Get your patient on Sirturo - Bedaquiline Fumarate tablet (Bedaquiline Fumarate)
Sirturo - Bedaquiline Fumarate tablet prescribing information
WARNING: QTc PROLONGATION
QTc prolongation can occur with SIRTURO. Use with drugs that prolong the QTc interval may cause additive QTc prolongation. Monitor ECGs. Discontinue SIRTURO if significant ventricular arrhythmia or QTc interval greater than 500 ms develops [see Warnings and Precautions (5.1) ] .
INDICATIONS AND USAGE
SIRTURO is a diarylquinoline antimycobacterial drug indicated as part of combination therapy in the treatment of adult and pediatric patients (2 years and older and weighing at least 8 kg) with pulmonary tuberculosis (TB) due to Mycobacterium tuberculosis resistant to at least rifampin and isoniazid.
Limitations of Use
- Do not use SIRTURO for the treatment of:
- Latent infection due to Mycobacterium tuberculosis ( M. tuberculosis )
- Drug-sensitive pulmonary TB
- Extra-pulmonary TB
- Infections caused by non-tuberculous mycobacteria
DOSAGE AND ADMINISTRATION
Important Administration Instructions
- Administer SIRTURO by directly observed therapy (DOT).
- Only use SIRTURO in combination with at least three other drugs to which the patient's TB isolate has been shown to be susceptible in vitro. If in vitro testing results are unavailable, SIRTURO treatment may be initiated in combination with at least four other drugs to which the patient's TB isolate is likely to be susceptible. Refer to the prescribing information of the drugs used in combination with SIRTURO for further information.
- SIRTURO (20 mg and 100 mg) must be taken with food.
- SIRTURO 20 mg are functionally scored tablets which can be split at the scored lines into two equal halves of 10 mg each to provide doses less than 20 mg [see Dosage and Administration (2.6) ].
- As an alternative method of administration, SIRTURO 20 mg tablets can be dispersed in water and administered or dispersed in water and further mixed with a beverage or soft food, or crushed and mixed with soft food, or administered through a feeding tube [see Dosage and Administration (2.6) ].
- Emphasize the need for compliance with the full course of therapy.
Required Testing Prior to Administration
Prior to treatment with SIRTURO, obtain the following:
- Susceptibility information for the background regimen against M. tuberculosis isolate if possible [see Dosage and Administration (2.1) ]
- ECG [see Warnings and Precautions (5.1) ]
- Serum potassium, calcium, and magnesium concentrations [see Warnings and Precautions (5.1) ]
- Liver enzymes [see Warnings and Precautions (5.4) ]
Recommended Dosage in Adult Patients
The recommended dosage of SIRTURO in adult patients is:
| Dosage Recommendation | |
|---|---|
| Weeks 1 and 2 | Weeks 3 to 24 At least 48 hours between doses |
| 400 mg (4 of the 100 mg tablets OR 20 of the 20 mg tablets) orally once daily | 200 mg (2 of the 100 mg tablets OR 10 of the 20 mg tablets) orally three times per week |
Recommended dosage in pediatric patients is described in Table 2 below [see Dosage and Administration (2.4) ].
Administer SIRTURO tablets with food. The total duration of treatment with SIRTURO in adults is 24 weeks. When treatment with SIRTURO is considered necessary beyond 24 weeks, treatment may be continued at a dose of 200 mg three times per week [see Clinical Studies (14.1) ] .
Recommended Dosage in Pediatric Patients (2 years and older and weighing at least 8 kg)
The recommended dosage of SIRTURO in pediatric patients (2 years and older and weighing at least 8 kg) is based on body weight and shown in Table 2:
| Body Weight | Dosage Recommendation | |
|---|---|---|
| Weeks 1 and 2 | Weeks 3 to 24 At least 48 hours between doses | |
| 8 kg to less than 10 kg | 80 mg (4 of the 20 mg tablets) orally once daily | 40 mg (2 of the 20 mg tablets) orally three times per week |
| 10 kg to less than 15 kg | 120 mg (6 of the 20 mg tablets) orally once daily | 60 mg (3 of the 20 mg tablets) orally three times per week |
| 15 kg to less than 30 kg | 200 mg (2 of the 100 mg tablets OR 10 of the 20 mg tablets) orally once daily | 100 mg (1 of the 100 mg tablets OR 5 of the 20 mg tablets) orally three times per week |
| Greater than or equal to 30 kg | 400 mg (4 of the 100 mg tablets OR 20 of the 20 mg tablets) orally once daily | 200 mg (2 of the 100 mg tablets OR 10 of the 20 mg tablets) orally three times per week |
Administer SIRTURO tablets with food. The total duration of treatment with SIRTURO is 24 weeks. When treatment with SIRTURO is considered necessary beyond 24 weeks in patients 16 years and older, and weighing at least 30 kg, treatment may be continued at a dose of 200 mg three times per week [see Clinical Studies (14.1) ] .
