Get your patient on Rifampin - Rifampin injection, Powder, Lyophilized, For Solution (Rifampin)
Rifampin - Rifampin injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE
In the treatment of both tuberculosis and the meningococcal carrier state, the small number of resistant cells present within large populations of susceptible cells can rapidly become the predominant type. Bacteriologic cultures should be obtained before the start of therapy to confirm the susceptibility of the organism to rifampin and they should be repeated throughout therapy to monitor the response to treatment. Since resistance can emerge rapidly, susceptibility tests should be performed in the event of persistent positive cultures during the course of treatment. If test results show resistance to rifampin and the patient is not responding to therapy, the drug regimen should be modified.
Tuberculosis
Rifampin is indicated in the treatment of all forms of tuberculosis.
A three-drug regimen consisting of rifampin, isoniazid, and pyrazinamide is recommended in the initial phase of short-course therapy which is usually continued for 2 months. The Advisory Council for the Elimination of Tuberculosis, the American Thoracic Society, and Centers for Disease Control and Prevention recommend that either streptomycin or ethambutol be added as a fourth drug in a regimen containing isoniazid (INH), rifampin, and pyrazinamide for initial treatment of tuberculosis unless the likelihood of INH resistance is very low. The need for a fourth drug should be reassessed when the results of susceptibility testing are known. If community rates of INH resistance are currently less than 4%, an initial treatment regimen with less than four drugs may be considered.
Following the initial phase, treatment should be continued with rifampin and isoniazid for at least 4 months. Treatment should be continued for longer if the patient is still sputum or culture positive, if resistant organisms are present, or if the patient is HIV positive.
Rifampin for injection is indicated for the initial treatment and retreatment of tuberculosis when the drug cannot be taken by mouth.
Meningococcal Carriers
Rifampin is indicated for the treatment of asymptomatic carriers of Neisseria meningitidis to eliminate meningococci from the nasopharynx. Rifampin is not indicated for the treatment of meningococcal infection because of the possibility of the rapid emergence of resistant organisms. (See WARNINGS .)
Rifampin should not be used indiscriminately, and, therefore, diagnostic laboratory procedures, including serotyping and susceptibility testing, should be performed for establishment of the carrier state and the correct treatment. So that the usefulness of rifampin in the treatment of asymptomatic meningococcal carriers is preserved, the drug should be used only when the risk of meningococcal disease is high.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of rifampin and other antibacterial drugs, rifampin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
Rifampin can be administered by IV infusion (see INDICATIONS AND USAGE ).
See CLINICAL PHARMACOLOGY for dosing information in patients with renal failure.
Tuberculosis
Adults: 10 mg/kg, in a single daily administration, not to exceed 600 mg/day, IV
Pediatric Patients: 10 mg/kg to 20 mg/kg, not to exceed 600 mg/day, IV
Rifampin is indicated in the treatment of all forms of tuberculosis. A three-drug regimen consisting of rifampin, isoniazid, and pyrazinamide is recommended in the initial phase of short-course therapy which is usually continued for 2 months. The Advisory Council for the Elimination of Tuberculosis, the American Thoracic Society, and the Centers for Disease Control and Prevention recommend that either streptomycin or ethambutol be added as a fourth drug in a regimen containing isoniazid (INH), rifampin, and pyrazinamide for initial treatment of tuberculosis unless the likelihood of INH resistance is very low. The need for a fourth drug should be reassessed when the results of susceptibility testing are known. If community rates of INH resistance are currently less than 4%, an initial treatment regimen with less than four drugs may be considered.
Following the initial phase, treatment should be continued with rifampin and isoniazid for at least 4 months. Treatment should be continued for longer if the patient is still sputum or culture positive, if resistant organisms are present, or if the patient is HIV positive.
