Get your patient on Rifabutin - Rifabutin capsule (Rifabutin)
Rifabutin - Rifabutin capsule prescribing information
INDICATIONS AND USAGE
Rifabutin capsules are indicated for the prevention of disseminated Mycobacterium avium complex (MAC) disease in patients with advanced HIV infection.
DOSAGE AND ADMINISTRATION
It is recommended that rifabutin capsules be administered at a dose of 300 mg once daily. For those patients with propensity to nausea, vomiting, or other gastrointestinal upset, administration of rifabutin at doses of 150 mg twice daily taken with food may be useful. Doses of Rifabutin capsules may be administered mixed with foods such as applesauce.
For patients with severe renal impairment (creatinine clearance less than 30 mL/min), consider reducing the dose of rifabutin by 50%, if toxicity is suspected. No dosage adjustment is required for patients with mild to moderate renal impairment. Reduction of the dose of rifabutin may also be needed for patients receiving concomitant treatment with certain other drugs (see PRECAUTIONS-Drug Interactions ).
Mild hepatic impairment does not require a dose modification. The pharmacokinetics of rifabutin in patients with moderate and severe hepatic impairment is not known.
CONTRAINDICATIONS
Rifabutin capsules are contraindicated in patients who have had clinically significant hypersensitivity to rifabutin or to any other rifamycins.
Rifabutin capsules are contraindicated in patients being treated with cabotegravir/rilpivirine prolonged-release injectable suspension (see PRECAUTIONS-Drug Interactions, Table 2 ).
ADVERSE REACTIONS
Adverse Reactions from Clinical Trials
Rifabutin capsules were generally well tolerated in the controlled clinical trials. Discontinuation of therapy due to an adverse event was required in 16% of patients receiving rifabutin, compared to 8% of patients receiving placebo in these trials. Primary reasons for discontinuation of rifabutin were rash (4% of treated patients), gastrointestinal intolerance (3%), and neutropenia (2%).
The following table enumerates adverse experiences that occurred at a frequency of 1% or greater, among the patients treated with rifabutin in studies 023 and 027.
| Adverse event | Rifabutin (n = 566) % | Placebo (n = 580) % |
|---|---|---|
Body as a whole | ||
Abdominal pain | 4 | 3 |
Asthenia | 1 | 1 |
Chest pain | 1 | 1 |
Fever | 2 | 1 |
Headache | 3 | 5 |
Pain | 1 | 2 |
Blood and lymphatic system | ||
Leucopenia | 10 | 7 |
Anemia | 1 | 2 |
Digestive System | ||
Anorexia | 2 | 2 |
Diarrhea | 3 | 3 |
Dyspepsia | 3 | 1 |
Eructation | 3 | 1 |
Flatulence | 2 | 1 |
Nausea | 6 | 5 |
Nausea and vomiting | 3 | 2 |
Vomiting | 1 | 1 |
Musculoskeletal system | ||
Myalgia | 2 | 1 |
Nervous system | ||
Insomnia | 1 | 1 |
Skin and appendages | ||
Rash | 11 | 8 |
Special senses | ||
Taste perversion | 3 | 1 |
Urogenital system | ||
Discolored urine | 30 | 6 |
CLINICAL ADVERSE EVENTS REPORTED IN <1% OF PATIENTS WHO RECEIVED RIFABUTIN
Considering data from the 023 and 027 pivotal trials, and from other clinical studies, rifabutin appears to be a likely cause of the following adverse events which occurred in less than 1% of treated patients: flu-like syndrome, hepatitis, hemolysis, arthralgia, myositis, chest pressure or pain with dyspnea, skin discoloration, thrombocytopenia, pancytopenia and jaundice.
The following adverse events have occurred in more than one patient receiving rifabutin, but an etiologic role has not been established: seizure, paresthesia, aphasia, confusion, and non-specific T wave changes on electrocardiogram.
When rifabutin was administered at doses from 1050 mg/day to 2400 mg/day, generalized arthralgia and uveitis were reported. These adverse experiences abated when rifabutin was discontinued.
