Get your patient on Octreoscan - Indium In -111 Pentetreotide (Indium In -111 Pentetreotide)
Octreoscan - Indium In -111 Pentetreotide prescribing information
INDICATIONS AND USAGE
Octreoscan, after radiolabeling, is an agent for the scintigraphic localization of primary and metastatic neuroendocrine tumors bearing somatostatin receptors.
DOSAGE AND ADMINISTRATION
Before administration, a patient should be well hydrated. After administration, the patient must be encouraged to drink fluids liberally. Elimination of extra fluid intake will help reduce the radiation dose by flushing out unbound, labelled pentetreotide by glomerular filtration. It is also recommended that a mild laxative (e.g., bisacodyl or lactulose) be given to the patient starting the evening before the radioactive drug is administered, and continuing for 48 hours. Ample fluid uptake is necessary during this period as a support both to renal elimination and the bowel-cleansing process. In a patient with an insulinoma, bowel-cleansing should be undertaken only after consultation with an endocrinologist.
The recommended intravenous dose for planar imaging is 111 MBq (3 mCi) of Indium In 111 Pentetreotide Injection prepared from an Octreoscan kit. The recommended intravenous dose for SPECT imaging is 222 MBq (6 mCi) of Indium In 111 Pentetreotide Injection.
The dose should be confirmed by a suitably calibrated radioactivity ionization chamber immediately before administration.
As with all intravenously administered products, Octreoscan should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Preparations containing particulate matter or discoloration should not be administered. They should be disposed of in a safe manner, in compliance with applicable regulations.
Aseptic techniques and effective shielding should be employed in withdrawing doses for administration to patients. Waterproof gloves should be worn during the administration procedure.
Do not administer Octreoscan in TPN solutions or through the same intravenous line.
Radiation Dosimetry
The estimated radiation doses Values listed include a correction for a maximum of 0.1% indium In-114m radiocontaminant at calibration. to the average adult (70 kg) from intravenous administration of 111 MBq (3 mCi) and 222 MBq (6 mCi) are presented in Table 4. These estimates were calculated by Oak Ridge Associated Universities using the data published by Krenning, et al. E.P. Krenning, W.H. Bakker, P.P.M. Kooij, W.A.P. Breeman, H.Y. Oei, M. de Jong, J.C. Reubi, T.J. Visser, C. Bruns, D.J. Kwekkeboom, A.E.M. Reijs, P.M. van Hagen, J.W. Koper, and S.W.J. Lamberts, “Somatostatin Receptor Scintigraphy with Indium-111-DTPA-D-Phe-1-Octreotide in Man: Metabolism, Dosimetry and Comparison with Iodine-123-Try-3-Octreotide,” The Journal of Nuclear Medicine, Vol. 33, No. 5, May 1992, pp. 652-658.
| PLANAR | SPECT | |||
| Organ | mGy/111 MBq | rads/3 mCi | mGy/222 MBq | rads/6 mCi |
| Kidneys | 54.16 | 5.42 | 108.32 | 10.83 |
| Liver | 12.15 | 1.22 | 24.31 | 2.43 |
| Spleen | 73.86 | 7.39 | 147.73 | 14.77 |
| Uterus | 6.34 | 0.63 | 12.67 | 1.27 |
| Ovaries | 4.89 | 0.49 | 9.79 | 0.98 |
| Testes | 2.90 | 0.29 | 5.80 | 0.58 |
| Red Marrow | 3.46 | 0.35 | 6.91 | 0.69 |
| Urinary Bladder Wall | 30.24 | 3.02 | 60.48 | 6.05 |
| GI Tract | ||||
| Stomach Wall | 5.67 | 0.57 | 11.34 | 1.13 |
| Small Intestine | 4.78 | 0.48 | 9.56 | 0.96 |
| Upper Large Intestine | 5.80 | 0.58 | 11.59 | 1.16 |
| Lower Large Intestine | 7.73 | 0.77 | 15.46 | 1.55 |
| Adrenals | 7.55 | 0.76 | 15.11 | 1.51 |
| Thyroid | 7.43 | 0.74 | 14.86 | 1.49 |
| mSv/111 MBq | rem/3 mCi | mSv/222 MBq | rem/6 mCi | |
| Effective Dose Estimated according to ICRP Publication 53. Equivalent | 13.03 | 1.30 | 26.06 | 2.61 |
CONTRAINDICATIONS
None known.
ADVERSE REACTIONS
The following adverse effects were observed in clinical trials at a frequency of less than 1% of 538 patients: dizziness, fever, flush, headache, hypotension, changes in liver enzymes, joint pain, nausea, sweating, and weakness. These adverse effects were transient. Also in clinical trials, there was one reported case of bradycardia and one case of decreased hematocrit and hemoglobin.