Preparation of Solution for IV Infusion
Reconstitute the lyophilized powder by transferring 10 mL of sterile water for injection to a vial containing 600 mg of rifampin for injection. Swirl vial gently to completely dissolve the antibiotic. The reconstituted solution contains 60 mg rifampin per mL and is stable at room temperature for up to 30 hours. Prior to administration, withdraw from the reconstituted solution a volume equivalent to the amount of rifampin calculated to be administered and add to 500 mL of infusion medium. Mix well and infuse at a rate allowing for complete infusion within 3 hours. Alternatively, the amount of rifampin calculated to be administered may be added to 100 mL of infusion medium and infused in 30 minutes.
Dilutions in dextrose 5% for injection (D5W) are stable at room temperature for up to 8 hours and should be prepared and used within this time. Precipitation of rifampin from the infusion solution may occur beyond this time. Dilutions in normal saline are stable at room temperature for up to 6 hours and should be prepared and used within this time. Other infusion solutions are not recommended.
Incompatibilities
Physical incompatibility (precipitate) was observed with undiluted (5 mg/mL) and diluted (1 mg/mL in normal saline) diltiazem hydrochloride and rifampin (6 mg/mL in normal saline) during simulated Y-site administration.
Meningococcal Carriers
Adults: For adults, it is recommended that 600 mg rifampin be administered twice daily for two days.
Pediatric Patients: Pediatric patients 1 month of age or older:
10 mg/kg (not to exceed 600 mg per dose) every 12 hours for two days.
Pediatric patients under 1 month of age: 5 mg/kg every 12 hours for two days.
CONTRAINDICATIONS
Rifampin is contraindicated in patients with a history of hypersensitivity to rifampin or any of the components, or to any of the rifamycins. (See WARNINGS .)
Rifampin is contraindicated in patients who are also receiving ritonavir-boosted saquinavir due to an increased risk of severe hepatocellular toxicity. (See PRECAUTIONS, Drug Interactions .)
Rifampin is contraindicated in patients who are also receiving atazanavir, darunavir, fosamprenavir, saquinavir, or tipranavir due to the potential of rifampin to substantially decrease plasma concentrations of these antiviral drugs, which may result in loss of antiviral efficacy and/or development of viral resistance.
Rifampin is contraindicated in patients receiving praziquantel since therapeutically effective blood levels of praziquantel may not be achieved. In patients receiving rifampin who need immediate treatment with praziquantel alternative agents should be considered. However, if treatment with praziquantel is necessary, rifampin should be discontinued 4 weeks before administration of praziquantel. Treatment with rifampin can then be restarted one day after completion of praziquantel treatment.
Rifampin is contraindicated in patients receiving lurasidone. Concomitant use of lurasidone with strong CYP3A4 inducers (e.g., rifampin) decreased the exposure of lurasidone compared to the use of lurasidone alone. (See PRECAUTIONS, Drug Interactions ).
ADVERSE REACTIONS
The following adverse reactions associated with the use of rifampin were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal
Heartburn, epigastric distress, anorexia, nausea, vomiting, jaundice, flatulence, cramps, and diarrhea have been noted in some patients. Although Clostridium difficile has been shown in vitro to be sensitive to rifampin, pseudomembranous colitis has been reported with the use of rifampin (and other broad-spectrum antibiotics). Therefore, it is important to consider this diagnosis in patients who develop diarrhea in association with antibiotic use. Tooth discoloration (which may be permanent) may occur.
Hepatic
Hepatotoxicity including transient abnormalities in liver function tests (e.g., elevations in serum bilirubin, alkaline phosphatase, serum transaminases, gamma-glutamyl transferase), hepatitis, shock-like syndrome with hepatic involvement and abnormal liver function tests, and cholestasis have been reported (see WARNINGS ).
Hematologic
Cases of thrombotic microangiopathy, including thrombotic thrombocytopenic purpura and hemolytic uremic syndrome, have been reported (see WARNINGS ).
Thrombocytopenia has occurred primarily with high dose intermittent therapy but has also been noted after resumption of interrupted treatment. It rarely occurs during well-supervised daily therapy. This effect is reversible if the drug is discontinued as soon as purpura occurs. Cerebral hemorrhage and fatalities have been reported when rifampin administration has been continued or resumed after the appearance of purpura.