Mild to severe, reversible uveitis has been reported less frequently when rifabutin is used at 300 mg as monotherapy in MAC prophylaxis versus rifabutin in combination with clarithromycin for MAC treatment (see also WARNINGS ).
Uveitis has been infrequently reported when rifabutin is used at 300 mg/day as monotherapy in MAC prophylaxis of HIV-infected persons, even with the concomitant use of fluconazole and/or macrolide antibacterials. However, if higher doses of rifabutin are administered in combination with these agents, the incidence of uveitis is higher.
Patients who developed uveitis had mild to severe symptoms that resolved after treatment with corticosteroids and/or mydriatic eye drops; in some severe cases, however, resolution of symptoms occurred after several weeks.
When uveitis occurs, temporary discontinuance of rifabutin and ophthalmologic evaluation are recommended. In most mild cases, rifabutin may be restarted; however, if signs or symptoms recur, use of rifabutin should be discontinued (Morbidity and Mortality Weekly Report, September 9, 1994).
Corneal deposits have been reported during routine ophthalmologic surveillance of some HIV-positive pediatric patients receiving rifabutin as part of a multiple drug regimen for MAC prophylaxis. The deposits are tiny, almost transparent, asymptomatic peripheral and central corneal deposits, and do not impair vision.
The following table enumerates the changes in laboratory values that were considered as laboratory abnormalities in Studies 023 and 027.
| Laboratory abnormalities | Rifabutin (n = 566) % | Placebo (n = 580) % |
|---|---|---|
| Includes grades 3 or 4 toxicities as specified: | ||
Chemistry | ||
Increased alkaline phosphatase All values >450 U/L | <1 | 3 |
Increased SGOT All values >150 U/L | 7 | 12 |
Increased SGPT | 9 | 11 |
Hematology | ||
Anemia All hemoglobin values <8.0 g/dL | 6 | 7 |
Eosinophilia | 1 | 1 |
Leukopenia All WBC values <1,500/mm 3 | 17 | 16 |
Neutropenia All ANC values <750/mm 3 | 25 | 20 |
Thrombocytopenia All platelet count values <50,000/mm 3 | 5 | 4 |
The incidence of neutropenia in patients treated with rifabutin was significantly greater than in patients treated with placebo (p = 0.03). Although thrombocytopenia was not significantly more common among patients treated with rifabutin in these trials, rifabutin has been clearly linked to thrombocytopenia in rare cases. One patient in Study 023 developed thrombotic thrombocytopenic purpura, which was attributed to rifabutin.
Adverse Reactions from Post-Marketing Experience
Adverse reactions identified through post-marketing surveillance by system organ class (SOC) are listed below:
Blood and lymphatic system disorders: White blood cell disorders (including agranulocytosis, lymphopenia, granulocytopenia, neutropenia, white blood cell count decreased, neutrophil count decreased), platelet count decreased.
Immune system disorders: Hypersensitivity, bronchospasm, rash, and eosinophilia.
Gastrointestinal disorders: Clostridioides difficile colitis/ Clostridioides difficile associated diarrhea.
Pyrexia, rash and other hypersensitivity reactions such as eosinophilia and bronchospasm might occur, as has been seen with other antibacterials.
A limited occurrence of skin discoloration has been reported.
Severe cutaneous adverse reactions (SCARs):
Rifabutin has been associated with the occurrence of DRESS as well as other SCARs such as SJS, TEN, and AGEP (see WARNINGS ).
Rifamycin hypersensitivity reactions:
Hypersensitivity to rifamycins have been reported including flu-like symptoms, bronchospasm, hypotension, urticaria, angioedema, conjunctivitis, thrombocytopenia or neutropenia.
Drug Interactions
Effect of Rifabutin on the Pharmacokinetics of Other Drugs
Rifabutin induces CYP3A enzymes and therefore may reduce the plasma concentrations of drugs metabolized by those enzymes. This effect may reduce the efficacy of standard doses of such drugs, which include itraconazole, clarithromycin, and saquinavir.