Pentetreotide is derived from octreotide which is used as a therapeutic agent to control symptoms from certain tumors. The usual dose for Indium In 111 Pentetreotide Injection is approximately 5 to 20 times less than for octreotide and is subtherapeutic. The following adverse reactions have been associated with octreotide in 3% to 10% of patients: nausea, injection site pain, diarrhea, abdominal pain/discomfort, loose stools, and vomiting. Hypertension and hyper- and hypoglycemia have also been reported with the use of octreotide.
The following adverse reactions have been identified during postapproval use of Octreoscan. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity reactions, predominantly rash, pruritus, less frequently angioedema or features of anaphylaxis.
DESCRIPTION
Octreoscan ™ is a kit for the preparation of Indium In 111 Pentetreotide Injection, a radioactive diagnostic agent. It is a kit consisting of two components:
1) A 10-mL Octreoscan Reaction Vial which contains a lyophilized mixture of: (i) 10 mcg pentetreotide [N-(diethylenetriamine-N,N,N′,N″-tetraacetic acid-N″-acetyl)-D-phenylalanyl-L-hemicystyl-L-phenylalanyl-D-tryptophyl-L-lysyl-L-threonyl-L-hemicystyl-L-threoninol cyclic (2→7) disulfide], (also known as octreotide DTPA), (ii) 2 mg gentisic acid [2, 5-dihydroxybenzoic acid], (iii) 4.9 mg trisodium citrate, anhydrous, (iv) 0.37 mg citric acid, anhydrous, and (v) 10 mg inositol.
Pentetreotide has the following structural formula:
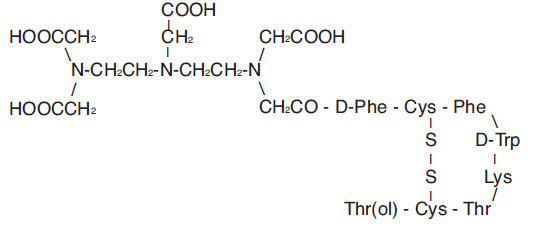
Prior to lyophilization, sodium hydroxide or hydrochloric acid may have been added for pH adjustment. The vial contents are sterile and nonpyrogenic. No bacteriostatic preservative is present.
2) A 10-mL vial of Indium In 111 Chloride Solution, which contains: 1.1 mL or 111 MBq/mL (3 mCi/mL) indium In 111 chloride in 0.02N HCl at time of calibration. The vial also contains ferric chloride at a concentration of 3.5 mcg/mL (ferric ion, 1.2 mcg/mL). The vial contents are sterile and nonpyrogenic. No bacteriostatic preservative is present.
Indium In 111 Pentetreotide Injection is prepared by combining the two kit components ( see INSTRUCTIONS FOR THE PREPARATION OF INDIUM In 111 PENTETREOTIDE INJECTION ). Indium In-111 reacts with the diethylenetriaminetetraacetic acid portion of the pentetreotide molecule to form indium In 111 pentetreotide. The pH of the resultant Indium In 111 Pentetreotide Injection is between 3.8 and 4.3. No bacteriostatic preservative is present.
The Indium In 111 Pentetreotide Injection is suitable for intravenous administration as is, or it may be diluted to a maximum volume of 3 mL with 0.9% Sodium Chloride Injection, USP, immediately before intravenous administration. In either case, the radiolabeling yield of Indium In 111 Pentetreotide Injection should be determined before administration to the patient. A method recommended for determining the radiolabeling yield is presented at the end of this package insert (see RECOMMENDED METHOD FOR DETERMINATION OF RADIOLABELING YIELD OF INDIUM In 111 PENTETREOTIDE INJECTION ).
Physical Characteristics
Indium In-111 decays by electron capture to cadmium-111 (stable) and has a physical half-life of 2.805 days (67.32 hours) ( see Table 2 ). From Radiopharmaceutical Internal Dosimetry Information Center, Oak Ridge Associated Universities, Oak Ridge, TN 37831-0117, February 1985. The principal photons that are useful for detection and imaging are listed in Table 1.
| Radiation | Mean Percent Per Disintegration | Energy (keV) |
| Gamma-2 | 90.2 | 171.3 |
| Gamma-3 | 94.0 | 245.4 |
The specific gamma ray constant for In-111 is 3.21 R/hr-mCi at 1 cm. The first half-value thickness of lead (Pb) for In-111 is 0.023 cm. Selected coefficients of attenuation are listed in Table 2 as a function of lead shield thickness. For example, the use of 0.834 cm of lead will attenuate the external radiation by a factor of about 1000.