Rare reports of disseminated intravascular coagulation have been observed.
Leukopenia, hemolytic anemia, decreased hemoglobin, bleeding, and vitamin K–dependent coagulation disorders (abnormal prolongation of prothrombin time or low vitamin K–dependent coagulation factors) have been observed.
Agranulocytosis has been reported very rarely.
Central Nervous System
Headache, fever, drowsiness, fatigue, ataxia, dizziness, inability to concentrate, mental confusion, behavioral changes, muscular weakness, pains in extremities, and generalized numbness have been observed.
Psychoses have been rarely reported.
Rare reports of myopathy have also been observed.
Ocular
Visual disturbances have been observed.
Endocrine
Menstrual disturbances have been observed.
Rare reports of adrenal insufficiency in patients with compromised adrenal function have been observed.
Renal
Elevations in BUN and serum uric acid have been reported. Rarely, hemolysis, hemoglobinuria, hematuria, interstitial nephritis, acute tubular necrosis, renal insufficiency, and acute renal failure have been noted. These are generally considered to be hypersensitivity reactions. They usually occur during intermittent therapy or when treatment is resumed following intentional or accidental interruption of a daily dosage regimen, and are reversible when rifampin is discontinued and appropriate therapy instituted.
Dermatologic
Cutaneous reactions are mild and self-limiting and do not appear to be hypersensitivity reactions. Typically, they consist of flushing and itching with or without a rash. More serious cutaneous reactions which may be due to hypersensitivity occur but are uncommon.
Hypersensitivity Reactions
Occasionally, pruritus, urticaria, rash, pemphigoid reaction, erythema multiforme, acute generalized exanthematous pustulosis, Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms syndrome (see WARNINGS ), vasculitis, eosinophilia, sore mouth, sore tongue, and conjunctivitis have been observed.
Anaphylaxis has been reported rarely.
Respiratory, Thoracic and Mediastinal Disorders
Pulmonary toxicity (including, but not limited to, interstitial lung disease, pneumonitis, hypersensitivity pneumonitis, eosinophilic pneumonia, pulmonary infiltrates, organizing pneumonia, respiratory failure, pulmonary fibrosis, and acute respiratory distress syndrome) has been observed (see WARNINGS ).
Miscellaneous
Paradoxical drug reaction has been reported with rifampin (see WARNINGS ). Edema of the face and extremities has been reported. Other reactions which have occurred with intermittent dosage regimens include "flu syndrome" (such as episodes of fever, chills, headache, dizziness, and bone pain), shortness of breath, wheezing, decrease in blood pressure and shock. The "flu syndrome" may also appear if rifampin is taken irregularly by the patient or if daily administration is resumed after a drug-free interval.
Drug Interactions
Pharmacodynamic Interactions
Healthy subjects who received rifampin 600 mg once daily concomitantly with saquinavir 1000 mg/ritonavir 100 mg twice daily (ritonavir-boosted saquinavir) developed severe hepatocellular toxicity. Therefore, concomitant use of these medications is contraindicated. (See CONTRAINDICATIONS .)
When rifampin is given concomitantly with other hepatotoxic medications such as halothane or isoniazid, the potential for hepatotoxicity is increased. The concomitant use of rifampin and halothane should be avoided. Patients receiving both rifampin and isoniazid should be monitored closely for hepatotoxicity.
Effect of Rifampin on Other Drugs
Induction of Drug Metabolizing Enzymes and Transporters
Drug metabolizing enzymes and transporters affected by rifampin include cytochromes P450 (CYP) 1A2, 2B6, 2C8, 2C9, 2C19, and 3A4, UDP-glucuronyltransferases (UGT), sulfotransferases, carboxylesterases, and transporters including P-glycoprotein (P-gp) and multidrug resistance-associated protein 2 (MRP2). Most drugs are substrates for one or more of these enzyme or transporter pathways and these pathways may be induced by rifampin simultaneously. Therefore, rifampin may increase the metabolism and decrease the activity of certain coadministered drugs or increase the activity of a coadministered pro-drug (where metabolic activation is required), and has the potential to perpetuate clinically important drug-drug interactions against many drugs and across many drug classes (Table 1).
