Get your patient on Ocuflox - Ofloxacin solution/ Drops (Ofloxacin)
Ocuflox - Ofloxacin solution/ Drops prescribing information
INDICATIONS AND USAGE
OCUFLOX ® ophthalmic solution is indicated for the treatment of infections caused by susceptible strains of the following bacteria in the conditions listed below:
| CONJUNCTIVITIS: | |
| Gram-positive bacteria: | Gram-negative bacteria: |
| Staphylococcus aureus | Enterobacter cloacae |
| Staphylococcus epidermidis | Haemophilus influenzae |
| Streptococcus pneumoniae | Proteus mirabilis |
| Pseudomonas aeruginosa | |
| CORNEAL ULCERS: | |
| Gram-positive bacteria: | Gram-negative bacteria: |
| Staphylococcus aureus | Pseudomonas aeruginosa |
| Staphylococcus epidermidis | Serratia marcescens• |
| Streptococcus pneumoniae | Anaerobic species: |
| Propionibacterium acnes |
•Efficacy for this organism was studied in fewer than 10 infections
DOSAGE AND ADMINISTRATION
The recommended dosage regimen for the treatment of bacterial conjunctivitis is:
| Days 1 and 2 | Instill one to two drops every two to four hours in the affected eye(s). |
| Days 3 through 7 | Instill one to two drops four times daily. |
| The recommended dosage regimen for the treatment of bacterial corneal ulcer is: | |
| Days 1 and 2 | Instill one to two drops into the affected eye every 30 minutes, while awake. Awaken at approximately four and six hours after retiring and instill one to two drops. |
| Days 3 through 7 to 9 | Instill one to two drops hourly, while awake. |
| Days 7 to 9 through | |
| treatment completion | Instill one to two drops, four times daily. |
CONTRAINDICATIONS
OCUFLOX ® solution is contraindicated in patients with a history of hypersensitivity to ofloxacin, to other quinolones, or to any of the components in this medication ( s ee Warnings ) .
ADVERSE REACTIONS
Ophthalmic Use
The most frequently reported drug-related adverse reaction was transient ocular burning or discomfort. Other reported reactions include stinging, redness, itching, chemical conjunctivitis/keratitis, ocular/periocular/facial edema, foreign body sensation, photophobia, blurred vision, tearing, dryness, and eye pain. Rare reports of dizziness and nausea have been received.
Refer to Warnings for additional adverse reactions.
Drug Interactions
Specific drug interaction studies have not been conducted with OCUFLOX ® ophthalmic solution. However, the systemic administration of some quinolones has been shown to elevate plasma concentrations of theophylline, interfere with the metabolism of caffeine, and enhance the effects of the oral anticoagulant warfarin and its derivatives, and has been associated with transient elevations in serum creatinine in patients receiving cyclosporine concomitantly.
DESCRIPTION
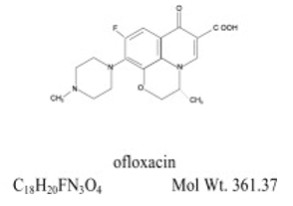
OCUFLOX ® (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical ophthalmic use.
Chemical Name:
(±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7 H -pyrido[1,2,3- de ]-1,4-benzoxazine-6-carboxylic acid.
Contains: Active: ofloxacin 0.3% (3 mg/mL). Preservative: benzalkonium chloride (0.005%).
Inactives : sodium chloride and purified water. May also contain hydrochloric acid and/or sodium hydroxide to adjust pH.
OCUFLOX ® solution is unbuffered and formulated with a pH of 6.4 (range - 6.0 to 6.8). It has an osmolality of 300 mOsm/kg. Ofloxacin is a fluorinated 4-quinolone which differs from other fluorinated 4-quinolones in that there is a six member (pyridobenzoxazine) ring from positions 1 to 8 of the basic ring structure.
CLINICAL PHARMACOLOGY
Pharmacokinetics
Serum, urine and tear concentrations of ofloxacin were measured in 30 healthy women at various time points during a ten-day course of treatment with OCUFLOX ® solution. The mean serum ofloxacin concentration ranged from 0.4 ng/mL to 1.9 ng/mL. Maximum ofloxacin concentration increased from 1.1 ng/mL on day one to 1.9 ng/mL on day 11 after QID dosing for 10 1/2 days. Maximum serum ofloxacin concentrations after ten days of topical ophthalmic dosing were more than 1000 times lower than those reported after standard oral doses of ofloxacin.
Tear ofloxacin concentrations ranged from 5.7 to 31 mcg/g during the 40 minute period following the last dose on day 11. Mean tear concentration measured four hours after topical ophthalmic dosing was 9.2 mcg/g.
Corneal tissue concentrations of 4.4 mcg/mL were observed four hours after beginning topical ocular application of two drops of OCUFLOX ® ophthalmic solution every 30 minutes. Ofloxacin was excreted in the urine primarily unmodified.
Microbiology
Ofloxacin has in vitro activity against a broad range of gram-positive and gram-negative aerobic and anaerobic bacteria. Ofloxacin is bactericidal at concentrations equal to or slightly greater than inhibitory concentrations. Ofloxacin is thought to exert a bactericidal effect on susceptible bacterial cells by inhibiting DNA gyrase, an essential bacterial enzyme which is a critical catalyst in the duplication, transcription, and repair of bacterial DNA.
