Promethazine Hydrochloride - Promethazine Hydrochloride syrup prescribing information
PROMETHAZINE SHOULD NOT BE USED IN PEDIATRIC PATIENTS LESS THAN 2 YEARS OF AGE BECAUSE OF THE POTENTIAL FOR FATAL RESPIRATORY DEPRESSION.
POSTMARKETING CASES OF RESPIRATORY DEPRESSION, INCLUDING FATALITIES, HAVE BEEN REPORTED WITH USE OF PROMETHAZINE IN PEDIATRIC PATIENTS LESS THAN 2 YEARS OF AGE. A WIDE RANGE OF WEIGHT-BASED DOSES OF PROMETHAZINE HAVE RESULTED IN RESPIRATORY DEPRESSION IN THESE PATIENTS.
CAUTION SHOULD BE EXERCISED WHEN ADMINISTERING PROMETHAZINE TO PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER. IT IS RECOMMENDED THAT THE LOWEST EFFECTIVE DOSE OF PROMETHAZINE BE USED IN PEDIATRIC PATIENTS 2 YEARS OF AGE AND OLDER AND CONCOMITANT ADMINISTRATION OF OTHER DRUGS WITH RESPIRATORY DEPRESSANT EFFECTS BE AVOIDED.
INDICATIONS AND USAGE
Promethazine is useful for:
Perennial and seasonal allergic rhinitis. Vasomotor rhinitis.
Allergic conjunctivitis due to inhalant allergens and foods.
Mild, uncomplicated allergic skin manifestations of urticaria and angioedema.
Amelioration of allergic reactions to blood or plasma.
Dermographism.
Anaphylactic reactions, as adjunctive therapy to epinephrine and other standard measures, after the acute manifestations have been controlled.
Preoperative, postoperative, or obstetric sedation.
Prevention and control of nausea and vomiting associated with certain types of anesthesia and surgery.
Therapy adjunctive to meperidine or other analgesics for control of post-operative pain.
Sedation in both children and adults, as well as relief of apprehension and production of light sleep from which the patient can be easily aroused.
Active and prophylactic treatment of motion sickness.
Antiemetic therapy in postoperative patients.
DOSAGE AND ADMINISTRATION
Promethazine Syrup is contraindicated for children under 2 years of age (see WARNINGS-Black Box Warning and Use in Pediatric Patients ).
Allergy
The average oral dose is 25 mg taken before retiring; however, 12.5 mg may be taken before meals and on retiring, if necessary. Single 25-mg doses at bedtime or 6.25 to 12.5 mg taken three times daily will usually suffice. After initiation of treatment in children or adults, dosage should be adjusted to the smallest amount adequate to relieve symptoms. The administration of promethazine HCl in 25-mg doses will control minor transfusion reactions of an allergic nature.
Motion Sickness
The average adult dose is 25 mg taken twice daily. The initial dose should be taken one-half to one hour before anticipated travel and be repeated 8 to 12 hours later, if necessary. On succeeding days of travel, it is recommended that 25 mg be given on arising and again before the evening meal. For children, Promethazine Syrup, 12.5 to 25 mg, twice daily, may be administered.
Nausea and Vomiting
Antiemetics should not be used in vomiting of unknown etiology in children and adolescents (see WARNINGS-Use in Pediatric Patients ).
The average effective dose of Promethazine for the active therapy of nausea and vomiting in children or adults is 25 mg. When oral medication cannot be tolerated, the dose should be given parenterally (cf. Promethazine Injection) or by rectal suppository. 12.5- to 25-mg doses may be repeated, as necessary, at 4- to 6-hour intervals.
For nausea and vomiting in children, the usual dose is 0.5 mg per pound of body weight, and the dose should be adjusted to the age and weight of the patient and the severity of the condition being treated.
For prophylaxis of nausea and vomiting, as during surgery and the postoperative period, the average dose is 25 mg repeated at 4- to 6-hour intervals, as necessary.
Sedation
This product relieves apprehension and induces a quiet sleep from which the patient can be easily aroused. Administration of 12.5 to 25 mg Promethazine by the oral route will provide sedation in children. Adults usually require 25 to 50 mg for nighttime, presurgical, or obstetrical sedation.
Pre- and Postoperative Use
Promethazine in 12.5- to 25-mg doses for children and 50-mg doses for adults the night before surgery relieves apprehension and produces a quiet sleep.
For preoperative medication, children require doses of 0.5 mg per pound of body weight in combination with an appropriately reduced dose of narcotic or barbiturate and the appropriate dose of an atropine-like drug. Usual adult dosage is 50 mg Promethazine with an appropriately reduced dose of narcotic or barbiturate and the required amount of a belladonna alkaloid.
Postoperative sedation and adjunctive use with analgesics may be obtained by the administration of 12.5 to 25 mg in children and 25- to 50-mg doses in adults.
Promethazine Syrup is contraindicated for children under 2 years of age.
CONTRAINDICATIONS
Promethazine Syrup is contraindicated for use in pediatric patients less than two years of age.
Promethazine Syrup is contraindicated in comatose states, and in individuals known to be hypersensitive or to have had an idiosyncratic reaction to promethazine or to other phenothiazines.
