Promethazine Hydrochloride - Promethazine Hydrochloride injection prescribing information
WARNINGS: RESPIRATORY DEPRESSION IN PEDIATRICS AND SEVERE TISSUE INJURY INCLUDING GANGRENE
Respiratory Depression – Pediatrics
- Promethazine hydrochloride injection should not be used in pediatric patients less than 2 years of age because of the potential for fatal respiratory depression.Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine in pediatric patients less than 2 years of age.Caution should be exercised when administering promethazine hydrochloride injection to pediatric patients 2 years of age and older (see WARNINGS – Respiratory Depression )
Severe Tissue Injury, Including Gangrene
- Severe chemical irritation and damage to tissues regardless of the route of parenteral administration has been reported in patients treated with promethazine hydrochloride injection including gangrene, tissue necrosis, and thrombophlebitis; and in some cases, surgical intervention including fasciotomy, skin graft, and/or amputation have been required (see WARNINGS – Severe Tissue Injury, Including Gangrene .).
- The use of promethazine hydrochloride injection by the following routes of administration is CONTRAINDICATED:
- Intravenous injection at concentrations greater than 1 mg/mL
- Intra-arterial injection
- Subcutaneous injection (see CONTRAINDICATIONS ).
- The preferred route of administration is by deep intramuscular administration.
- Promethazine hydrochloride injection may be administered intravenously after dilution through an intravenous catheter inserted in a large vein. Preferably through a central venous catheter (see DOSAGE AND ADMINISTRATION ).
- If pain occurs at the injection site during intravenous infusion, immediately discontinue the infusion and evaluate for possible arterial injection or perivascular extravasation, and initiate appropriate medical management.
INDICATIONS AND USAGE
Promethazine hydrochloride injection is indicated for the following conditions:
- Amelioration of allergic reactions to blood or plasma.
- In anaphylaxis as an adjunct to epinephrine and other standard measures after the acute symptoms have been controlled.
- For other uncomplicated allergic conditions of the immediate type when oral therapy is impossible or contraindicated.
- For sedation and relief of apprehension and to produce light sleep from which the patient can be easily aroused.
- Active treatment of motion sickness.
- Prevention and control of nausea and vomiting associated with certain types of anesthesia and surgery.
- As an adjunct to analgesics for the control of postoperative pain.
- Preoperative, postoperative, and obstetric (during labor) sedation.
- Intravenously in special surgical situations, such as repeated bronchoscopy, ophthalmic surgery, and poor-risk patients, with reduced amounts of meperidine or other narcotic analgesic as an adjunct to anesthesia and analgesia.
DOSAGE AND ADMINISTRATION
Important Administration Information for Adult and Pediatric Patients 2 Years of Age and Older
- The preferred route of administration of promethazine hydrochloride injection is by deep intramuscular administration (see WARNINGS - Severe Tissue Injury, Including Gangrene ).
- Promethazine hydrochloride injection may be administered intravenously after dilution as recommended below (see Preparation and Administration ). If pain occurs at the injection site during intravenous infusion, immediately discontinue the infusion, and evaluate for possible arterial injection or perivascular extravasation.
Allergic Conditions
The average adult dose is 25 mg. This dose may be repeated within two hours if necessary, but continued therapy, if indicated, should be via the oral route as soon as existing circumstances permit. After initiation of treatment, dosage should be adjusted to the smallest amount adequate to relieve symptoms. The average adult dose for amelioration of allergic reactions to blood or plasma is 25 mg.
Sedation
In hospitalized adult patients, nighttime sedation may be achieved by a dose of 25 to 50 mg of promethazine hydrochloride injection.
Nausea and Vomiting
For control of nausea and vomiting, the usual adult dose is 12.5 to 25 mg, not to be repeated more frequently than every four hours. When used for control of postoperative nausea and vomiting, the dosage of analgesics and barbiturates should be reduced accordingly (see PRECAUTIONS - Drug Interactions ).
Antiemetics should not be used in vomiting of unknown etiology in children and adolescents (see PRECAUTIONS - Pediatric Use ).
Preoperative and Postoperative Use
As an adjunct to preoperative or postoperative medication, 25 to 50 mg of promethazine hydrochloride injection in adults may be combined with appropriately reduced doses of analgesics and atropine-like drugs as desired. Dosage of concomitant analgesic or hypnotic medication should be reduced accordingly (see PRECAUTIONS - Drug Interactions ).