| Shield Thickness (Pb) cm | Coefficient of Attenuation |
| 0.023 | 0.5 |
| 0.203 | 0.1 |
| 0.513 | 0.01 |
| 0.834 | 0.001 |
| 1.12 | 0.0001 |
Table 3 lists fractions remaining at selected time intervals before and after calibration. This information may be used to correct for physical decay of the radionuclide.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
| -72 | 2.100 | 0 Calibration time | 1.000 |
| -60 | 1.854 | 3 | 0.970 |
| -48 | 1.639 | 6 | 0.940 |
| -36 | 1.448 | 12 | 0.885 |
| -24 | 1.280 | 24 | 0.781 |
| -12 | 1.131 | 36 | 0.690 |
| -6 | 1.064 | 48 | 0.610 |
CLINICAL PHARMACOLOGY
General
Pentetreotide is a DTPA conjugate of octreotide, which is a long-acting analog of the human hormone, somatostatin. Indium In 111 pentetreotide binds to somatostatin receptors on cell surfaces throughout the body. Within an hour of injection, most of the dose of indium In 111 pentetreotide distributes from plasma to extravascular body tissues and concentrates in tumors containing a high density of somatostatin receptors. After background clearance, visualization of somatostatin receptor-rich tissue is achieved. In addition to somatostatin receptor-rich tumors, the normal pituitary gland, thyroid gland, liver, spleen and urinary bladder also are visualized in most patients, as is the bowel, to a lesser extent. Excretion is almost exclusively via the kidneys.
Pharmacokinetics
Radioactivity leaves the plasma rapidly; one third of the radioactive injected dose remains in the blood pool at 10 minutes after administration. Plasma levels continue to decline so that by 20 hours post-injection, about 1% of the radioactive dose is found in the blood pool. The biological half-life of indium In 111 pentetreotide is 6 hours.
Half of the injected dose is recoverable in urine within 6 hours after injection, 85% is recovered in the first 24 hours, and over 90% is recovered in urine by two days.
Hepatobiliary excretion represents a minor route of elimination, and less than 2% of the injected dose is recovered in feces within three days after injection.
Metabolism
For several hours after administration, plasma radioactivity is predominantly in parent form. Ten percent of the radioactivity excreted is nonpeptide-bound.
Pharmacodynamics
Indium In 111 pentetreotide binds to cell surface receptors for somatostatin. In nonclinical pharmacologic studies, the hormonal effect of Octreoscan in vitro is one-tenth that of octreotide. Since diagnostic imaging doses of indium In 111 pentetreotide are lower than the therapeutic doses of octreotide, indium In 111 pentetreotide is not expected to exert clinically significant somatostatin effects.
Indium In 111 pentetreotide is cleared from the body primarily by renal excretion. Indium In 111 pentetreotide elimination has not been studied in anephric patients or in those with poorly functioning kidneys. It is not known whether indium In 111 pentetreotide can be removed by dialysis. Dosage adjustments in patients with decreased renal function have not been studied.
Clinical Trials
Octreoscan was studied in nine unblinded clinical studies in a total of 365 patients. Of these patients, 174 were male and 191 were female. Their mean age was 54.0 years (range 1.8 to 86 years). One patient was under the age of 2 and 2 patients were between the ages of 2 and 12; 223 patients (61.1%) were between 18 and 60 years; and 136 patients (37.3%) were older than 60 years. A racial distribution is not available.
Eligible patients had a demonstrated or high clinical suspicion of a neuroendocrine tumor. The most common tumors were carcinoids (132 of 309 evaluable patients). Scintigraphic results were compared to results of conventional localization procedures (CT, ultrasound, MRI, angiography, surgery and/or biopsy). The mean dose of radioactivity administered was 173.4 MBq (4.7 mCi).
Octreoscan results were consistent with the final diagnosis (success) in 267 of 309 evaluable patients (86.4%). Compared with carcinoids and gastrinomas, lower success rates were noted for localization of insulinomas, neuroblastomas, pituitary adenomas and medullary thyroid carcinomas. Octreoscan success was observed in 27 of 32 patients (84.4%) with clinically nonfunctioning neuroendocrine tumors (i.e., no symptom of a clinical syndrome mediated by abnormally elevated hormones).
