Prochlorperazine Maleate - Prochlorperazine Maleate tablet prescribing information
WARNING
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5% compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Prochlorperazine maleate is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS ).
INDICATIONS AND USAGE
For control of severe nausea and vomiting.
For the treatment of schizophrenia.
Prochlorperazine is effective for the short-term treatment of generalized non-psychotic anxiety. However, prochlorperazine is not the first drug to be used in therapy for most patients with non-psychotic anxiety, because certain risks associated with its use are not shared by common alternative treatments (e.g., benzodiazepines).
When used in the treatment of non-psychotic anxiety, prochlorperazine should not be administered at doses of more than 20 mg per day or for longer than 12 weeks, because the use of prochlorperazine at higher doses or for longer intervals may cause persistent tardive dyskinesia that may prove irreversible (see WARNINGS ).
The effectiveness of prochlorperazine as treatment for non-psychotic anxiety was established in 4-week clinical studies of outpatients with generalized anxiety disorder. This evidence does not predict that prochlorperazine will be useful in patients with other non-psychotic conditions in which anxiety, or signs that mimic anxiety, are found (e.g., physical illness, organic mental conditions, agitated depression, character pathologies, etc.).
Prochlorperazine has not been shown effective in the management of behavioral complications in patients with mental retardation.
DOSAGE AND ADMINISTRATION
DOSAGE AND ADMINISTRATION–ADULTS
(For children’s dosage and administration, see below.) Dosage should be increased more gradually in debilitated or emaciated patients.
Elderly Patients: In general, dosages in the lower range are sufficient for most elderly patients. Since they appear to be more susceptible to hypotension and neuromuscular reactions, such patients should be observed closely. Dosage should be tailored to the individual, response carefully monitored and dosage adjusted accordingly. Dosage should be increased more gradually in elderly patients.
1. To Control Severe Nausea and Vomiting: Adjust dosage to the response of the individual. Begin with the lowest recommended dosage.
Oral Dosage–Tablets: Usually one 5 mg or 10 mg tablet 3 or 4 times daily. Daily dosages above 40 mg should be used only in resistant cases.
2. In Adult Psychiatric Disorders: Adjust dosage to the response of the individual and according to the severity of the condition. Begin with the lowest recommended dose. Although response ordinarily is seen within a day or 2, longer treatment is usually required before maximal improvement is seen.
Oral Dosage: Non-Psychotic Anxiety--Usual dosage is 5 mg 3 or 4 times daily. Do not administer in doses of more than 20 mg per day or for longer than 12 weeks.
Psychotic Disorders including Schizophrenia -- In relatively mild conditions, as seen in private psychiatric practice or in outpatient clinics, dosage is 5 or 10 mg 3 or 4 times daily.
In moderate to severe conditions, for hospitalized or adequately supervised patients, usual starting dosage is 10 mg 3 or 4 times daily. Increase dosage gradually until symptoms are controlled or side effects become bothersome. When dosage is increased by small increments every 2 or 3 days, side effects either do not occur or are easily controlled. Some patients respond satisfactorily on 50 to 75 mg daily.
In more severe disturbances, optimum dosage is usually 100 to 150 mg daily.
DOSAGE AND ADMINISTRATION–CHILDREN
Do not use in pediatric surgery.
Children seem more prone to develop extrapyramidal reactions, even on moderate doses. Therefore, use lowest effective dosage. Tell parents not to exceed prescribed dosage, since the possibility for adverse reactions increases as dosage rises.
Occasionally the patient may react to the drug with signs of restlessness and excitement; if this occurs, do not administer additional doses. Take particular precaution in administering the drug to children with acute illnesses or dehydration (see under Dystonias).
1. Severe Nausea and Vomiting in Children: Prochlorperazine should not be used in pediatric patients under 20 pounds in weight or 2 years of age. It should not be used in conditions for which children’s dosages have not been established. Dosage and frequency of administration should be adjusted according to the severity of the symptoms and the response of the patient. The duration of activity following intramuscular administration may last up to 12 hours. Subsequent doses may be given by the same route if necessary.
Oral Dosage: More than 1 day’s therapy is seldom necessary.
| Weight | Usual Dosage | Not to Exceed |
|---|---|---|
under 20 lbs not recommended | ||
20 to 29 lbs | 2½ mg 1 or 2 times a day | 7.5 mg per day |
30 to 39 lbs | 2½ mg 2 or 3 times a day | 10 mg per day |
40 to 85 lbs | 2½ mg 3 times a day or 5 mg 2 times a day | 15 mg per day |
2. Children with schizophrenia:
Oral Dosage: For children 2 to 12 years, starting dosage is 2½ mg 2 or 3 times daily. Do not give more than 10 mg the first day. Then increase dosage according to patient’s response.
FOR AGES 2 to 5, total daily dosage usually does not exceed 20 mg.
