Phytonadione - Phytonadione injection, Emulsion prescribing information
WARNING — INTRAVENOUS AND INTRAMUSCULAR USE
Severe reactions, including fatalities, have occurred during and immediately after INTRAVENOUS injection of phytonadione, even when precautions have been taken to dilute the phytonadione and to avoid rapid infusion. Severe reactions, including fatalities, have also been reported following INTRAMUSCULAR administration. Typically these severe reactions have resembled hypersensitivity or anaphylaxis, including shock and cardiac and/or respiratory arrest. Some patients have exhibited these severe reactions on receiving phytonadione for the first time. Therefore the INTRAVENOUS and INTRAMUSCULAR routes should be restricted to those situations where the subcutaneous route is not feasible and the serious risk involved is considered justified.
INDICATIONS AND USAGE
Phytonadione Injectable Emulsion is indicated in the following coagulation disorders which are due to faulty formation of factors II, VII, IX and X when caused by vitamin K deficiency or interference with vitamin K activity.
Phytonadione Injectable Emulsion is indicated in: • anticoagulant-induced prothrombin deficiency caused by coumarin or indanedione derivatives; • prophylaxis and therapy of hemorrhagic disease of the newborn; • hypoprothrombinemia due to antibacterial therapy; • hypoprothrombinemia secondary to factors limiting absorption or synthesis of vitamin K, e.g., obstructive jaundice, biliary fistula, sprue, ulcerative colitis, celiac disease, intestinal resection, cystic fibrosis of the pancreas, and regional enteritis; • other drug-induced hypoprothrombinemia where it is definitely shown that the result is due to interference with vitamin K metabolism, e.g., salicylates.
DOSAGE AND ADMINISTRATION
Whenever possible, Phytonadione Injectable Emulsion, USP should be given by the subcutaneous route (See Box Warning). When intravenous administration is considered unavoidable, the drug should be injected very slowly, not exceeding 1 mg per minute.
Protect from light at all times.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Directions for Dilution
Phytonadione injectable emulsion, USP may be diluted with 0.9% Sodium Chloride Injection, 5% Dextrose Injection, or 5% Dextrose and Sodium Chloride Injection. Benzyl alcohol as a preservative has been associated with toxicity in newborns. Therefore, all of the above diluents should be preservative-free (See WARNINGS). Other diluents should not be used . When dilutions are indicated, administration should be started immediately after mixture with the diluent, and unused portions of the dilution should be discarded, as well as unused contents of the ampul.
Prophylaxis of Hemorrhagic Disease of the Newborn
The American Academy of Pediatrics recommends that Phytonadione be given to the newborn. A single intramuscular dose of phytonadione injectable emulsion, USP 0.5 to 1 mg within one hour of birth is recommended.
Treatment of Hemorrhagic Disease of the Newborn Empiric administration of Phytonadione should not replace proper laboratory evaluation of the coagulation mechanism. A prompt response (shortening of the prothrombin time in 2 to 4 hours) following administration of Phytonadione is usually diagnostic of hemorrhagic disease of the newborn, and failure to respond indicates another diagnosis or coagulation disorder.
Phytonadione injectable emulsion, USP 1 mg should be given either subcutaneously or intramuscularly. Higher doses may be necessary if the mother has been receiving oral anticoagulants.
Whole blood or component therapy may be indicated if bleeding is excessive. This therapy, however, does not correct the underlying disorder and phytonadione injectable emulsion, USP should be given concurrently.
Anticoagulant-Induced Prothrombin Deficiency in Adults To correct excessively prolonged prothrombin time caused by oral anticoagulant therapy—2.5 to 10 mg or up to 25 mg initially is recommended. In rare instances 50 mg may be required. Frequency and amount of subsequent doses should be determined by prothrombin time response or clinical condition (See WARNINGS). If in 6 to 8 hours after parenteral administration the prothrombin time has not been shortened satisfactorily, the dose should be repeated.
Phytonadione Injectable Emulsion, USP Summary of Dosage Guidelines (See circular text for details)
Newborns | Dosage |
Hemorrhagic Disease of the Newborn Prophylaxis | 0.5 to 1 mg IM within 1 hour of birth |
Treatment | 1 mg SC or IM (Higher doses may be necessary if the mother has been receiving oral anticoagulants) |
Adults | Initial Dosage |
Anticoagulant-Induced Prothrombin Deficiency (caused by coumarin or indanedione derivatives) | 2.5 mg to 10 mg or up to 25 mg (rarely 50 mg) |
Hypoprothrombinemia Due to other causes (Antibiotics; Salicylates or other drugs; Factors limiting absorption or synthesis) | 2.5 mg to 25 mg or more (rarely up to 50 mg) |
In the event of shock or excessive blood loss, the use of whole blood or component therapy is indicated.