Table 1 summarizes the effect of rifampin on other drugs or drug classes. Adjust dosages of concomitant drugs based on approved drug labeling and if applicable, therapeutic drug monitoring, unless otherwise specified.
| AUC = area under the time-concentration curve | ||
Drug or Drug Class and Prevention or Management | Clinical Effect | |
Antiretrovirals Prevention or Management : Concomitant use is contraindicated (see CONTRAINDICATIONS ) | ||
Atazanavir | Decrease AUC by 72% | |
Darunavir Rifampin dosage used concomitantly with the drug(s) is not specified in the proposed package insert. | Substantial decrease in exposure, which may result in loss of therapeutic effect and development of resistance. | |
Tipranavir | ||
Fosamprenavir Administered with rifampin 300 mg daily | Decrease AUC by 82% | |
Saquinavir | Decrease AUC by 70% Coadministration may result in severe hepatocellular toxicity. | |
Antiretrovirals Prevention or Management : Avoid concomitant use | ||
Zidovudine | Decrease AUC by 47% | |
Indinavir | Decrease AUC by 92% | |
Efavirenz | Decrease AUC by 26% | |
Cortisol Receptor Blocker | ||
Mifepristone Prevention or Management: Avoid concomitant use | Decrease exposure | |
Hepatitis C Antiviral Prevention or Management : Avoid concomitant use | ||
Daclatasvir | Decrease AUC by 79% | |
Simeprevir | Decrease AUC by 48% | |
Sofosbuvir | Decrease AUC by 72% Coadministration of sofosbuvir with rifampin may decrease sofosbuvir plasma concentrations, leading to reduced therapeutic effect of sofosbuvir. | |
Telaprevir | Decrease AUC by 92% | |
Systemic Hormonal Contraceptives Prevention or Management : Advise patients to change to non-hormonal methods of birth control during rifampin therapy | ||
Estrogens | Decrease exposure | |
Progestins | ||
Anticonvulsants | ||
Phenytoin Administered with rifampin 450 mg daily | Decrease exposure | |
Antiarrhythmics | ||
Disopyramide | Decrease exposure | |
Mexiletine | Decrease exposure | |
Quinidine | Decrease exposure | |
Propafenone | Decrease AUC by 50% to 67% | |
Tocainide | Decrease exposure | |
Antiestrogens | ||
Tamoxifen | Decrease AUC by 86% | |
Toremifene | Decrease steady state concentrations of toremifene in serum | |
Antithrombotic Agents | ||
Clopidogrel Prevention or Management : Concomitant use of clopidogrel and rifampin should be discouraged | Increase active metabolite exposure and risk of bleeding | |
Ticagrelor Prevention or Management : Avoid use | Decrease exposure | |
Antipsychotics | ||
Haloperidol | Decrease plasma concentrations by 70% | |
Lurasidone Prevention or Management : Concomitant use is contraindicated (See CONTRAINDICATIONS ) | Decrease exposure | |
Oral Anticoagulants Prevention or Management : Perform prothrombin time daily or as frequently as necessary to establish and maintain the required dose of anticoagulant | ||
Warfarin | Decrease exposure | |
Antifungals | ||
Fluconazole | Decrease AUC by 23% | |
Itraconazole Prevention or Management : Not recommended 2 weeks before and during itraconazole treatment | Decrease exposure | |
Ketoconazole | Decrease exposure | |
Caspofungin Prevention or Management: Refer to the caspofungin prescribing information for caspofungin dose adjustment. | Decrease exposure | |
Beta-blockers | ||
Metoprolol | Decrease exposure | |
Propranolol | Decrease exposure | |
Benzodiazepines | ||
Diazepam, Administered with rifampin 1200 mg daily | Decrease exposure | |
Benzodiazepine-related drugs | ||
Zopiclone | Decrease AUC by 82% | |
Zolpidem | Decrease AUC by 73% | |
Calcium Channel Blockers | ||
Diltiazem | Decrease exposure | |
Nifedipine Rifampin 1200 mg administered as a single oral dose 8 hours before administering a single oral dose of nifedipine 10 mg | Decrease exposure | |
Verapamil | Decrease exposure | |