Effect of Other Drugs on Rifabutin Pharmacokinetics
Some drugs that inhibit CYP3A may significantly increase the plasma concentration of rifabutin. Therefore, carefully monitor for rifabutin associated adverse events in those patients also receiving CYP3A inhibitors, which include fluconazole and clarithromycin. In some cases, the dosage of rifabutin may need to be reduced when it is co-administered with CYP3A inhibitors.
Table 2 summarizes the results and magnitude of the pertinent drug interactions assessed with rifabutin. The clinical relevance of these interactions and subsequent dose modifications should be judged in light of the population studied, severity of the disease, patient's drug profile, and the likely impact on the risk/benefit ratio.
| Co-administered drug | Dosing regimen of co-administered drug | Dosing regimen of rifabutin | Study population (n) | Effect on rifabutin | Effect on co-administered drug | Recommendation |
|---|---|---|---|---|---|---|
| ↑ indicates increase; ↓ indicates decrease; ↔ indicates no significant change | ||||||
| ND - No Data | ||||||
| AUC - Area under the Concentration vs. Time Curve; C max - Maximum serum concentration; C min – Minimum serum concentration | ||||||
ANTIRETROVIRALS | ||||||
Amprenavir | 1200 mg twice a day for 10 days | 300 mg once a day for 10 days | Healthy male subjects (6) | ↑ AUC by 193%, ↑ C max by 119% | ↔ | Reduce rifabutin dose by at least 50%. Monitor closely for adverse reactions. |
Atazanavir/Ritonavir | 300/100 mg once daily | 150 mg twice weekly | Healthy adult subjects | 48% ↑ in AUC, 149% ↑ C max of rifabutin. 990% ↑ in AUC, 677% ↑ C max of 25‑O‑desacetyl-rifabutin. | No significant change in pharmacokinetics. | A reduction in the dose of rifabutin (to 150 mg every other day or 3 times a week) is recommended. Increased monitoring for adverse reactions is warranted. |
Bictegravir | 75 mg once a day | 300 mg once a day (fasted) | Healthy subjects | ND | ↓ AUC 38% ↓ C min 56% ↓ C max 20% | Co-administration of rifabutin with Biktarvy (bictegravir/emtricitabine/ tenofovir alafenamide) is not recommended due to an expected decrease in tenofovir alafenamide in addition to the reported reduction in bictegravir. Refer to Biktarvy prescribing information for additional information. |
Darunavir/Ritonavir | 600/100 mg twice a day for 12 days | 150 mg every other day for 12 days | Healthy HIV- negative adults | No significant change in rifabutin pharmacokinetics. 881% ↑ in AUC, 377% ↑ C max of 25‑O‑desacetyl-rifabutin. | 57% ↑ in AUC, 42% ↑ C max of darunavir. 66% ↑ in AUC, 68% ↑ C max of ritonavir. | A reduction in the dose of rifabutin (to 150 mg every other day or 3 times a week) is recommended. Increased monitoring for adverse reactions is warranted. |
Delavirdine | 400 mg three times a day | 300 mg once a day | HIV-infected patients (7) | ↑ AUC by 230%, ↑ C max by 128% | ↓ AUC by 80%, ↓ C max by 75%, ↓ C min by 17% | CONTRAINDICATED |
Didanosine | 167 or 250 mg twice a day for 12 days | 300 or 600 mg once a day for 12 days | HIV-infected patients (11) | ↔ | ↔ | |
Dolutegravir | 50 mg daily for 14 days | 300 mg daily for 14 days | Healthy adult subjects | ND | No significant change in dolutegravir pharmacokinetics at steady state. | |