Hypoprothrombinemia Due to Other Causes in Adults
A dosage of 2.5 to 25 mg or more (rarely up to 50 mg) is recommended, the amount and route of administration depending upon the severity of the condition and response obtained. If possible, discontinuation or reduction of the dosage of drugs interfering with coagulation mechanisms (such as salicylates; antibiotics) is suggested as an alternative to administering concurrent phytonadione injectable emulsion, USP. The severity of the coagulation disorder should determine whether the immediate administration of phytonadione injectable emulsion, USP is required in addition to discontinuation or reduction of interfering drugs.
CONTRAINDICATION
Hypersensitivity to any component of this medication.
ADVERSE REACTIONS
Deaths have occurred after intravenous and intramuscular administration. (See Box Warning.)
Transient "flushing sensations" and "peculiar" sensations of taste have been observed, as well as rare instances of dizziness, rapid and weak pulse, profuse sweating, brief hypotension, dyspnea, and cyanosis.
Pain, swelling, and tenderness at the injection site may occur.
The possibility of allergic sensitivity including an anaphylactoid reaction, should be kept in mind.
Infrequently, usually after repeated injection, erythematous, indurated, pruritic plaques have occurred; rarely, these have progressed to scleroderma-like lesions that have persisted for long periods. In other cases, these lesions have resembled erythema perstans.
Hyperbilirubinemia has been observed in the newborn following administration of phytonadione. This has occurred rarely and primarily with doses above those recommended (See PRECAUTIONS, Pediatric Use ).
DESCRIPTION
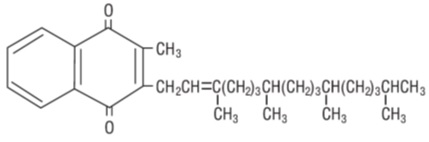
Phytonadione is a vitamin, which is a clear, yellow to amber, viscous, odorless or nearly odorless liquid. It is practically insoluble in water, sparingly soluble in ethanol and miscible with fatty oils. It has a molecular weight of 450.70. Phytonadione is 2-Methyl-3’,7’,11’,15’-tetramethyl-hexadec-2’-en-1-yl-naphthalene-1,4 dione. Its empirical formula is C 31 H 46 O 2 and its structural formula is:
Phytonadione Injectable Emulsion, USP is a yellow, sterile, nonpyrogenic aqueous dispersion available for injection by the intravenous, intramuscular and subcutaneous routes. Each milliliter contains phytonadione, USP 10 mg, polyoxyl 35 castor oil 70 mg, dextrose monohydrate 37.5 mg in water for injection; benzyl alcohol 9 mg added as preservative. May contain hydrochloric acid for pH adjustment. pH is 6.3 (5.0 to 7.0). Phytonadione is oxygen sensitive.
CLINICAL PHARMACOLOGY
Phytonadione Injectable Emulsion aqueous dispersion of Phytonadione for parenteral injection, possesses the same type and degree of activity as does naturally-occurring vitamin K, which is necessary for the production via the liver of active prothrombin (factor II), proconvertin (factor VII), plasma thromboplastin component (factor IX), and Stuart factor (factor X). The prothrombin test is sensitive to the levels of three of these four factors−II, VII, and X. Vitamin K is an essential cofactor for a microsomal enzyme that catalyzes the post-translational carboxylation of multiple, specific, peptide-bound glutamic acid residues in inactive hepatic precursors of factors II, VII, IX, and X. The resulting gamma-carboxy-glutamic acid residues convert the precursors into active coagulation factors that are subsequently secreted by liver cells into the blood.
Phytonadione is readily absorbed following intramuscular administration. After absorption, phytonadione is initially concentrated in the liver, but the concentration declines rapidly. Very little vitamin K accumulates in tissues. Little is known about the metabolic fate of vitamin K. Almost no free unmetabolized vitamin K appears in bile or urine.
In normal animals and humans, phytonadione is virtually devoid of pharmacodynamic activity. However, in animals and humans deficient in vitamin K, the pharmacological action of vitamin K is related to its normal physiological function, that is, to promote the hepatic biosynthesis of vitamin K dependent clotting factors.
The action of the aqueous dispersion, when administered intravenously, is generally detectable within an hour or two and hemorrhage is usually controlled within 3 to 6 hours. A normal prothrombin level may often be obtained in 12 to 14 hours. In the prophylaxis and treatment of hemorrhagic disease of the newborn, phytonadione has demonstrated a greater margin of safety than that of the water-soluble vitamin K analogues.
HOW SUPPLIED
Phytonadione Injectable Emulsion, USP is supplied as follows:
Unit of Sale | Concentration |
NDC 68462-758-25 25 ampuls (Bundle of 5 clamcells, each clamcell containing 5 single-dose ampuls) | 10 mg/mL |
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Protect from light. Keep ampuls in tray until time of use.
Distributed by:

Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
August 2025