Corticosteroids Numerous cases in the literature describe a decrease in glucocorticoid effect when used concomitantly with rifampin. The literature contains reports of acute adrenal crisis or adrenal insufficiency induced by the combination of rifampin-isoniazid-ethambutol or rifampin-isoniazid in patients with Addison’s disease. | ||
Prednisolone | Decrease exposure | |
Cardiac Glycosides | ||
Digoxin Prevention or Management : Measure serum digoxin concentrations before initiating rifampin. Continue monitoring and increase digoxin dose by approximately 20% to 40% as necessary. | Decrease exposure | |
Digitoxin | Decrease exposure | |
Fluoroquinolones | ||
Pefloxacin Administered with rifampin 900 mg daily | Decrease exposure | |
Moxifloxacin, | Decrease exposure | |
Oral Hypoglycemic Agents (e.g., sulfonylureas) | ||
Glyburide | Decrease exposure Rifampin may worsen glucose control of glyburide. | |
Glipizide | Decrease exposure | |
Immunosuppressive Agents | ||
Cyclosporine | Decrease exposure | |
Tacrolimus Prevention or Management : Monitoring of whole blood concentrations and appropriate dosage adjustments of tacrolimus are recommended when rifampin and tacrolimus are used concomitantly. | Decrease AUC by 56% | |
Narcotic Analgesics | ||
Oxycodone | Decrease AUC by 86% | |
Morphine | Decrease exposure | |
Progestin Antagonist | ||
Mifepristone Prevention or Management: Refer to the post-treatment assessment in the mifepristone prescribing information to verify that treatment has been successful. | Decrease exposure | |
Selective 5-HT 3 Receptor Antagonists | ||
Ondansetron | Decrease exposure | |
Statins Metabolized by CYP3A4 | ||
Simvastatin | Decrease exposure | |
Thiazolidinediones | ||
Rosiglitazone | Decrease AUC by 66% | |
Tricyclic Antidepressants | ||
Nortriptyline A tuberculosis treatment regimen including rifampin (600 mg/day), isoniazid (300 mg/day), pyrazinamide (500 mg 3× per day), and pyridoxine (25 mg) was associated with higher than expected doses of nortriptyline were required to obtain a therapeutic drug level. Following the discontinuation of rifampin, the patient became drowsy and the serum nortriptyline levels rose precipitously (3-fold) into the toxic range. | Decrease exposure | |
Other Drugs | ||
Enalapril | Decrease active metabolite exposure | |
Chloramphenicol Concomitant use with rifampin in 2 children | Decrease exposure | |
Clarithromycin | Decrease exposure | |
Dapsone | Rifampin has been shown to increase the clearance of dapsone and, accordingly, decrease dapsone exposure. Rifampin has also been shown to increase the production of the hydroxylamine metabolite of dapsone which could increase the risk of methemoglobinemia. | |
Doxycycline Administered with rifampin (10 mg/kg daily) | Decrease exposure | |
Irinotecan Administered with an antibiotic regimen including rifampin (450 mg/day), isoniazid (300 mg/day), and streptomycin (0.5 g/day) IM Prevention or Management : Avoid the use of rifampin, a strong CYP3A4 inducer, if possible. Substitute non-enzyme inducing therapies at least 2 weeks prior to initiation of irinotecan therapy | Decrease irinotecan and active metabolite exposure | |
Levothyroxine | Decrease exposure | |
Losartan | Parent | Decrease AUC by 30% |
Active metabolite (E3174) | Decrease AUC by 40%. | |
Methadone | In patients well-stabilized on methadone, concomitant administration of rifampin resulted in a marked reduction in serum methadone levels and a concurrent appearance of withdrawal symptoms. | |
Praziquantel Prevention or Management : Concomitant use is contraindicated (see CONTRAINDICATIONS ) | Decrease plasma praziquantel concentrations to undetectable levels. | |
Quinine Prevention or Management : Avoid concomitant use | Decrease AUC by 75% to 85% | |
Telithromycin | Decrease AUC by 86% | |
Theophylline | Decrease exposure by 20% to 40% | |
Effect of Other Drugs on Rifampin
Concomitant antacid administration may reduce the absorption of rifampin. Daily doses of rifampin should be given at least 1 hour before the ingestion of antacids.
