Get your patient on Oxybutynin Chloride - Oxybutynin Chloride tablet (Oxybutynin Chloride)
Oxybutynin Chloride - Oxybutynin Chloride tablet prescribing information
INDICATIONS & USAGE
Oxybutynin chloride tablets, USP are indicated for the relief of symptoms of bladder instability associated with voiding in patients with uninhibited neurogenic or reflex neurogenic bladder (i.e., urgency, frequency, urinary leakage, urge incontinence, dysuria).
DOSAGE & ADMINISTRATION
Adults
The usual dose is one 5-mg tablet two to three times a day. The maximum recommended dose is one 5-mg tablet four times a day. A lower starting dose of 2.5 mg two or three times a day is recommended for the frail elderly.
Pediatric patients over 5 years of age
The usual dose is one 5-mg tablet two times a day. The maximum recommended dose is one 5-mg tablet three times a day.
CONTRAINDICATIONS
Oxybutynin chloride tablets are contraindicated in patients with urinary retention, gastric retention and other severe decreased gastrointestinal motility conditions, uncontrolled narrow-angle glaucoma and in patients who are at risk for these conditions.
Oxybutynin chloride tablets are also contraindicated in patients who have demonstrated hypersensitivity to the drug substance or other components of the product.
ADVERSE REACTIONS
The safety and efficacy of oxybutynin chloride was evaluated in a total of 199 patients in three clinical trials. These participants were treated with oxybutynin chloride 5 to 20 mg/day for up to 6 weeks. Table 3 shows the incidence of adverse events judged by investigators to be at least possibly related to treatment and reported by at least 5% of patients.
Table 3 Incidence (%) of Adverse Events Reported by ≥ 5% of Patients Using Oxybutynin Chloride (5 to 20 mg/day)
| Body System | Adverse Event | Oxybutynin Chloride (5 to 20 mg/day) (n=199) |
| Infections and Infestations | Urinary tract infection | 6.5% |
| Psychiatric Disorders | Insomnia | 5.5% |
| Nervousness | 6.5% | |
| Nervous System Disorders | Dizziness | 16.6% |
| Somnolence | 14.0% | |
| Headache | 7.5% | |
| Eye Disorders | Blurred vision | 9.6% |
| Gastrointestinal Disorders | Dry mouth | 71.4% |
| Constipation | 15.1% | |
| Nausea | 11.6% | |
| Dyspepsia | 6.0% | |
| Renal and Urinary Disorders | Urinary Hesitation | 8.5% |
| Urinary Retention | 6.0% |
The most common adverse events reported by patients receiving oxybutynin chloride 5 to 20 mg/day were the expected side effects of anticholinergic agents. The incidence of dry mouth was dose-related.
In addition, the following adverse events were reported by 1 to <5% of patients using oxybutynin chloride (5 to 20 mg/day) in all studies.
Infections and Infestations : nasopharyngitis, upper respiratory tract infection, bronchitis, cystitis, fungal infection; Metabolism and Nutrition Disorders : fluid retention; Psychiatric Disorders : confusional state; Nervous System Disorders : dysgeusia, sinus headache; Eye Disorders : kerato conjunctivitis sicca, eye irritation; Cardiac Disorders : palpitations, sinus arrhythmia; Vascular Disorders : flushing; Respiratory, thoracic and Mediastinal Disorders : nasal dryness, cough, pharyngolaryngeal pain, dry throat, sinus congestion, hoarseness, asthma, nasal congestion; Gastrointestinal Disorders : diarrhea, abdominal pain, loose stools, flatulence, vomiting, abdominal pain upper, dysphagia, aptyalism, eructation, tongue coated; Skin and Subcutaneous Tissue Disorders : dry skin, pruritis; Musculoskeletal and Connective Tissue Disorders : back pain, arthralgia, pain in extremity, flank pain; Renal and Urinary Disorders : dysuria, pollakiuria; General Disorders and Administration Site Conditions : fatigue, edema peripheral, asthenia, pain, thirst, edema; Investigations : blood pressure increased, blood glucose increased, blood pressure decreased; Injury, Poisoning, and Procedural Complications : fall.
Postmarketing Surveillance
Because postmarketing adverse events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following additional adverse events have been reported from worldwide postmarketing experience with oral oxybutynin chloride: Psychiatric Disorders : psychotic disorder, agitation, hallucinations, memory impairment; Nervous System Disorders : convulsions; Eye Disorders : cycloplegia, mydriasis, glaucoma; Cardiac Disorders : tachycardia, QT interval prolongation, chest discomfort; Gastrointestinal Disorders : decreased gastrointestinal motility, frequent bowel movements; Skin and Subcutaneous Tissue Disorders : rash, decreased sweating; Renal and Urinary Disorders : impotence; Reproductive System and Breast Disorders : Suppression of lactation; General Disorders and Administration Site Conditions: hypersensitivity reactions, including angioedema with airway obstruction, urticaria, and face edema; rare anaphylactic reactions requiring hospitalization for emergency treatment; Metabolism and Nutrition Disorders : anorexia; Respiratory, Thoracic and Mediastinal Disorders : dysphonia.