Missed Dose
If a dose is missed during the first 2 weeks of treatment, do not administer the missed dose (skip the dose and then continue the daily dosing regimen). From Week 3 onwards, if a dose is missed, administer the missed dose as soon as possible, and then resume the 3 times a week dosing regimen. The total dose of SIRTURO during a 7-day period should not exceed the recommended weekly dose (with at least 24 hours between each intake).
Method of Administration
There is one method of administration of SIRTURO 100 mg tablet and four different methods of administration of SIRTURO 20 mg tablet as follows, each of which must be taken with food:
- For SIRTURO 100 mg tablet, administer the tablet whole with water. Take with food.
- For SIRTURO 20 mg tablet, the four different methods of administration are outlined below. Each administration method requires SIRTURO to be taken with food in addition to any soft food or beverage used to administer SIRTURO by the different methods for patients who cannot swallow intact SIRTURO 20 mg tablets.
Methods of Administration of SIRTURO 20 mg Tablet
Administration of 20 mg Tablets to Patients Who Can Swallow Intact Tablets:
Administer SIRTURO 20 mg tablet whole or split in half along the functional score line into two equal halves of 10 mg each. Administer SIRTURO 20 mg tablet with water. Take with food.
Administration of 20 mg Tablets to Patients Who Cannot Swallow Intact Tablets:
Dispersed in Water and Mixed with Beverage or Soft Food
For patients who have difficulty swallowing intact tablets, SIRTURO 20 mg tablet can be dispersed in water prior to administration with food. If needed to aid with administration, the dispersed mixture in water can be further mixed with a beverage (e.g., water, milk products, apple juice, orange juice, cranberry juice or carbonated beverage) or soft food (e.g., yogurt, apple sauce, mashed banana or porridge) as follows:
- Disperse tablets in water (maximum of 5 tablets in 5 mL of water) in a drinking cup.
- Mix the contents of the cup well until the tablets are completely dispersed and then orally administer the contents of the cup immediately with food. If needed to aid with oral administration, the dispersed mixture in water can be further mixed with at least 5 mL of beverage or 1 teaspoonful of soft food and then orally administer the contents of the cup immediately.
- If the total dose requires more than 5 tablets, repeat the above preparation steps with the appropriate number of additional tablets until the desired dose is reached.
- Ensure no tablet residue is left in the cup, rinse with beverage or add more soft food and orally administer the contents of the cup immediately.
- Take with food in addition to any beverage or soft food used to aid in administration.
Crushed and Mixed with Soft Food
SIRTURO 20 mg tablet can be crushed and mixed with soft food (e.g., yogurt, apple sauce, mashed banana or porridge) immediately prior to use and administered orally. To ensure no tablet residue is left in the container, add more soft food and administer the contents immediately. Take with food in addition to any beverage or soft food used to aid in administration.
Administration Through a Feeding Tube
SIRTURO 20 mg tablet can be administered through a feeding tube (8 French or greater) as follows:
- Disperse 5 tablets or less in 50 mL of non-carbonated water and mix well. Mixture should be white to almost white with visible particles expected.
- Administer through the feeding tube immediately.
- Repeat with additional tablets until desired dose is reached.
- Rinse and flush with 25 mL of additional water to ensure no tablet residue is left in materials used for preparation or the feeding tube.
- Administer with food.
DOSAGE FORMS AND STRENGTHS
- SIRTURO 20 mg tablet: uncoated, white to almost white oblong functionally scored tablet, with a score line on both sides, debossed with "2" and "0" on one side and plain on the other side.