Antihistamines are contraindicated for use in the treatment of lower respiratory tract symptoms including asthma.
ADVERSE REACTIONS
Central Nervous System
Drowsiness is the most prominent CNS effect of this drug. Sedation, somnolence, blurred vision, dizziness; confusion, disorientation, and extrapyramidal symptoms such as oculogyric crisis, torticollis, and tongue protrusion; lassitude, tinnitus, incoordination, fatigue, euphoria, nervousness, diplopia, insomnia, tremors, convulsive seizures, excitation, catatonic-like states, hysteria. Hallucinations have also been reported.
Cardiovascular -Increased or decreased blood pressure, tachycardia, bradycardia, faintness.
Dermatologic -Dermatitis, photosensitivity, urticaria.
Hematologic -Leukopenia, thrombocytopenia, thrombocytopenic purpura, agranulocytosis.
Gastrointestinal -Dry mouth, nausea, vomiting, jaundice.
Respiratory -Asthma, nasal stuffiness, respiratory depression (potentially fatal) and apnea (potentially fatal). (See WARNINGS-Respiratory Depression .)
Other -Angioneurotic edema. Neuroleptic malignant syndrome (potentially fatal) has also been reported. (See WARNINGS-Neuroleptic Malignant Syndrome .)
Paradoxical Reactions
Hyperexcitability and abnormal movements have been reported in patients following a single administration of promethazine HCl. Consideration should be given to the discontinuation of promethazine HCl and to the use of other drugs if these reactions occur. Respiratory depression, nightmares, delirium, and agitated behavior have also been reported in some of these patients.
Drug Interactions
CNS Depressants
Promethazine Syrup may increase, prolong, or intensify the sedative action of other central-nervous-system depressants, such as alcohol, sedatives/hypnotics (including barbiturates), narcotics, narcotic analgesics, general anesthetics, tricyclic antidepressants, and tranquilizers; therefore, such agents should be avoided or administered in reduced dosage to patients receiving promethazine HCl. When given concomitantly with Promethazine Syrup, the dose of barbiturates should be reduced by at least one-half, and the dose of narcotics should be reduced by one-quarter to one-half. Dosage must be individualized. Excessive amounts of promethazine HCl relative to a narcotic may lead to restlessness and motor hyperactivity in the patient with pain; these symptoms usually disappear with adequate control of the pain.
Epinephrine
Because of the potential for Promethazine to reverse epinephrine's vasopressor effect, epinephrine should NOT be used to treat hypotension associated with Promethazine Syrup overdose.
Anticholinergics
Concomitant use of other agents with anticholinergic properties should be undertaken with caution.
Monoamine Oxidase Inhibitors (MAOI)
Drug interactions, including an increased incidence of extrapyramidal effects, have been reported when some MAOI and phenothiazines are used concomitantly. This possibility should be considered with Promethazine Syrup.
DESCRIPTION
Each 5 mL (teaspoonful) of Promethazine contains 6.25 mg of promethazine HCl. The inactive ingredients present are artificial banana flavor, artificial fruit flavor, artificial strawberry flavor, ascorbic acid, citric acid anhydrous, dehydrated alcohol, D&C Yellow No. 10, edetate disodium, FD&C Blue No. 1, FD&C Red No. 40, glycerin, sucrose, methylparaben, purified water, saccharin sodium, sodium benzoate, sodium citrate dihydrate, and sodium propionate.
Promethazine HCl is a racemic compound; the molecular formula is C17H20N2S•HCl and its molecular weight is 320.88.
Promethazine HCl, a phenothiazine derivative, is designated chemically as 10H-Phenothiazine-10-ethanamine, N,N, -trimethyl-, monohydrochloride, (±)- with the following structural formula:
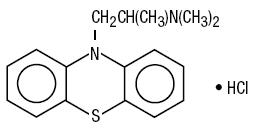
Promethazine HCl occurs as a white to faint yellow, practically odorless, crystalline powder which slowly oxidizes and turns blue on prolonged exposure to air. It is freely soluble in water and soluble in alcohol.
CLINICAL PHARMACOLOGY
Promethazine is a phenothiazine derivative which differs structurally from the antipsychotic phenothiazines by the presence of a branched side chain and no ring substitution. It is thought that this configuration is responsible for its relative lack (1/10 that of chlorpromazine) of dopamine antagonist properties.
Promethazine is an H1 receptor blocking agent. In addition to its antihistaminic action, it provides clinically useful sedative and antiemetic effects.
Promethazine is well absorbed from the gastrointestinal tract. Clinical effects are apparent within 20 minutes after oral administration and generally last four to six hours, although they may persist as long as 12 hours. Promethazine is metabolized by the liver to a variety of compounds; the sulfoxides of promethazine and N-demethylpromethazine are the predominant metabolites appearing in the urine.
HOW SUPPLIED
Promethazine Syrup Plain 6.25 mg/5 mL is available in PET plastic bottles as follows:
- NDC# 0116-4023-16: Pint (473 mL)
STORAGE AND HANDLING
Keep tightly closed.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]
Protect from light.
Dispense in light-resistant, tight container.