Concomitant use with probenecid and cotrimoxazole increases the concentration of rifampin which may increase the risk of rifampin toxicities. Monitor for adverse reactions associated with rifampin during coadministration.
Other Interactions
Atovaquone: Concomitant use of rifampin with atovaquone decrease concentrations of atovaquone and increase concentrations of rifampin which may increase the risk of rifampin toxicities. Coadministration of rifampin with atovaquone is not recommended.
DESCRIPTION
Rifampin for injection, USP is a red colored lyophilized powder or cake that contains rifampin USP 600 mg, sodium formaldehyde sulfoxylate 10 mg, and sodium hydroxide to adjust pH.
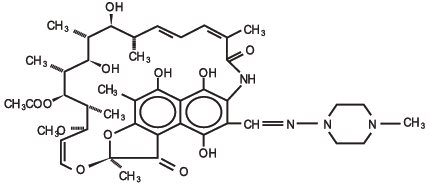
Rifampin is a semisynthetic antibiotic derivative of rifamycin SV. Rifampin USP is a red to brown crystalline powder very slightly soluble in water at neutral pH, freely soluble in chloroform, soluble in ethyl acetate and in methanol. Its molecular weight is 822.95 and its chemical formula is C 43 H 58 N 4 O 12 . The chemical name for rifampin is either:
3-[[(4-Methyl-1-piperazinyl)imino]methyl]rifamycin
or
5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,18,20,22–heptamethyl-8-[N-(4-methyl-1-piperazinyl)formimidoyl]-2,7-(epoxypentadeca[1,11,13]trienimino)naphtho[2,1- b ]furan-1,11(2H)-dione 21-acetate.
Its structural formula is:
CLINICAL PHARMACOLOGY
Oral Administration
Rifampin is readily absorbed from the gastrointestinal tract. Peak serum concentrations in healthy adults and pediatric populations vary widely from individual to individual. Following a single 600 mg oral dose of rifampin in healthy adults, the peak serum concentration averages 7 mcg/mL but may vary from 4 to 32 mcg/mL. Absorption of rifampin is reduced by about 30% when the drug is ingested with food.
Rifampin is widely distributed throughout the body. It is present in effective concentrations in many organs and body fluids, including cerebrospinal fluid. Rifampin is about 80% protein bound. Most of the unbound fraction is not ionized and, therefore, diffuses freely into tissues.
In healthy adults, the mean biological half-life of rifampin in serum averages 3.35±0.66 hours after a 600 mg oral dose, with increases up to 5.08±2.45 hours reported after a 900 mg dose. With repeated administration, the half-life decreases and reaches average values of approximately 2 to 3 hours. The half-life does not differ in patients with renal failure at doses not exceeding 600 mg daily, and, consequently, no dosage adjustment is required. The half-life of rifampin at a dose of 720 mg daily has not been established in patients with renal failure. Following a single 900 mg oral dose of rifampin in patients with varying degrees of renal insufficiency, the mean half-life increased from 3.6 hours in healthy adults to 5.0, 7.3, and 11.0 hours in patients with glomerular filtration rates of 30 to 50 mL/min, less than 30 mL/min, and in anuric patients, respectively. Refer to the WARNINGS section for information regarding patients with hepatic insufficiency.