- SIRTURO 100 mg tablet: uncoated, white to almost white round biconvex tablet with debossing of "T" over "207" on one side and "100" on the other side.
USE IN SPECIFIC POPULATIONS
- Lactation: Breastfeeding is not recommended unless infant formula is not available. If an infant is exposed to bedaquiline through breast milk, monitor for signs of bedaquiline-related adverse reactions, such as hepatotoxicity. (6 , 8.2 )
- Pediatrics: The safety and effectiveness of SIRTURO in pediatric patients less than 2 years of age and/or weighing less than 8 kg have not been established. (8.4 )
- Use with caution in patients with severe hepatic impairment and only when the benefits outweigh the risks. Monitor for SIRTURO-related adverse reactions. (8.6 )
- Use with caution in patients with severe renal impairment. (8.7 )
Pregnancy
Risk Summary
Available data from published literature of SIRTURO use in pregnant women are insufficient to evaluate a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks associated with active TB during pregnancy (see Clinical Considerations ) .
Reproduction studies performed in rats and rabbits have revealed no evidence of harm to the fetus due to oral administration of bedaquiline to pregnant rats and rabbits during organogenesis at exposures up to 6 times the clinical dose based on AUC comparisons (see Data ) .
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Active TB in pregnancy is associated with adverse maternal and neonatal outcomes including maternal anemia, caesarean delivery, preterm birth, low birth weight, birth asphyxia, and perinatal infant death.
Data
Animal Data
Pregnant rats were treated with bedaquiline at 5, 15 and 45 mg/kg (approximately 0.7, 2 and 6 times the clinical dose based on AUC comparisons) during the period of organogenesis (gestational Days 6 to 17, inclusive). Pregnant rabbits were treated with bedaquiline at 10, 30 and 100 mg/kg (approximately 0.05, 0.2 and 1.5 times the clinical dose based on AUC comparisons) during the period of organogenesis (gestational Days 6 to 19, inclusive). No embryotoxic effects were found in rats or rabbits at dose exposures up to 6 times the clinical dose exposures based on AUC comparisons.
Lactation
Risk Summary
Data from a published clinical lactation study demonstrate higher bedaquiline concentrations in breast milk compared to maternal plasma, suggesting that bedaquiline accumulates in breast milk (see Data ) . Data are insufficient to determine effects of the drug on the breastfed infants. No data are available on the effects of the drug on milk production. Because of the potential for serious adverse reactions in a breastfed infant, including hepatotoxicity, advise patients that breastfeeding is not recommended during treatment with SIRTURO and for 27.5 months (5 times the half-life) after the last dose unless infant formula is not available.
Clinical Considerations
If an infant is exposed to bedaquiline through breast milk, monitor for signs of bedaquiline-related adverse reactions, such as hepatotoxicity [see Adverse Reactions (6) ].
Data
A clinical lactation study was conducted in two lactating women who were approximately 7 weeks' postpartum. Bedaquiline and M2, its active metabolite, levels were measured between approximately 27 and 48 hours after the last bedaquiline dose, and concentrations of bedaquiline and M2 ranged from 2.61 to 8.11 mg/L and 0.27 to 0.81 mg/L, respectively. The milk:plasma ratios for bedaquiline and M2 at 27 to 48 hours after the last dose of bedaquiline ranged from approximately 19 to 29 and 4 to 6, respectively.
Pediatric Use
The safety and effectiveness of SIRTURO have been established in pediatric patients 2 years and older weighing at least 8 kg. The use of SIRTURO in this pediatric population is supported by evidence from the study of SIRTURO in adults together with additional pharmacokinetic and safety data from the single-arm, open-label, multi-cohort trial that enrolled 45 pediatric patients 2 years to less than 18 years of age with confirmed or probable pulmonary TB caused by M. tuberculosis resistant to at least rifampin who were treated with SIRTURO for 24 weeks in combination with a background regimen [see Dosage and Administration (2.4) , Adverse Reactions (6.1) , Clinical Pharmacology (12.3) and Clinical Studies (14.2) ] .