Drug Interactions
The concomitant use of oxybutynin with other anticholinergic drugs or with other agents which produce dry mouth, constipation, somnolence (drowsiness), and/or other anticholinergic-like effects may increase the frequency and/or severity of such effects.
Anticholinergic agents may potentially alter the absorption of some concomitantly administered drugs due to anticholinergic effects on gastrointestinal motility. This may be of concern for drugs with a narrow therapeutic index. Anticholinergic agents may also antagonize the effects of prokinetic agents, such as metoclopramide.
Mean oxybutynin chloride plasma concentrations were approximately 3 to 4 fold higher when oxybutynin was administered with ketoconazole, a potent CYP3A4 inhibitor.
Other inhibitors of the cytochrome P450 3A4 enzyme system, such as antimycotic agents (e.g., itraconazole and miconazole) or macrolide antibiotics (e.g., erythromycin and clarithromycin), may alter oxybutynin mean pharmacokinetic parameters (i.e., C max and AUC). The clinical relevance of such potential interactions is not known. Caution should be used when such drugs are co-administered.
DESCRIPTION
Each scored oxybutynin chloride tablet, USP contains 5 mg of oxybutynin chloride. Chemically, oxybutynin chloride is d, l (racemic) 4-diethylamino-2-butynyl phenylcyclohexylglycolate hydrochloride. The molecular formula of oxybutynin chloride is C 22 H 31 NO 3 •HCl. The structural formula appears below:
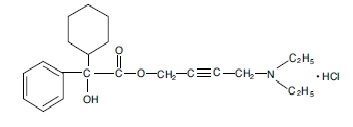
Oxybutynin chloride, USP is a white crystalline solid with a molecular weight of 393.95. It is readily soluble in water and acids, but relatively insoluble in alkalis.
Oxybutynin chloride Tablets, USP also contains anhydrous lactose, croscarmellose sodium, magnesium stearate, microcrystalline cellulose.
Oxybutynin chloride tablets, USP are for oral administration.
Therapeutic Category: Antispasmodic, anticholinergic. Meets USP Dissolution Test 2.
CLINICAL PHARMACOLOGY
Oxybutynin chloride exerts a direct antispasmodic effect on smooth muscle and inhibits the muscarinic action of acetylcholine on smooth muscle. Oxybutynin chloride exhibits only one fifth of the anticholinergic activity of atropine on the rabbit detrusor muscle, but four to ten times the antispasmodic activity. No blocking effects occur at skeletal neuromuscular junctions or autonomic ganglia (antinicotinic effects).
Oxybutynin chloride relaxes bladder smooth muscle. In patients with conditions characterized by involuntary bladder contractions, cystometric studies have demonstrated that oxybutynin chloride increases bladder (vesical) capacity, diminishes the frequency of uninhibited contractions of the detrusor muscle, and delays the initial desire to void. Oxybutynin chloride thus decreases urgency and the frequency of both incontinent episodes and voluntary urination.
Antimuscarinic activity resides predominately in the R-isomer. A metabolite, desethyloxybutynin, has pharmacological activity similar to that of oxybutynin in in vitro studies.
Pharmacokinetics
Absorption
Following oral administration of oxybutynin chloride tablets, oxybutynin is rapidly absorbed achieving C max within an hour, following which plasma concentration decreases with an effective half-life of approximately 2 to 3 hours. The absolute bioavailability of oxybutynin is reported to be about 6% (range 1.6 to 10.9%) for the tablets. Wide interindividual variation in pharmacokinetic parameters is evident following oral administration of oxybutynin.
The mean pharmacokinetic parameters for R- and S-oxybutynin are summarized in Table 1 . The plasma concentration-time profiles for R- and S-oxybutynin are similar in shape; Figure 1 shows the profile for R-oxybutynin.