Octreoscan localized previously unidentified tumors in 57/204 patients. In 55/195 patients, indium In 111 pentetreotide uptake occurred in lesions not thought to have somatostatin receptors. In a small subgroup of 39 patients who had tissue confirmation, the sensitivity rate for Octreoscan scintigraphy was 85.7%; for CT/MRI the rate was 68%. The specificity rate for Octreoscan scintigraphy was 50%, the rate for CT/MRI was 12%. Larger studies are needed to confirm these comparisons. Overall, including all tumor types with or without the presence of somatostatin receptors, there were 3/508 false positives and 104/508 false negatives.
Of the 309 patients, 87 had received octreotide for therapeutic purposes within 72 hours of Octreoscan administration. These patients had an overall 95% success rate. The effect of different dose levels of octreotide on success rates has not been evaluated.
Clinical Trials
Octreoscan was studied in nine unblinded clinical studies in a total of 365 patients. Of these patients, 174 were male and 191 were female. Their mean age was 54.0 years (range 1.8 to 86 years). One patient was under the age of 2 and 2 patients were between the ages of 2 and 12; 223 patients (61.1%) were between 18 and 60 years; and 136 patients (37.3%) were older than 60 years. A racial distribution is not available.
Eligible patients had a demonstrated or high clinical suspicion of a neuroendocrine tumor. The most common tumors were carcinoids (132 of 309 evaluable patients). Scintigraphic results were compared to results of conventional localization procedures (CT, ultrasound, MRI, angiography, surgery and/or biopsy). The mean dose of radioactivity administered was 173.4 MBq (4.7 mCi).
Octreoscan results were consistent with the final diagnosis (success) in 267 of 309 evaluable patients (86.4%). Compared with carcinoids and gastrinomas, lower success rates were noted for localization of insulinomas, neuroblastomas, pituitary adenomas and medullary thyroid carcinomas. Octreoscan success was observed in 27 of 32 patients (84.4%) with clinically nonfunctioning neuroendocrine tumors (i.e., no symptom of a clinical syndrome mediated by abnormally elevated hormones).
Octreoscan localized previously unidentified tumors in 57/204 patients. In 55/195 patients, indium In 111 pentetreotide uptake occurred in lesions not thought to have somatostatin receptors. In a small subgroup of 39 patients who had tissue confirmation, the sensitivity rate for Octreoscan scintigraphy was 85.7%; for CT/MRI the rate was 68%. The specificity rate for Octreoscan scintigraphy was 50%, the rate for CT/MRI was 12%. Larger studies are needed to confirm these comparisons. Overall, including all tumor types with or without the presence of somatostatin receptors, there were 3/508 false positives and 104/508 false negatives.
Of the 309 patients, 87 had received octreotide for therapeutic purposes within 72 hours of Octreoscan administration. These patients had an overall 95% success rate. The effect of different dose levels of octreotide on success rates has not been evaluated.
HOW SUPPLIED
The Octreoscan kit (NDC 69945-050-40) is supplied with the following components:
- A 10-mL Octreoscan Reaction Vial which contains a lyophilized mixture of:
(i) 10 mcg pentetreotide [N-(diethylenetriamine-N,N,N',N”-tetraacetic acid-N”-acetyl)-D-phenylalanyl-L-hemicystyl-L-phenylalanyl-D-tryptophyl-L-lysyl-L-threonyl-L-hemicystyl-L-threoninol cyclic (2→7) disulfide], (also known as octreotide DTPA), (ii) 2 mg gentisic acid [2, 5-dihydroxybenzoic acid], (iii) 4.9 mg trisodium citrate, anhydrous, (iv) 0.37 mg citric acid, anhydrous, and (v) 10 mg inositol. Before lyophilization, sodium hydroxide or hydrochloric acid may have been added for pH adjustment. The vial contents are sterile and nonpyrogenic. No bacteriostatic preservative is present.
- A 10-mL vial of Indium In 111 Chloride Solution, which contains 1.1 mL or 111 MBq/mL (3 mCi/mL) Indium In 111 chloride in 0.02 N HCl at time of calibration. The vial also contains ferric chloride at a concentration of 3.5 mcg/mL (ferric ion, 1.2 mcg/mL). The vial contents are sterile and nonpyrogenic. No bacteriostatic preservative is present.
In addition, the kit also contains the following items: (1) a 25 G x 5/8” needle (B-D, Monoject) used to transfer Indium In 111 Chloride Solution to the Octreoscan Reaction Vial, (2) pressure sensitive label, and (3) a package insert.
Storage
The Octreoscan kit should be stored refrigerated at 2° to 8°C (36° to 46°F).
After reconstitution, store at controlled room temperature 20° to 25°C (68° to 77°F). Indium In 111 Pentetreotide Injection must be used within 6 hours of preparation.