The safety, effectiveness and dosage of SIRTURO in pediatric patients less than 2 years of age and/or weighing less than 8 kg have not been established.
Geriatric Use
Clinical studies of SIRTURO did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger adult patients [see Clinical Pharmacology (12.3) ] .
Hepatic Impairment
The pharmacokinetics of bedaquiline were assessed after single-dose administration to adult patients with moderate hepatic impairment (Child-Pugh B) [see Clinical Pharmacology (12.3) ] . Based on these results, no dose adjustment is necessary for SIRTURO in patients with mild or moderate hepatic impairment. SIRTURO has not been studied in patients with severe hepatic impairment and should be used with caution in these patients only when the benefits outweigh the risks. Clinical monitoring for SIRTURO-related adverse reactions is recommended [see Warnings and Precautions (5.4) ] .
Renal Impairment
SIRTURO has mainly been studied in adult patients with normal renal function. Renal excretion of unchanged bedaquiline is not substantial (less than or equal to 0.001%). No dose adjustment is required in patients with mild or moderate renal impairment. In patients with severe renal impairment or end stage renal disease requiring hemodialysis or peritoneal dialysis, SIRTURO should be used with caution [see Clinical Pharmacology (12.3) ] . Monitor adult and pediatric patients for adverse reactions of SIRTURO when administered to patients with severe renal impairment or end stage renal disease requiring hemodialysis or peritoneal dialysis.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
QTc Prolongation
SIRTURO prolongs the QTc interval [see Clinical Pharmacology (12.2) ] . Use with drugs that prolong the QTc interval may cause additive QTc prolongation [see Adverse Reactions (6) ] . In Study 4, where SIRTURO was administered with the QTc prolonging drugs clofazimine and levofloxacin, 5% of patients in the 40-week SIRTURO treatment group experienced a QTc ≥500 ms and 43% of patients experienced an increase in QTc ≥60 ms over baseline. Of the clofazimine- and levofloxacin-treated patients in the 40-week control arm, 7% of patients experienced a QTc ≥500 ms and 39% experienced an increase in QTc ≥60 ms over baseline.
Obtain an ECG before initiation of treatment, 2 weeks after initiation, during treatment, as clinically indicated and at the expected time of maximum increase in the QTc interval of the concomitantly administered QTc prolonging drugs (as applicable). Obtain electrolytes at baseline and during treatment and correct electrolytes as clinically indicated.
The following may increase the risk for QTc prolongation when patients are taking SIRTURO:
- use with other QTc prolonging drugs
- a history of Torsade de Pointes
- a history of congenital long QTc syndrome
- a history of or ongoing hypothyroidism
- a history of or ongoing bradyarrhythmias
- a history of uncompensated heart failure
- serum calcium, magnesium, or potassium levels below the lower limits of normal
Discontinue SIRTURO if the patient develops:
- Clinically significant ventricular arrhythmia
- A QTc interval of greater than 500 ms (confirmed by repeat ECG)
If syncope occurs, obtain an ECG to detect QTc prolongation.
Mortality Imbalance in Clinical Trials
An increased risk of death was seen in the SIRTURO treatment group (9/79, 11.4%) compared to the placebo treatment group (2/81, 2.5%) in one placebo-controlled trial in adults (Study 1; based on the 120 week visit window). One death occurred during the 24 weeks of administration of SIRTURO. The imbalance in deaths is unexplained. No discernible pattern between death and sputum culture conversion, relapse, sensitivity to other drugs used to treat tuberculosis, HIV status, or severity of disease could be observed. In a subsequent active-controlled trial in adults (Study 4), deaths by Week 132 occurred in 11/211 (5.2%) patients in the 40-week SIRTURO treatment group, 8/202 (4%) patients in the active-control treatment group including four of 29 patients who received SIRTURO as part of a salvage treatment, and 2/143 (1.4%) patients in the 28-week SIRTURO treatment group [see Adverse Reactions (6.1) ] .
Risk of Development of Resistance to Bedaquiline
A potential for development of resistance to bedaquiline in M. tuberculosis exists [see Microbiology (12.4) ]. Bedaquiline must only be used in an appropriate combination regimen for the treatment of pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid, to reduce the risk of development of resistance to bedaquiline [see Indications and Usage (1) ] .