Promethazine hydrochloride is contraindicated for use in pediatric patients less than two years of age.
Obstetrics
Promethazine hydrochloride injection in doses of 50 mg will provide sedation and relieve apprehension in the early stages of labor. When labor is definitely established, 25 to 75 mg (average dose, 50 mg) promethazine hydrochloride injection may be given with an appropriately reduced dose of any desired narcotic (see PRECAUTIONS - Drug Interactions ). If necessary, promethazine hydrochloride injection with a reduced dose of analgesic may be repeated once or twice at four-hour intervals in the course of a normal labor. A maximum total dose of 100 mg of promethazine hydrochloride injection may be administered during a 24-hour period to patients in labor.
Pediatric Patients
Promethazine hydrochloride injection is contraindicated for use in pediatric patients less than 2 years of age (see WARNINGS - Respiratory Depression ). Caution should be exercised when administering promethazine hydrochloride to pediatric patients 2 years of age or older. It is recommended that the lowest effective dose of promethazine hydrochloride be used in pediatric patients 2 years of age and older and concomitant administration of other drugs with respiratory depressant effects be avoided (see WARNINGS - Respiratory Depression ).
In pediatric patients 2 years of age and older, the dosage should not exceed half that of the suggested adult dose. As an adjunct to premedication, the suggested dose is 1.1 mg per kg of body weight in combination with an appropriately reduced dose of narcotic or barbiturate and the appropriate dose of an atropine-like drug (see PRECAUTIONS - Drug Interactions ). Antiemetics should not be used in vomiting of unknown etiology in pediatric patients.
Preparation and Administration Instructions for Diluted Intravenous Infusion in Adults
Inspect the solution for particulate matter and discoloration, before dilution, after dilution, and before administration . Discard the vial, ampule, or bag if particulates and/or discoloration are observed.
1. Determine the recommended dose of promethazine hydrochloride injection.
2. Aseptically withdraw the required volume from the vial or ampule and transfer it into an infusion bag.
- Use a filter needle when withdrawing promethazine hydrochloride injection from the ampule.
3. Dilute with the recommended volume of 0.9% Sodium Chloride Injection, see Table 1. Avoid mixing and/or diluting with any other drugs or solutions other than 0.9% Sodium Chloride Injection.
4. Gently invert the infusion bag.
5. Check patency of the access site before administration.
6. Administer the diluted solution through an intravenous catheter, inserted in a large vein (preferably through a central venous catheter), over 20 to 40 minutes; for maximum infusion rates, see Table 1.
- Do NOT administer via an intravenous catheter in the hand or wrist.
7. If pain occurs at the injection site during intravenous infusion, immediately discontinue the infusion and evaluate for possible arterial injection or perivascular extravasation.
| Dose of Promethazine Hydrochloride Injection | Volume of 0.9% Sodium Chloride Injection for Dilution | Maximum Concentration of the Diluted Promethazine Hydrochloride Injection Solution | Maximum Rate of Infusion |
| 12.5 mg | 50 mL | 1 mg/mL | 2.5 mL/minute |
| 25 mg | 50 mL | 2.5 mL/minute | |
| 50 mg | 50 mL | 2.5 mL/minute | |
| 75 mg | 100 mL | 5 mL/minute |
Preparation and Administration Instructions for Diluted Intravenous Infusion to Pediatric Patients 2 Years of Age and Older.
Inspect the solution for particulate matter and discoloration, before dilution, after dilution, and before administration. Discard the vial, ampule, syringe or bag if particulates and/or discoloration are observed.
1. Determine the recommended dose of promethazine hydrochloride injection for pediatric patients 2 years of age and older.
2. Aseptically withdraw the required volume from the vial or ampule and transfer it into an appropriately sized syringe or bag for use with an infusion pump.
- Use a filter needle when withdrawing promethazine hydrochloride injection from the ampule.
3. Dilute with the recommended volume of 0.9% Sodium Chloride Injection, see Table 2. Avoid mixing and/or diluting with any other drugs or solutions other than 0.9% Sodium Chloride Injection.