Cross-resistance has been observed between ofloxacin and other fluoroquinolones. There is generally no cross-resistance between ofloxacin and other classes of antibacterial agents such as beta-lactams or aminoglycosides.
Ofloxacin has been shown to be active against most strains of the following organisms both in vitro and clinically, in conjunctival and/or corneal ulcer infections ( see Indications and Usage ) .
| AEROBES, GRAM-POSITIVE: | AEROBES, GRAM-NEGATIVE: |
| Staphylococcus aureus | Enterobacter cloacae |
| Staphylococcus epidermidis | Haemophilus influenzae |
| Streptococcus pneumoniae | Proteus mirabilis |
| ANAEROBIC SPECIES: | Pseudomonas aeruginosa |
| Propionibacterium acnes | Serratia marcescens• |
•Efficacy for this organism was studied in fewer than 10 infections
The safety and effectiveness of OCUFLOX ® ophthalmic solution in treating ophthalmologic infections due to the following organisms have not been established in adequate and well-controlled clinical trials. OCUFLOX ® ophthalmic solution has been shown to be active in vitro against most strains of these organisms but the clinical significance in ophthalmologic infections is unknown.
| AEROBES, GRAM-POSITIVE: | |
| Enterococcus faecalis | Staphylococcus hominus |
| Listeria monocytogenes | Staphylococcus simulans |
| Staphylococcus capitis | Streptococcus pyogenes |
| AEROBES, GRAM-NEGATIVE: | |
| Acinetobacter calcoaceticus var. anitratus | Klebsiella pneumoniae |
| Acinetobacter calcoaceticus var. lwoffii | Moraxella ( Branhamella ) catarrhalis |
| Citrobacter diversus | Moraxella lacunata |
| Citrobacter freundii | Morganella morganii |
| Enterobacter aerogenes | Neisseria gonorrhoeae |
| Enterobacter agglomerans | Pseudomonas acidovorans |
| Escherichia coli | Pseudomonas fluorescens |
| Haemophilus parainfluenzae | Shigella sonnei |
| Klebsiella oxytoca | |
| OTHER: | |
| Chlamydia trachomatis |
Clinical Studies
Conjunctivitis
In a randomized, double-masked, multicenter clinical trial, OCUFLOX ® ophthalmic solution was superior to its vehicle after 2 days of treatment in patients with conjunctivitis and positive conjunctival cultures. Clinical outcomes for the trial demonstrated a clinical improvement rate of 86% (54/63) for the ofloxacin treated group versus 72% (48/67) for the placebo treated group after 2 days of therapy. Microbiological outcomes for the same clinical trial demonstrated an eradication rate for causative pathogens of 65% (41/63) for the ofloxacin treated group versus 25% (17/67) for the vehicle treated group after 2 days of therapy. Please note that microbiologic eradication does not always correlate with clinical outcome in anti-infective trials.
Corneal Ulcers
In a randomized, double-masked, multi-center clinical trial of 140 subjects with positive cultures, OCUFLOX ® ophthalmic solution treated subjects had an overall clinical success rate (complete re-epithelialization and no progression of the infiltrate for two consecutive visits) of 82% (61/74) compared to 80% (53/66) for the fortified antibiotic group, consisting of 1.5% tobramycin and 10% cefazolin solutions. The median time to clinical success was 11 days for the ofloxacin treated group and 10 days for the fortified treatment group.
Clinical Studies
Conjunctivitis
In a randomized, double-masked, multicenter clinical trial, OCUFLOX ® ophthalmic solution was superior to its vehicle after 2 days of treatment in patients with conjunctivitis and positive conjunctival cultures. Clinical outcomes for the trial demonstrated a clinical improvement rate of 86% (54/63) for the ofloxacin treated group versus 72% (48/67) for the placebo treated group after 2 days of therapy. Microbiological outcomes for the same clinical trial demonstrated an eradication rate for causative pathogens of 65% (41/63) for the ofloxacin treated group versus 25% (17/67) for the vehicle treated group after 2 days of therapy. Please note that microbiologic eradication does not always correlate with clinical outcome in anti-infective trials.
Corneal Ulcers
In a randomized, double-masked, multi-center clinical trial of 140 subjects with positive cultures, OCUFLOX ® ophthalmic solution treated subjects had an overall clinical success rate (complete re-epithelialization and no progression of the infiltrate for two consecutive visits) of 82% (61/74) compared to 80% (53/66) for the fortified antibiotic group, consisting of 1.5% tobramycin and 10% cefazolin solutions. The median time to clinical success was 11 days for the ofloxacin treated group and 10 days for the fortified treatment group.
HOW SUPPLIED
OCUFLOX ® (ofloxacin ophthalmic solution) 0.3% is supplied sterile in opaque white LDPE plastic bottles and white dropper tips with tan high impact polystyrene (HIPS) caps as follows:
5mL in 10mL bottle - NDC 11980-779-05
Note: Store at 15°-25°C (59°-77°F)
Rx only
Revised : 0 6 /2024
Distributed by: AbbVie, Inc. North Chicago, IL 60064
© 2024 AbbVie. All rights reserved. Ocuflox and its design are trademarks of Allergan, Inc., an AbbVie company.
20085528