After absorption, rifampin is rapidly eliminated in the bile, and an enterohepatic circulation ensues. During this process, rifampin undergoes progressive deacetylation so that nearly all the drug in the bile is in this form in about 6 hours. This metabolite has antibacterial activity. Intestinal reabsorption is reduced by deacetylation, and elimination is facilitated. Up to 30% of a dose is excreted in the urine, with about half of this being unchanged drug.
Intravenous Administration
After intravenous administration of a 300 or 600 mg dose of rifampin infused over 30 minutes to healthy male volunteers (n=12), mean peak plasma concentrations were 9.0±3.0 and 17.5±5.0 mcg/mL, respectively. Total body clearances after the 300 and 600 mg IV doses were 0.19±0.06 and 0.14±0.03 L/hr/kg, respectively. Volumes of distribution at steady state were 0.66±0.14 and 0.64±0.11 L/kg for the 300 and 600 mg IV doses, respectively. After intravenous administration of 300 or 600 mg doses, rifampin plasma concentrations in these volunteers remained detectable for 8 and 12 hours, respectively (see Table).
Plasma Concentrations (mean ± standard deviation, mcg/mL) | ||||||
Rifampin Dosage IV | 30 min | 1 hr | 2 hr | 4 hr | 8 hr | 12 hr |
300 mg | 8.9±2.9 | 4.9±1.3 | 4.0±1.3 | 2.5±1.0 | 1.1±0.6 | <0.4 |
600 mg | 17.4±5.1 | 11.7±2.8 | 9.4±2.3 | 6.4±1.7 | 3.5±1.4 | 1.2±0.6 |
Plasma concentrations after the 600 mg dose, which were disproportionately higher (up to 30% greater than expected) than those found after the 300 mg dose, indicated that the elimination of larger doses was not as rapid.
After repeated once-a-day infusions (3-hr duration) of 600 mg in patients (n=5) for 7 days, concentrations of IV rifampin decreased from 5.81±3.38 mcg/mL 8 hours after the infusion on day 1 to 2.6±1.88 mcg/mL 8 hours after the infusion on day 7.
Rifampin is widely distributed throughout the body. It is present in effective concentrations in many organs and body fluids, including cerebrospinal fluid. Rifampin is about 80% protein bound. Most of the unbound fraction is not ionized and therefore diffuses freely into tissues.
Rifampin is rapidly eliminated in the bile and undergoes progressive enterohepatic circulation and deacetylation to the primary metabolite, 25-desacetyl-rifampin. This metabolite is microbiologically active. Less than 30% of the dose is excreted in the urine as rifampin or metabolites. Serum concentrations do not differ in patients with renal failure at a studied dose of 300 mg, and consequently, no dosage adjustment is required.
Pediatrics
Oral Administration
In one study, pediatric patients 6 to 58 months old were given rifampin suspended in simple syrup or as dry powder mixed with applesauce at a dose of 10 mg/kg body weight. Peak serum concentrations of 10.7±3.7 and 11.5±5.1 mcg/mL were obtained 1 hour after preprandial ingestion of the drug suspension and the applesauce mixture, respectively. After the administration of either preparation, the t ½ of rifampin averaged 2.9 hours. It should be noted that in other studies in pediatric populations, at doses of 10 mg/kg body weight, mean peak serum concentrations of 3.5 mcg/mL to 15 mcg/mL have been reported.
Intravenous Administration
In pediatric patients 0.25 to 12.8 years old (n=12), the mean peak serum concentration of rifampin at the end of a 30-minute infusion of approximately 300 mg/m 2 was 25.9±1.3 mcg/mL; individual peak concentrations 1 to 4 days after initiation of therapy ranged from 11.7 to 41.5 mcg/mL; individual peak concentrations 5 to 14 days after initiation of therapy were 13.6 to 37.4 mcg/mL. The individual serum half-life of rifampin changed from 1.04 to 3.81 hours early in therapy to 1.17 to 3.19 hours 5 to 14 days after therapy was initiated.