4. Gently invert the syringe or bag.
5. Check patency of the access site before administration.
6. Administer the diluted solution through an intravenous catheter, inserted in a large vein (preferably through a central venous catheter) with a maximum infusion rate of 1.25 mL/minute.
- Do NOT administer via an intravenous catheter in the hand or wrist.
7. If pain occurs at the injection site during intravenous infusion, immediately discontinue the infusion and evaluate for possible arterial injection or perivascular extravasation.
| Dose of Promethazine Hydrochloride Injection | Volume of 0.9% Sodium Chloride Injection for Dilution | Maximum Concentration of the Diluted Promethazine Hydrochloride Injection Solution | Maximum Rate of Infusion |
| Up to 25 mg | 25 mL | 1 mg/mL | 1.25 mL/minute |
| 25 mg to 50 mg | 50 mL |
•Avoid mixing and/or diluting with any other drugs or solutions besides 0.9% Sodium Chloride Injection
Storage of Diluted Promethazine Hydrochloride Injection
Diluted solution may be stored up to 4 hours at room temperature, 20°C to 25°C (68°F to 77°F), or refrigerated at 2°C to 8°C (36° to 46°F), up to 24 hours.
CONTRAINDICATIONS
The use of promethazine hydrochloride injection is contraindicated:
• In pediatric patients less than 2 years of age due to the risk of respiratory depression (see WARNINGS – Respiratory Depression ).
• For use as an intravenous injection at concentrations greater than 1 mg/mL due to the risk of perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration (see WARNINGS - Severe Tissue Injury, Including Gangrene and DOSAGE AND ADMINISTRATION ).
• For use as an intra-arterial injection due to the likelihood of severe arteriospasm and the possibility of resultant gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene ).
• For use as a subcutaneous injection because chemical irritation and necrotic lesions have been reported (see WARNINGS - Severe Tissue Injury, Including Gangrene ).
• In patients in a comatose state.
• In patients who have demonstrated an idiosyncratic reaction or hypersensitivity to promethazine hydrochloride or other phenothiazines.
ADVERSE REACTIONS
Respiratory Depression
Promethazine hydrochloride injection is contraindicated in pediatric patients less than 2 years of age, because of the potential for fatal respiratory depression. Promethazine hydrochloride injection should be used with caution in pediatric patients 2 years of age and older (see WARNINGS - Respiratory Depression ).
Severe Tissue Injury, Including Gangrene
Promethazine hydrochloride injection has been reported to cause severe chemical irritation and damage to tissues, including gangrene, regardless of the route of parenteral administration . Intra-arterial and subcutaneous injection have resulted in more significant complications (see C ONTRAINDICATIONS ). Reported adverse reactions included burning, pain, erythema, swelling, sensory loss, palsies, paralysis, severe spasm of distal vessels, thrombophlebitis, venous thrombosis, phlebitis, abscesses, tissue necrosis, and gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene ).
Central Nervous System
Drowsiness is the most prominent CNS effect of this drug. Sedation, somnolence, blurred vision, dizziness, confusion, disorientation, and extrapyramidal symptoms such as oculogyric crisis, torticollis, and tongue protrusion; lassitude, tinnitus, incoordination, fatigue, euphoria, nervousness, diplopia, insomnia, tremors, convulsive seizures, excitation, catatonic-like states, hysteria. Hallucinations have also been reported.
Cardiovascular
Increased or decreased blood pressure, tachycardia, bradycardia, faintness.
Dermatologic
Dermatitis, photosensitivity, urticaria.
Hematologic
Leukopenia, thrombocytopenia, thrombocytopenic purpura, agranulocytosis.
Gastrointestinal
Dry mouth, nausea, vomiting, jaundice.
Respiratory
Asthma, nasal stuffiness, respiratory depression (potentially fatal) and apnea (potentially fatal). (See WARNINGS - Respiratory Depression .)
Other
Angioneurotic edema. Neuroleptic Malignant Syndrome (potentially fatal) has also been reported. (See WARNINGS - Neuroleptic Malignant Syndrome .)
Paradoxical Reactions
Hyperexcitability and abnormal movements have been reported in patients following a single administration of promethazine hydrochloride injection. Consideration should be given to the discontinuation of promethazine hydrochloride injection and to the use of other drugs if these reactions occur. Respiratory depression, nightmares, delirium, and agitated behavior have also been reported in some of these patients.
