Get your patient on Norgesic Forte - Orphenadrine Citrate, Aspirin And Caffeine tablet, Multilayer (Orphenadrine Citrate, Aspirin And Caffeine)
Norgesic Forte - Orphenadrine Citrate, Aspirin And Caffeine tablet, Multilayer prescribing information
INDICATIONS AND USAGE
Norgesic Forte 50 mg/770mg/60 mg Tablets are indicated in:
- Symptomatic relief of mild to moderate pain of acute musculoskeletal disorders.
- The orphenadrine component is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute painful musculoskeletal conditions.
The mode of action of orphenadrine has not been clearly identified, but may be related to its analgesic properties. Norgesic Forte Tablets do not directly relax tense skeletal muscles in man.
DOSAGE AND ADMINISTRATION
Norgesic Forte Tablets: Adults 1/2 to 1 tablet 3 to 4 times daily.
CONTRAINDICATIONS
Because of the mild anti-cholinergic effect of orphenadrine, Norgesic Forte Tablets should not be used in patients with glaucoma, pyloric or duodenal obstruction, achalasia, prostatic hypertrophy, or obstructions at the bladder neck. Norgesic Forte Tablets are also contraindicated in patients with myasthenia gravis and in patients known to be sensitive to aspirin or caffeine.
The drug is contraindicated in patients who have demonstrated a previous hypersensitivity to the drug.
ADVERSE REACTIONS
Side effects of Norgesic Forte Tablets are those seen with aspirin and caffeine or those usually associated with mild anticholinergic agents. These may include tachycardia, palpitation, urinary hesitancy or retention, dry mouth, blurred vision, dilation of the pupil, increased intraocular tension, weakness, nausea, vomiting, headache, dizziness, constipation, drowsiness, and rarely, urticaria and other dermatoses. Infrequently, an elderly patient may experience some degree of confusion. Mild central excitation and occasional hallucinations may be observed. These mild side effects can usually be eliminated by reduction in dosage. One case of aplastic anemia associated with the use of Orphenadrine Citrate, Aspirin, and Caffeine Tablets has been reported. No causal relationship has been established. Rare G.I. hemorrhage due to aspirin content may be associated with the administration of Norgesic Forte Tablets. Some patients may experience transient episodes of lightheadedness, dizziness or syncope.
DESCRIPTION
Each Norgesic Forte tablet, for oral administration, contains Orphenadrine Citrate 50 mg, Aspirin 770 mg, Caffeine 60 mg.