FOR AGES 6 to 12, total daily dosage usually does not exceed 25 mg.
CONTRAINDICATIONS
Do not use in patients with known hypersensitivity to phenothiazines.
Do not use in comatose states or in the presence of large amounts of central nervous system depressants (alcohol, barbiturates, narcotics, etc.).
Do not use in pediatric surgery.
Do not use in pediatric patients under 2 years of age or under 20 lbs. Do not use in children for conditions for which dosage has not been established.
ADVERSE REACTIONS
Drowsiness, dizziness, amenorrhea, blurred vision, skin reactions and hypotension may occur. Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs (see WARNINGS ).
Cholestatic jaundice has occurred. If fever with grippe-like symptoms occurs, appropriate liver studies should be conducted. If tests indicate an abnormality, stop treatment. There have been a few observations of fatty changes in the livers of patients who have died while receiving the drug. No causal relationship has been established.
Leukopenia and agranulocytosis have occurred. Warn patients to report the sudden appearance of sore throat or other signs of infection. If white blood cell and differential counts indicate leukocyte depression, stop treatment and start antibiotic and other suitable therapy.
Neuromuscular (Extrapyramidal) Reactions
These symptoms are seen in a significant number of hospitalized mental patients. They may be characterized by motor restlessness, be of the dystonic type, or they may resemble parkinsonism.
Depending on the severity of symptoms, dosage should be reduced or discontinued. If therapy is reinstituted, it should be at a lower dosage. Should these symptoms occur in children or pregnant patients, the drug should be stopped and not reinstituted. In most cases barbiturates by suitable route of administration will suffice. (Or, injectable Benadryl ®|| may be useful.) In more severe cases, the administration of an anti-parkinsonism agent, except levodopa (see PDR ), usually produces rapid reversal of symptoms. Suitable supportive measures such as maintaining a clear airway and adequate hydration should be employed.
Dystonia
Class effect: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
These usually subside within a few hours, and almost always within 24 to 48 hours, after the drug has been discontinued.
Motor Restlessness: Symptoms may include agitation or jitteriness and sometimes insomnia. These symptoms often disappear spontaneously. At times these symptoms may be similar to the original neurotic or psychotic symptoms. Dosage should not be increased until these side effects have subsided.
If these symptoms become too troublesome, they can usually be controlled by a reduction of dosage or change of drug. Treatment with anti-parkinsonian agents, benzodiazepines or propranolol may be helpful.
Pseudo-Parkinsonism: Symptoms may include: mask-like facies; drooling; tremors; pillrolling motion; cog-wheel rigidity; and shuffling gait. Reassurance and sedation are important. In most cases these symptoms are readily controlled when an anti-parkinsonism agent is administered concomitantly. Anti-parkinsonism agents should be used only when required. Generally, therapy of a few weeks to 2 or 3 months will suffice. After this time patients should be evaluated to determine their need for continued treatment. (Note: Levodopa has not been found effective in pseudo-parkinsonism.) Occasionally it is necessary to lower the dosage of prochlorperazine or to discontinue the drug.
Tardive Dyskinesia: As with all antipsychotic agents, tardive dyskinesia may appear in some patients on long-term therapy or may appear after drug therapy has been discontinued. The syndrome can also develop, although much less frequently, after relatively brief treatment periods at low doses. This syndrome appears in all age groups. Although its prevalence appears to be highest among elderly patients, especially elderly women, it is impossible to rely upon prevalence estimates to predict at the inception of antipsychotic treatment which patients are likely to develop the syndrome. The symptoms are persistent and in some patients appear to be irreversible. The syndrome is characterized by rhythmical involuntary movements of the tongue, face, mouth or jaw (e.g., protrusion of tongue, puffing of cheeks, puckering of mouth, chewing movements). Sometimes these may be accompanied by involuntary movements of extremities. In rare instances, these involuntary movements of the extremities are the only manifestations of tardive dyskinesia. A variant of tardive dyskinesia, tardive dystonia, has also been described.
There is no known effective treatment for tardive dyskinesia; antiparkinsonism agents do not alleviate the symptoms of this syndrome. It is suggested that all antipsychotic agents be discontinued if these symptoms appear.
Should it be necessary to reinstitute treatment, or increase the dosage of the agent, or switch to a different antipsychotic agent, the syndrome may be masked.
It has been reported that fine vermicular movements of the tongue may be an early sign of the syndrome and if the medication is stopped at that time the syndrome may not develop.
Adverse Reactions Reported with Prochlorperazine or Other Phenothiazine Derivatives: Adverse reactions with different phenothiazines vary in type, frequency and mechanism of occurrence, i.e., some are dose-related, while others involve individual patient sensitivity. Some adverse reactions may be more likely to occur, or occur with greater intensity, in patients with special medical problems, e.g., patients with mitral insufficiency or pheochromocytoma have experienced severe hypotension following recommended doses of certain phenothiazines.