Drug Interactions
CNS Depressants
Promethazine hydrochloride injection may increase, prolong, or intensify the sedative action of central-nervous-system depressants, such as alcohol, sedative/hypnotics (including barbiturates), general anesthetics, narcotics, narcotic analgesics, tricyclic antidepressants, and tranquilizers; therefore, such agents should be avoided or administered in reduced dosage to patients receiving promethazine hydrochloride. When given concomitantly with promethazine hydrochloride injection, the dose of barbiturates should be reduced by at least one-half, and the dose of narcotics should be reduced by one-quarter to one-half. Dosage must be individualized. Excessive amounts of promethazine hydrochloride injection relative to a narcotic may lead to restlessness and motor hyperactivity in the patient with pain; these symptoms usually disappear with adequate control of the pain.
Epinephrine
Because of the potential for promethazine hydrochloride to reverse epinephrine’s vasopressor effect, epinephrine should NOT be used to treat hypotension associated with promethazine hydrochloride injection overdose.
Anticholinergics
Concomitant use of other agents with anticholinergic properties should be undertaken with caution.
Monoamine Oxidase (MAO) Inhibitors
Drug interactions, including an increased incidence of extrapyramidal effects, have been reported when some MAO Inhibitors and phenothiazines are used concomitantly. This possibility should be considered with promethazine hydrochloride injection.
DESCRIPTION
Promethazine hydrochloride injection, USP is a sterile, pyrogen-free solution for deep intramuscular or intravenous administration. Promethazine hydrochloride (10 H -Phenothiazine-10-ethanamine, N,N,α -trimethyl-, monohydrochloride, (±)-) is a racemic compound and has the following structural formula:
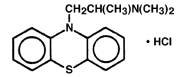
C 17 H 21 ClN 2 S MW 320.88
Each mL contains promethazine hydrochloride, either 25 mg or 50 mg, edetate disodium 0.1 mg, calcium chloride 0.04 mg, sodium metabisulfite 0.25 mg and phenol 5 mg in Water for Injection. pH 4.0 to 5.5; buffered with acetic acid-sodium acetate.
Promethazine hydrochloride injection is a clear, colorless solution. The product is light sensitive. It should be inspected before use and discarded if either color or particulate is observed.
CLINICAL PHARMACOLOGY
Promethazine hydrochloride is a phenothiazine derivative which possesses antihistaminic, sedative, antimotion-sickness, antiemetic, and anticholinergic effects. Promethazine is a competitive H 1 receptor antagonist, but does not block the release of histamine. Structural differences from the neuroleptic phenothiazines result in its relative lack (1 / 10 that of chlorpromazine) of dopamine antagonist properties. Clinical effects are generally apparent within 5 minutes of an intravenous injection and within 20 minutes of an intramuscular injection. Duration of action is four to six hours, although effects may persist up to 12 hours. Promethazine hydrochloride is metabolized in the liver, with the sulfoxides of promethazine and N-desmethylpromethazine being the predominant metabolites appearing in the urine. Following intravenous administration in healthy volunteers, the plasma half-life for promethazine has been reported to range from 9 to 16 hours. The mean plasma half-life for promethazine after intramuscular administration in healthy volunteers has been reported to be 9.8 ± 3.4 hours.
HOW SUPPLIED
Promethazine Hydrochloride Injection, USP is available as follows:
25 mg/mL 1 mL ampuls packaged in 25s (NDC 0641-0948-35)
50 mg/mL 1 mL ampuls packaged in 25s (NDC 0641-0949-35)
Storage
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Protect from light. Keep covered in carton until time of use. Do not use if solution has developed color or contains a precipitate.
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Pharmaceuticals USA Inc. at 1-877-845-0689, or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
For Product Inquiry call 1-877-845-0689.
Manufactured by: HIKMA FARMACÊUTICA (PORTUGAL), S.A. Estrada do Rio da Mό, 8, 8A e 8B – Fervença –2705-906 Terrugem SNT, PORTUGAL
Distributed by: Hikma Pharmaceuticals USA Inc. Berkeley Heights, NJ 07922
Revised December 2023
462-640-03
PIN293-NOV/4
Novaplus is a registered trademark of Vizient, Inc.
novaplus+