In addition, each tablet contains the following inactive ingredients, anhydrous lactous, colloidal silicon dioxide, D&C yellow #10, FD&C blue #1, zinc stearate, providone, pregetanized starch, and stearic acid.
Orphenadrine citrate, (2-dimethylaminoethyl 2-methybenzhydryl ether citrate). It is as a white, practically odorless, crystalline powder, having a bitter taste. It is sparingly soluble in water, slightly soluble in alcohol. It has the following structural formula:
.jpg)
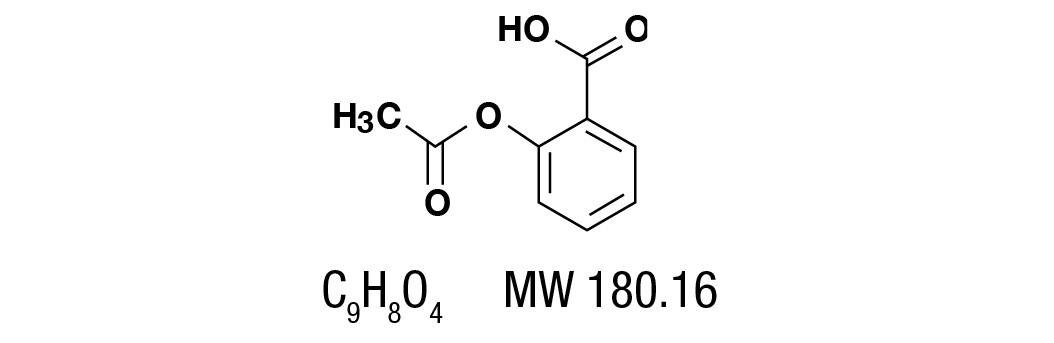
Aspirin, salicylic acid acetate, is a non-opiate analgesic, anti-inflammatory and antipyretic agent. It occurs as a white, crystalline tabular or needle like powder and is odorless or has a faint odor. It is sparingly soluble in water, freely soluble in alcohol and chloroform. It has the following structural formula:
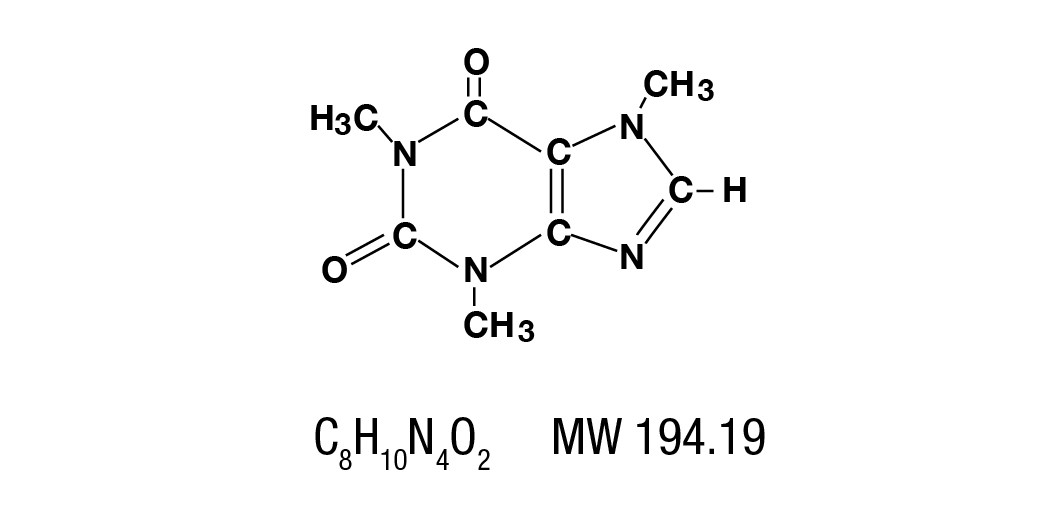
Caffeine is a central nervous system stimulant which occurs as a white powder or white glistening needles, usually matted together. It is sparingly soluble in alcohol, and freely soluble in chloroform. The chemical name for caffeine is 1,3,7-Trimethylxanthine. It has the following structural formula:
CLINICAL PHARMACOLOGY
Orphenadrine citrate is a centrally acting (brain stem) compound which in animals selectively blocks facilitatory functions of the reticular formulation. Orphenadrine does not produce myoneural block, nor does it affect crossed extensor reflexes. Orphenadrine prevents nicotine-induced convulsions but not those produced by strychnine.
Chronic administration of Orphenadrine Citrate, Aspirin, and Caffeine to dogs and rats has revealed no drug-related toxicity. No blood or urine changes were observed, nor were there any macroscopic or microscopic pathological changes detected. Extensive experience with combinations containing aspirin and caffeine has established them as safe agents. The addition of orphenadrine citrate does not alter the toxicity of aspirin and caffeine.
The mode of therapeutic action of orphenadrine has not been clearly identified, but may be related to its analgesic properties. Orphenadrine citrate also possesses anticholinergic actions.
HOW SUPPLIED
Norgesic Forte Tablets (Orphenadrine Citrate 50mg, Aspirin 770mg, and Caffeine 60mg) Two-layered, white/green capsule shaped, bisected tablets debossed "GA" and "473" with bisect on the white side and plain on the green side are available in bottles of 60 tablets (NDC 50991-999-60).
Store below 30°C (86°F)
Rx Only
Storage
Store at 20°-25°C (68°-77° F) (see USP Controlled Room Temperature).
Protect from moisture.