Not all of the following adverse reactions have been observed with every phenothiazine derivative, but they have been reported with 1 or more and should be borne in mind when drugs of this class are administered: extrapyramidal symptoms (opisthotonos, oculogyric crisis, hyperreflexia, dystonia, akathisia, dyskinesia, parkinsonism) some of which have lasted months and even years–particularly in elderly patients with previous brain damage; grand mal and petit mal convulsions, particularly in patients with EEG abnormalities or history of such disorders; altered cerebrospinal fluid proteins; cerebral edema; intensification and prolongation of the action of central nervous system depressants (opiates, analgesics, antihistamines, barbiturates, alcohol), atropine, heat, organophosphorus insecticides; autonomic reactions (dryness of mouth, nasal congestion, headache, nausea, constipation, obstipation, adynamic ileus, ejaculatory disorders/impotence, priapism, atonic colon, urinary retention, miosis and mydriasis); reactivation of psychotic processes, catatonic-like states; hypotension (sometimes fatal); cardiac arrest; blood dyscrasias (pancytopenia, thrombocytopenic purpura, leukopenia, agranulocytosis, eosinophilia, hemolytic anemia, aplastic anemia); liver damage (jaundice, biliary stasis); endocrine disturbances (hyperglycemia, hypoglycemia, glycosuria, lactation, galactorrhea, gynecomastia, menstrual irregularities, false-positive pregnancy tests); skin disorders (photosensitivity, itching, erythema, urticaria, eczema up to exfoliative dermatitis); other allergic reactions (asthma, laryngeal edema, angioneurotic edema, anaphylactoid reactions); peripheral edema; reversed epinephrine effect; hyperpyrexia; mild fever after large I.M. doses; increased appetite; increased weight; a systemic lupus erythematosus-like syndrome; pigmentary retinopathy; with prolonged administration of substantial doses, skin pigmentation, epithelial keratopathy, and lenticular and corneal deposits.
EKG changes–particularly nonspecific, usually reversible Q and T wave distortions–have been observed in some patients receiving phenothiazines.
Although phenothiazines cause neither psychic nor physical dependence, sudden discontinuance in long-term psychiatric patients may cause temporary symptoms, e.g., nausea and vomiting, dizziness, tremulousness.
Note: There have been occasional reports of sudden death in patients receiving phenothiazines. In some cases, the cause appeared to be cardiac arrest or asphyxia due to failure of the cough reflex.
DESCRIPTION
Prochlorperazine, USP is a phenothiazine derivative, present in prochlorperazine tablets as the maleate. Its chemical name is 2-chloro-10-[3-(4-methyl-1-piperazinyl)propyl]phenothiazine maleate (1:2), empirical formula is C 20 H 24 CIN 3 S•2C 4 H 4 O 4 , molecular weight is 606.09 and has the following structure.
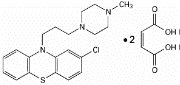
Prochlorperazine Maleate, USP
Prochlorperazine maleate, USP is white or pale yellow crystalline powder. It is practically insoluble in water and in alcohol; slightly soluble in warm chloroform.
Each tablet, for oral administration contains prochlorperazine maleate, USP equivalent to 5 mg or 10 mg of prochlorperazine. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, diethyl phthalate, ethyl cellulose, FD&C blue no. 2 aluminium lake, FD&C red no. 40 aluminium lake, hydroxypropyl methylcellulose 2910, lactose monohydrate, magnesium stearate, microcrystalline cellulose, poloxamer 188, pregelatinized starch, stearic acid 50, talc and titanium dioxide.
HOW SUPPLIED
Prochlorperazine Maleate Tablets, USP are available in the following strengths and package sizes:
5 mg: Reddish pink to pink, round, film-coated tablet debossed with “Y” on the top and “32” on the bottom of the score line on one side and plain on the other side.
- Bottles of 100 NDC 68462-889-01 Bottles of 1,000 NDC 68462-889-10
- 10 mg: Reddish pink to pink, round, film-coated tablet debossed with “Y” and “133” on the one side and scoreline on the other side.
- Bottles of 100 NDC 68462-947-01 Bottles of 1,000 NDC 68462-947-10
STORAGE
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from light. Dispense in a tight, light-resistant container.
•norepinephrine bitartrate, Abbott Laboratories. †phenylephrine hydrochloride, Abbott Laboratories. ‡phenytoin, Parke-Davis. §metrizamide, Sanofi Pharmaceuticals. ||diphenhydramine hydrochloride, Parke-Davis.
All trademarks are the property of their respective owners.
Distributed by:

Glenmark Pharmaceuticals Inc., USA
Elmwood Park, NJ 07407
Questions? 1 (888) 721-7115
August 2025