In a pharmacokinetics (PK) study of 22 pediatric patients (median gestational age 27.5 weeks [range; 23 to 41 weeks] and median postnatal age (PNA) 7.5 days [range: 0 to 56 days]) receiving intravenous rifampin 10 to 20 mg/kg/day over 30 minutes, pediatric patients ≤7 days old had a median clearance of 0.09 L/kg/hr (range: 0.056 to 0.14 L/kg/hr) and a median area under the plasma drug concentration-time curve at infinity (AUC ∞ ) of 105 mg•hr/L (range: 80172 mg•hr/L); pediatric patients >7 days PNA had a median clearance of 0.23 L/kg/hr (range: 0.14 to 0.32 L/kg/hr) and a median AUC ∞ of 78 mg•hr/L (range: 51 to 170 mg•hr/L).
Based on PK modeling and simulation analyses, a dose of 12 mg/kg infused over 60 minutes once daily in pediatric patients ≤7 days PNA resulted in median (90% prediction interval) for maximum plasma concentration (C max ) of 8.4 mcg/mL (range: 5.3 to 13.3 mcg/mL) and AUC ∞ of 96.4 mg•hr/L (range: 64.5 to 141.6 mg•hr/L); in pediatric patients >7 days and < 90 days PNA a dose of 14 mg/kg infused over 60 minutes once daily resulted in a median (90% prediction interval) for C max of 9.6 mcg/mL (range: 6.l to 15.1 mcg/mL) and AUC ∞ of 73.l mg•hr/L (range: 45.6 to 131.6 mg•hr/L) . All simulated AUC ∞ were within the range of AUC ∞ values reported in other studies in adults treated for tuberculosis.
Microbiology
Mechanism of Action
Rifampin inhibits DNA-dependent RNA polymerase activity in susceptible Mycobacterium tuberculosis organisms. Specifically, it interacts with bacterial RNA polymerase but does not inhibit the mammalian enzyme.
Resistance
Organisms resistant to rifampin are likely to be resistant to other rifamycins.
In the treatment of both tuberculosis and the meningococcal carrier state (see INDICATIONS AND USAGE ), the small number of resistant cells present within large populations of susceptible cells can rapidly become predominant. In addition, resistance to rifampin has been determined to occur as single-step mutations of the DNA-dependent RNA polymerase. Since resistance can emerge rapidly, appropriate susceptibility tests should be performed in the event of persistent positive cultures.
Activity in vitro and in vivo
Rifampin has bactericidal activity in vitro against slow and intermittently growing M. tuberculosis organisms.
Rifampin has been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections (see INDICATIONS AND USAGE ):
Aerobic Gram-Negative Microorganisms: Neisseria meningitidis
“Other” Microorganisms: Mycobacterium tuberculosis
The following in vitro data are available, but their clinical significance is unknown . At least 90% of the following bacteria exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for rifampin against isolates of similar genus or organism group. However, the efficacy of rifampin in treating clinical infections caused by these bacteria has not been established in adequate and well-controlled clinical trials.
Aerobic Gram-Positive Microorganisms: Staphylococcus aureus (including Methicillin-Resistant S. aureus /MRSA) Staphylococcus epidermidis
Aerobic Gram-Negative Microorganisms: Haemophilus influenzae
“Other” Microorganisms: Mycobacterium leprae
ß-lactamase production should have no effect on rifampin activity.
Susceptibility Testing
For specific information regarding susceptibility test criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: www.fda.gov/STIC.
HOW SUPPLIED
Rifampin for injection, USP is available in sterile glass vials containing 600 mg rifampin (NDC 67457-445-60).
Storage: Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.] Avoid excessive heat (temperatures above 40°C or 104°F). Protect from light.
Manufactured for: Mylan Institutional LLC Morgantown, WV 26505 U.S.A.
Manufactured by: OneSource Specialty Pharma Limited (Sterile Product Division) Bangalore, India
50105007 1200012531
MAY 2025
Mechanism of Action
Rifampin inhibits DNA-dependent RNA polymerase activity in susceptible Mycobacterium tuberculosis organisms. Specifically, it interacts with bacterial RNA polymerase but does not inhibit the mammalian enzyme.