232 treated with orchiectomy and placebo), the NILANDRON group showed a statistically significant benefit in time to progression and time to death. The results are summarized below.
| NILANDRON | PLACEBO | |
| Median Survival (months) | 27.3 | 23.6 |
| Progression-Free Survival (months) | 21.1 | 14.9 |
| Complete or Partial Regression | 41% | 24% |
| Improvement in Bone Pain | 54% | 37% |
Clinical Studies
Nilutamide through its antiandrogenic activity can complement surgical castration, which suppresses only testicular androgens. The effects of the combined therapy were studied in patients with previously untreated metastatic prostate cancer.
In a double-blind, randomized, multicenter study that enrolled 457 patients (225 treated with orchiectomy and NILANDRON, 232 treated with orchiectomy and placebo), the NILANDRON group showed a statistically significant benefit in time to progression and time to death. The results are summarized below.
| NILANDRON | PLACEBO | |
| Median Survival (months) | 27.3 | 23.6 |
| Progression-Free Survival (months) | 21.1 | 14.9 |
| Complete or Partial Regression | 41% | 24% |
| Improvement in Bone Pain | 54% | 37% |
HOW SUPPLIED
NILANDRON 150 mg tablets are supplied in boxes of 30 tablets. Each box contains 3 child-resistant, PVC, aluminum foil-backed blisters of 10 tablets (NDC 59212-111-14). Each white, biconvex, cylindrical (10 mm in diameter) tablet has a triangular logo on one side and an internal reference number (168D) on the other.
Store at 25°C (77°F); excursions permitted between 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Protect from light. Keep out of reach of children. Rev. 11/23 Manufactured for: Advanz Pharma (US) Corp. Bannockburn, IL 60015
NILANDRON ® is a registered trademark of Mercury Pharma Group Limited. Distributed by Advanz Pharma (US) Corp. under license.
Mechanism of Action
Prostate cancer is known to be androgen sensitive and responds to androgen ablation. In animal studies, nilutamide has demonstrated antiandrogenic activity without other hormonal (estrogen, progesterone, mineralocorticoid, and glucocorticoid) effects. In vitro, nilutamide blocks the effects of testosterone at the androgen receptor level. In vivo, nilutamide interacts with the androgen receptor and prevents the normal androgenic response.
Nilandron - Nilutamide tablet PubMed™ news
- Journal Article • 2026 JanExploring the interplay of electron density distribution and electrostatic potential in the interaction of nilutamide and flutamide with androgen receptors using quantum crystallography.
- Journal Article • 2025 SepNanoengineered scheelite-structured SrWO(4) decorated on MXene sheets: A novel platform for electrochemical sensing of the anti-cancer drug nilutamide with DFT insights.
- Journal Article • 2025 AugMetabolic activation of flunitrazepam via nitroreduction mediated by aldo-keto reductase 1C3.
- Journal Article • 2025 Jul3D Nanoflowers of Binary Metal-Selenide for Improved Electrochemical Sensing and High-Energy-Density Energy Storage.
- Journal Article • 2025 JulPolymorphic Control in Pharmaceutical Gel-Mediated Crystallization: Exploiting Solvent-Gelator Synergy in FmocFF Organogels.