Doravirine | 100 mg single dose | 300 mg once a day for 16 days | Healthy subjects (12) | ND | ↓ 50% in AUC, ↓ 68% in C 24 ↔ in C max | If concomitant use is necessary, increase the doravirine dosage as instructed in doravirine-containing product prescribing information. |
Elvitegravir/Cobicistat | 150/50 mg daily | 300 mg daily Or 150 mg every other day | Healthy subjects (12) | No significant change in rifabutin pharmacokinetics. 6.3-fold ↑ in AUC, 4.8-fold ↑ C max of 25‑O‑desacetyl-rifabutin. | No change in elvitegravir except 67% ↓ C trough of elvitegravir. No change in cobicistat exposure. | Co-administration of rifabutin with elvitegravir/ cobicistat is not recommended due to an expected decrease in elvitegravir exposure. |
Etravirine | 800 mg twice daily for 21 days | 300 mg daily on days 8 to 21 | Healthy volunteers (18) | No significant change in rifabutin pharmacokinetics. | 37% ↓ in AUC, 37% ↓ in C max and 35% ↓ in C min | No dose adjustment of rifabutin is required when etravirine is not co-administered with protease inhibitor/ritonavir. Rifabutin should not be co-administered with etravirine and boosted PIs due to potential for decreased effectiveness of etravirine. |
Fosamprenavir/ritonavir | 700 mg twice a day plus ritonavir 100 mg twice a day for 2 weeks | 150 mg every other day for 2 weeks | Healthy subjects (15) | ↔ AUC compared to rifabutin 300 mg once a day alone ↓ C max by 15% | ↑ AUC by 35% compared to historical control (fosamprenavir/ritonavir 700/100 mg twice a day) , ↑ C max by 36%, ↑ C min by 36% | Reduce rifabutin dose by at least 75% (to a maximum 150 mg every other day or three times per week) when given with fosamprenavir/ritonavir combination. |
Indinavir | 800 mg three times a day for 10 days | 300 mg once a day for 10 days | Healthy subjects (10) | ↑ AUC by 173%, ↑ C max by 134% | ↓ AUC by 34%, ↓ C max by 25%, ↓ C min by 39% | Reduce rifabutin dose by 50%, and increase indinavir dose from 800 mg to 1000 mg three times a day. |
Lopinavir/ ritonavir | 400/100 mg twice a day for 20 days | 150 mg once a day for 10 days | Healthy subjects (14) | ↑ AUC by 203% also taking zidovudine 500 mg once a day ↓ C max by 112% | ↔ | Reduce rifabutin dose by at least 75% (to a maximum 150 mg every other day or three times per week) when given with lopinavir/ritonavir combination. Monitor closely for adverse reactions. Reduce rifabutin dosage further, as needed. |
Saquinavir/ritonavir | 1000/100 mg twice a day for 14 or 22 days | 150 mg every 3 days for 14 or 22 days | Healthy subjects | ↑ AUC by 53% compared to rifabutin 150 mg once a day alone ↑ C max by 88% (n=11) | ↓ AUC by 13%, ↓ C max by 15%, (n=19) | Reduce rifabutin dose by at least 75% (to a maximum 150 mg every other day or three times per week) when given with saquinavir/ritonavir combination. Monitor closely for adverse reactions. |
Rilpivirine | 25 mg once a day | 300 mg once a day | Healthy subjects (18) | ND | ↓ AUC by 42% ↓ C min by 48% ↓ C max by 31% | Co-administration of rifabutin with Odefsey (rilpivirine/tenofovir alafenamide/emtricitabine) is not recommended, due to an expected decrease in tenofovir alafenamide in addition to the reported reduction in rilpivirine. Refer to Odefsey prescribing information for additional information. Co-administration of rifabutin with cabotegravir/rilpivirine prolonged-release injectable suspension is contraindicated. |