Table 1 Mean (SD) R-and S-Oxybutynin Pharmacokinetic Parameters Following Three Doses of Oxybutynin Chloride 5 mg Administered every 8 Hours (n=23)
| Parameters (units) | R-Oxybutynin | S-Oxybutynin |
| C max (ng/mL) | 3.6 (2.2) | 7.8 (4.1) |
| T max (h) | 0.89 (0.34) | 0.65 (0.32) |
| AUC t (ng•h/mL) | 22.6 (11.3) | 35.0 (17.3) |
| AUC inf (ng•h/mL) | 24.3 (12.3) | 37.3 (18.7) |
Figure 1 . Mean R-Oxybutynin Plasma Concentrations Following Three Doses of Oxybutynin Chloride 5 mg Administered Every 8 Hours for 1 Day in 23 Healthy Adult Volunteers. 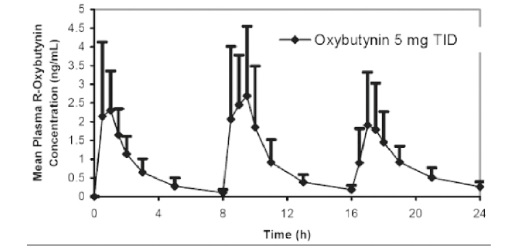
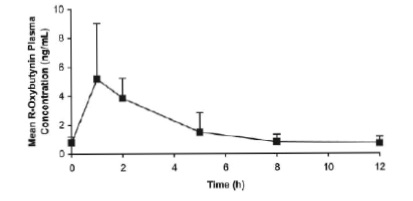
Oxybutynin chloride steady-state pharmacokinetics were also studied in 11 pediatric patients with detrusor overactivity associated with a neurological condition (e.g., spina bifida). These pediatric patients were on oxybutynin chloride tablets with total daily dose ranging from 7.5 mg to 15 mg (0.22 to 0.53 mg/kg). Overall, most patients (86.9%) were taking a total daily oxybutynin chloride dose between 10 mg and 15 mg. Sparse sampling technique was used to obtain serum samples. When all available data are normalized to an equivalent of 5 mg twice daily oxybutynin chloride, the mean pharmacokinetic parameters derived for R- and S-oxybutynin and R- and S-desethyloxybutynin are summarized in Table 2 . The plasma-time concentration profiles for R- and S-oxybutynin are similar in shape; Figure 2 shows the profile for R-oxybutynin when all available data are normalized to an equivalent of 5 mg twice daily.
Table 2 Mean ± SD R- and S-Oxybutynin and R- and S-Desethyloxybutynin Pharmacokinetic Parameters in Children Aged 5 to 15 Following Administration of 7.5 mg to 15 mg Total Daily Dose of Oxybutynin Chloride Tablets (N=11)
All Available Data Normalized to an Equivalent of Oxybutynin Chloride Tablets 5 mg BID or TID at Steady State
| R-Oxybutynin | S-Oxybutynin | R-Desethyloxybutynin | S-Desethyloxybutynin | |
| C max • (ng/mL) | 6.1 ± 3.2 | 10.1 ± 7.5 | 55.4 ± 17.9 | 28.2 ± 10.0 |
| T max (hr) | 1.0 | 1.0 | 2.0 | 2.0 |
| AUC † (ng•hr/mL) | 19.8 ± 7.4 | 28.4 ± 12.7 | 238.8 ± 77.6 | 119.5 ± 50.7 |
•Reflects C max for pooled data
† AUC 0-end of dosing interval
Figure 2 . Mean steady-state (±SD) R-oxybutynin plasma concentrations following administration of total daily Oxybutynin Chloride Tablet dose of 7.5 mg to 15 mg (0.22 mg/kg to 0.53 mg/kg) in children 5 to 15 years of age. – Plot represents all available data normalized to the equivalent of oxybutynin chloride 5 mg BID or TID at steady state
Food Effects
Data in the literature suggests that oxybutynin solution co-administered with food resulted in a slight delay in absorption and an increase in its bioavailability by 25% (n=18). 1
Distribution
Oxybutynin is widely distributed in body tissues following systemic absorption. The volume of distribution is 193 L after intravenous administration of 5 mg oxybutynin chloride. Both enantiomers of oxybutynin are highly bound (>99%) to plasma proteins. Both enantiomers of desethyloxybutynin are also highly bound (>97%) to plasma proteins. The major binding protein is alpha-1 acid glycoprotein.
Metabolism
Oxybutynin is metabolized primarily by the cytochrome P450 enzyme systems, particularly CYP3A4 found mostly in the liver and gut wall. Its metabolic products include phenylcyclohexylglycolic acid, which is pharmacologically inactive, and desethyloxybutynin, which is pharmacologically active.
Excretion
Oxybutynin is extensively metabolized by the liver, with less than 0.1% of the administered dose excreted unchanged in the urine. Also, less than 0.1% of the administered dose is excreted as the metabolite desethyloxybutynin
HOW SUPPLIED
Oxybutynin Chloride Tablets, USP 5 mg are white, round, biconvex tablets debossed with “n005” on one side and bisect on the other side. They are supplied as follows:
NDC 70954- 005 -10 Bottles of 100 Tablets
NDC 70954- 005 -20 Bottles of 500 Tablets
NDC 70954- 005- 30 Bottles of 1000 Tablets
NDC 70954- 005- 50 Unit dose blister packages of 100 (10 cards of 10 tablets each)
Pharmacist: Dispense in tight, light-resistant container as defined in the USP.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].