Ritonavir | 500 mg twice a day for 10 days | 150 mg once a day for 16 days | Healthy subjects (5) | ↑ AUC by 300%, ↑ C max by 150% | ND | Reduce rifabutin dose by at least 75% (to a maximum 150 mg every other day or three times per week) when given with lopinavir/ritonavir combination. Monitor closely for adverse reactions. Reduce rifabutin dosage further, as needed. |
Tipranavir/ritonavir | 500/200 twice a day for 15 doses | 150 mg single dose | Healthy subjects (20) | ↑ AUC by 190%, ↑ C max by 70% | ↔ | Reduce rifabutin dose by at least 75% (to a maximum 150 mg every other day or three times per week) when given with tipranavir/ritonavir combination. Monitor closely for adverse reactions. Reduce rifabutin dosage further, as needed. |
Nelfinavir | 1250 mg twice a day for 7–8 days | 150 mg once a day for 8 days | HIV-infected patients (11) | ↑ AUC by 83%, compared to rifabutin 300 mg once a day alone ↑ C max by 19% | ↔ | Reduce rifabutin dose by 50% (to 150 mg once a day) and increase the nelfinavir dose to 1250 mg twice a day. |
Zidovudine | 100 or 200 mg every four hours | 300 or 450 mg once a day | HIV-infected patients (16) | ↔ | ↓ AUC by 32%, ↓ C max by 48%, | Because zidovudine levels remained within the therapeutic range during co-administration of rifabutin, dosage adjustments are not necessary. |
ANTI-HCV DRUGS | ||||||
Sofosbuvir | 400 mg on day 1 and day 21 | 300 mg daily on day 10 to day 29 | Healthy subjects (20) | ND | 36% ↓ in C max and 24% ↓ AUC | Co-administration of rifabutin with sofosbuvir (alone or in combination) is not recommended. |
ANTIFUNGALS | ||||||
Fluconazole | 200 mg once a day for 2 weeks | 300 mg once a day for 2 weeks | HIV-infected patients (12) | ↑ AUC by 82%, ↑ C max by 88% | ↔ | Monitor for rifabutin associated adverse events. Reduce rifabutin dose or suspend rifabutin use if toxicity is suspected. |
Posaconazole | 200 mg once a day for 10 days | 300 mg once a day for 17 days | Healthy subjects (8) | ↑ AUC by 72%, ↑ C max by 31% | ↓ AUC by 49%, ↓ C max by 43% | If co-administration of these two drugs cannot be avoided, patients should be monitored for adverse events associated with rifabutin administration, and lack of posaconazole efficacy. |
Itraconazole | 200 mg once a day | 300 mg once a day | HIV-Infected patients (6) | ↑ data from a case report | ↓ AUC by 70%, ↓ C max by 75%, | If co-administration of these two drugs cannot be avoided, patients should be monitored for adverse events associated with rifabutin administration, and lack of itraconazole efficacy. In a separate study, one case of uveitis was associated with increased serum rifabutin levels following co-administration of rifabutin (300 mg once a day) with itraconazole (600–900 mg once a day). |
Voriconazole | 400 mg twice a day for 7 days (maintenance dose) | 300 mg once a day for 7 days | Healthy male subjects (12) | ↑ AUC by 331%, ↑ C max by 195% | ↑ AUC by ~100%, ↑ C max by ~100% compared to voriconazole 200 mg twice a day alone | CONTRAINDICATED |
ANTI-PCP (Pneumocystis carinii pneumonia) | ||||||
Dapsone | 50 mg once a day | 300 mg once a day | HIV-infected patients (16) | ND | ↓ AUC by 27 – 40% | |
Sulfamethoxazole-Trimethoprim | 800/160 mg | 300 mg once a day | HIV-infected patients (12) | ↔ | ↓ AUC by 15–20% | |
ANTI-MAC (Mycobacterium avium intracellulare complex) | ||||||
Azithromycin | 500 mg once a day for 1 day, then 250 mg once a day for 9 days | 300 mg once a day | Healthy subjects (6) | ↔ | ↔ | |
Clarithromycin | 500 mg twice a day | 300 mg once a day | HIV-infected patients (12) | ↑ AUC by 75% | ↓ AUC by 50% | Monitor for rifabutin associated adverse events. Reduce dose or suspend use of rifabutin if toxicity is suspected. Alternative treatment for clarithromycin should be considered when treating patients receiving rifabutin |
ANTI-TB (Tuberculosis) | ||||||
Ethambutol | 1200 mg | 300 mg once a day for 7 days | Healthy subjects (10) | ND | ↔ | |
Isoniazid | 300 mg | 300 mg once a day for 7 days | Healthy subjects (6) | ND | ↔ | |
Bedaquiline | 400 mg daily on day 1 and day 29 | 300 mg daily | Healthy subjects (17) | ND | No change in bedaquiline pharmacokinetics. 1.4-fold ↑ in M2 and approximately 3.0-fold ↑ in M3 metabolites of bedaquiline. | Avoid bedaquiline co‑administration with rifabutin due to the adverse reactions associated with increased bedaquiline metabolite concentrations. |
OTHER | ||||||
Methadone | 20 – 100 mg once a day | 300 mg once a day for 13 days | HIV – infected patients (24) | ND | ↔ | |
Ethinylestradiol (EE)/ Norethindrone (NE) | 35 mg EE / 1 mg NE for 21 days | 300 mg once a day for 10 days | Healthy female subjects (22) | ND | EE: ↓ AUC by 35%, ↓ C max by 20% NE: ↓ AUC by 46% | Patients should be advised to use additional or alternative methods of contraception. |
Theophylline | 5 mg/kg | 300 mg for 14 days | Healthy subjects (11) | ND | ↔ | |
Other drugs
The structurally similar drug, rifampin, is known to reduce the plasma concentrations of a number of other drugs (see prescribing information for rifampin). Although a weaker enzyme inducer than rifampin, rifabutin may be expected to have some effect on those drugs as well.
DESCRIPTION
Rifabutin capsules for oral administration contain 150 mg of the rifamycin antimycobacterial agent rifabutin, USP, per capsule, along with the inactive ingredients, microcrystalline cellulose, magnesium stearate, red iron oxide, silica gel, sodium lauryl sulfate, titanium dioxide, and edible white ink.
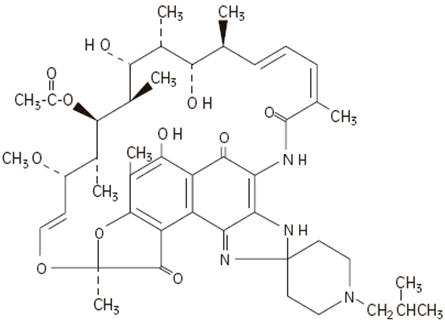
The chemical name for rifabutin is 1',4-didehydro-1-deoxy-1,4-dihydro-5'-(2-methylpropyl)-1-oxorifamycin XIV (Chemical Abstracts Service, 9th Collective Index) or (9 S ,12 E ,14 S ,15 R ,16 S ,17 R ,18 R ,19 R ,20 S ,21 S ,22 E ,24 Z )-6,16,18,20-tetrahydroxy-1'-isobutyl-14-methoxy-7,9,15,17,19,21,25-heptamethyl-spiro [9,4-(epoxypentadeca[1,11,13]trienimino)-2 H -furo[2',3':7,8]naphth[1,2-d] imidazole-2,4'-piperidine]-5,10,26-(3 H ,9 H )-trione-16-acetate. Rifabutin has a molecular formula of C 46 H 62 N 4 O 11 , a molecular weight of 847.02 and the following structure:
Rifabutin is a red-violet powder soluble in chloroform and methanol, sparingly soluble in ethanol, and very slightly soluble in water (0.19 mg/mL). Its log P value (the base 10 logarithm of the partition coefficient between n-octanol and water) is 3.2 (n-octanol/water).
CLINICAL PHARMACOLOGY
Pharmacokinetics
Absorption
Following a single oral dose of 300 mg to nine healthy adult volunteers, rifabutin was readily absorbed from the gastrointestinal tract with mean (±SD) peak plasma levels (C max ) of 375 (±267) ng/mL (range: 141 to 1033 ng/mL) attained in 3.3 (±0.9) hours (T max range: 2 to 4 hours). Absolute bioavailability assessed in five HIV -positive patients, who received both oral and intravenous doses, averaged 20%. Total recovery of radioactivity in the urine indicates that at least 53% of the orally administered rifabutin dose is absorbed from the gastrointestinal tract. The bioavailability of rifabutin from the capsule dosage form, relative to an oral solution, was 85% in 12 healthy adult volunteers. High-fat meals slow the rate without influencing the extent of absorption from the capsule dosage form. Plasma concentrations post-C max declined in an apparent biphasic manner. Pharmacokinetic dose-proportionality was established over the 300 mg to 600 mg dose range in nine healthy adult volunteers (crossover design) and in 16 early symptomatic human immunodeficiency virus (HIV)-positive patients over a 300 mg to 900 mg dose range.
Distribution
Due to its high lipophilicity, rifabutin demonstrates a high propensity for distribution and intracellular tissue uptake. Following intravenous dosing, estimates of apparent steady-state distribution volume (9.3 ± 1.5 L/kg) in five HIV-positive patients exceeded total body water by approximately 15-fold. Substantially higher intracellular tissue levels than those seen in plasma have been observed in both rat and man. The lung-to-plasma concentration ratio, obtained at 12 hours, was approximately 6.5 in four surgical patients who received an oral dose. Mean rifabutin steady-state trough levels (C p,min ss ; 24-hour post-dose) ranged from 50 to 65 ng/mL in HIV-positive patients and in healthy adult volunteers. About 85% of the drug is bound in a concentration-independent manner to plasma proteins over a concentration range of 0.05 to 1 µg/mL. Binding does not appear to be influenced by renal or hepatic dysfunction. Rifabutin was slowly eliminated from plasma in seven healthy adult volunteers, presumably because of distribution-limited elimination, with a mean terminal half-life of 45 (±17) hours (range: 16 to 69 hours). Although the systemic levels of rifabutin following multiple dosing decreased by 38%, its terminal half-life remained unchanged.
Metabolism
Of the five metabolites that have been identified, 25-O-desacetyl and 31-hydroxy are the most predominant, and show a plasma metabolite:parent area under the curve ratio of 0.10 and 0.07, respectively. The former has an activity equal to the parent drug and contributes up to 10% to the total antimicrobial activity.
Excretion
A mass-balance study in three healthy adult volunteers with 14 C-labeled rifabutin showed that 53% of the oral dose was excreted in the urine, primarily as metabolites. About 30% of the dose is excreted in the feces. Mean systemic clearance (CL s /F) in healthy adult volunteers following a single oral dose was 0.69 (±0.32) L/hr/kg (range: 0.46 to 1.34 L/hr/kg). Renal and biliary clearance of unchanged drug each contribute approximately 5% to CL s /F.
Pharmacokinetics in Special Populations
Geriatric
Compared to healthy volunteers, steady-state pharmacokinetics of rifabutin are more variable in elderly patients (>70 years).
Pediatric
The pharmacokinetics of rifabutin have not been studied in subjects under 18 years of age.
Renal Impairment
The disposition of rifabutin (300 mg) was studied in 18 patients with varying degrees of renal function. Area under plasma concentration time curve (AUC) increased by about 71% in patients with severe renal impairment (creatinine clearance below 30 mL/min) compared to patients with creatinine clearance (Cr cl ) between 61–74 mL/min. In patients with mild to moderate renal impairment (Cr cl between 30–61 mL/min), the AUC increased by about 41%. In patients with severe renal impairment, carefully monitor for rifabutin associated adverse events. A reduction in the dosage of rifabutin is recommended for patients with Cr cl <30 mL/min if toxicity is suspected (see DOSAGE AND ADMINISTRATION ).
Hepatic Impairment
Mild hepatic impairment does not require a dose modification. The pharmacokinetics of rifabutin in patients with moderate and severe hepatic impairment is not known.
Malabsorption in HIV-Infected Patients
Alterations in gastric pH due to progressing HIV disease has been linked with malabsorption of some drugs used in HIV-positive patients (e.g., rifampin, isoniazid). Drug serum concentrations data from AIDS patients with varying disease severity (based on CD4+ counts) suggests that rifabutin absorption is not influenced by progressing HIV disease.
Drug-Drug Interactions
(see also PRECAUTIONS-Drug Interactions )
Multiple dosing of rifabutin has been associated with induction of hepatic metabolic enzymes of the CYP3A subfamily. Rifabutin's predominant metabolite (25-desacetyl rifabutin: LM565), may also contribute to this effect. Metabolic induction due to rifabutin is likely to produce a decrease in plasma concentrations of concomitantly administered drugs that are primarily metabolized by the CYP3A enzymes. Similarly concomitant medications that competitively inhibit the CYP3A activity may increase plasma concentrations of rifabutin.
CLINICAL STUDIES
Two randomized, double-blind clinical trials (Study 023 and Study 027) compared rifabutin (300 mg/day) to placebo in patients with CDC-defined AIDS and CD4 counts ≤200 cells/µL. These studies accrued patients from 2/90 through 2/92. Study 023 enrolled 590 patients, with a median CD4 cell count at study entry of 42 cells/µL (mean 61). Study 027 enrolled 556 patients with a median CD4 cell count at study entry of 40 cells/µL (mean 58).
Endpoints included the following:
- MAC bacteremia, defined as at least one blood culture positive for Mycobacterium avium complex (MAC) bacteria.
- Clinically significant disseminated MAC disease, defined as MAC bacteremia accompanied by signs or symptoms of serious MAC infection, including one or more of the following: fever, night sweats, rigors, weight loss, worsening anemia, and/or elevations in alkaline phosphatase.
- Survival.
MAC Bacteremia
Participants who received rifabutin were one-third to one-half as likely to develop MAC bacteremia as were participants who received placebo. These results were statistically significant (Study 023: p<0.001; Study 027: p = 0.002).
In Study 023, the one-year cumulative incidence of MAC bacteremia, on an intent to treat basis, was 9% for patients randomized to rifabutin and 22% for patients randomized to placebo. In Study 027, these rates were 13% and 28% for patients receiving rifabutin and placebo, respectively.
Most cases of MAC bacteremia (approximately 90% in these studies) occurred among participants whose CD4 count at study entry was ≤100 cells/µL. The median and mean CD4 counts at onset of MAC bacteremia were 13 cells/µL and 24 cells/µL, respectively. These studies did not investigate the optimal time to begin MAC prophylaxis.
Clinically Significant Disseminated MAC Disease
In association with the decreased incidence of bacteremia, patients on rifabutin showed reductions in the signs and symptoms of disseminated MAC disease, including fever, night sweats, weight loss, fatigue, abdominal pain, anemia, and hepatic dysfunction.
Survival
The one-year survival rates in Study 023 were 77% for the group receiving rifabutin and 77% for the placebo group. In Study 027, the one-year survival rates were 77% for the group receiving rifabutin and 70% for the placebo group.
These differences were not statistically significant.
HOW SUPPLIED
Rifabutin capsules, USP are supplied as hard gelatin capsules having an opaque red-brown cap and body, imprinted with MYCOBUTIN/PHARMACIA & UPJOHN in white ink, each containing 150 mg of rifabutin, USP.
Rifabutin is available as follows:
NDC 59762-1350-1 Bottles of 100 capsules
Keep tightly closed and dispense in a tight container as defined in the USP. Store at 25°C (77°F); excursions permitted to 15°–30°C (59°–86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Rifabutin inhibits DNA-dependent RNA polymerase in susceptible strains of Escherichia coli and Bacillus subtilis but not in mammalian cells. In resistant strains of E. coli , rifabutin, like rifampin, did not inhibit this enzyme. It is not known whether rifabutin inhibits DNA-dependent RNA polymerase in Mycobacterium avium or in M. intracellulare which comprise M. avium complex (MAC).