Hepatotoxicity
In clinical trials, more hepatic-related adverse reactions were reported in adults with the use of SIRTURO plus other drugs used to treat TB compared to other drugs used to treat TB without the addition of SIRTURO. Alcohol and other hepatotoxic drugs should be avoided while on SIRTURO, especially in patients with impaired hepatic function. Hepatic-related adverse reactions have also been reported in pediatric patients 5 years of age and older [see Adverse Reactions (6.1) ] .
Monitor symptoms (such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness and hepatomegaly) and laboratory tests (ALT, AST, alkaline phosphatase, and bilirubin) at baseline, monthly while on treatment, and as needed. Test for viral hepatitis and discontinue other hepatotoxic medications if evidence of new or worsening liver dysfunction occurs. Discontinue SIRTURO if:
- transaminase elevations are accompanied by total bilirubin elevation greater than two times the upper limit of normal
- transaminase elevations are greater than eight times the upper limit of normal
- transaminase elevations are greater than five times the upper limit of normal and persist beyond two weeks
Drug Interactions
CYP3A4 Inducers
Coadministration of SIRTURO with a moderate or strong CYP3A4 inducer decreases the systemic exposure of bedaquiline and may reduce the therapeutic effect of SIRTURO. Avoid coadministration of SIRTURO with moderate or strong CYP3A4 inducers, such as efavirenz and rifamycins (i.e., rifampin, rifapentine and rifabutin) [see Drug Interactions (7.1) ] .
CYP3A4 Inhibitors
Coadministration of SIRTURO with CYP3A4 inhibitors increases the systemic exposure of bedaquiline, which may increase the risk of adverse reactions. Closely monitor patient safety (e.g., liver function) when SIRTURO is coadministered with CYP3A4 inhibitors [see Drug Interactions (7.1) ] .
ADVERSE REACTIONS
The following serious adverse reactions are discussed elsewhere in the labeling:
- QTc Prolongation [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2) ]
- Mortality Imbalance in Clinical Trials [see Warnings and Precautions (5.2) ]
- Hepatotoxicity [see Warnings and Precautions (5.4) ]
- Drug Interactions [see Warnings and Precautions (5.5) ]
Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice. Refer to the prescribing information of the drugs used in combination with SIRTURO for their respective adverse reactions.
Clinical Studies Experience in Adults
Adverse reactions for SIRTURO were identified from safety data from 335 patients who received SIRTURO for eight weeks (Study 2) and 24 weeks (Studies 1 and 3), and 354 patients who received SIRTURO for 40 weeks or 28 weeks (Study 4). In these studies, patients received SIRTURO in combination with other antimycobacterial drugs.
Studies 1 and 2 were randomized, double-blind, placebo-controlled trials in newly diagnosed patients with pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid. Study 3 was an open-label, noncomparative study with SIRTURO administered as part of an individualized treatment regimen in previously treated patients with pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid, including patients resistant to second-line injectables and/or fluroquinolones.
In Study 1, 35% were Black, 17% were Hispanic, 13% were White, 9% were Asian, and 26% were of another race. Eight of 79 (10%) patients in the SIRTURO group and 16 of 81 (20%) patients in the placebo treatment group were HIV infected. Seven (8.9%) SIRTURO-treated patients and six (7.4%) placebo-treated patients discontinued Study 1 because of an adverse reaction.
Study 4 was an open-label, randomized, active-controlled trial in patients with pulmonary TB due to M. tuberculosis resistant to at least rifampin that evaluated a 40-week arm of SIRTURO in combination with other oral antimycobacterial drugs compared with a 40-week active control arm that included an injectable antimycobacterial drug in combination with other oral antimycobacterial drugs. A 28-week arm including SIRTURO, an injectable and other antimycobacterial drugs, was also evaluated in the trial, but recruitment was stopped early due to changes in the standard of care. In the population treated in the two 40-week arms (N=413), the median age was 32.8 years, 61% were male, 46% were Asian, 36% were Black, 18% were White and 16% were HIV infected.
Common Adverse Reactions
Table 3 presents select adverse reactions that occurred more frequently in the SIRTURO arm than the placebo arm in Study 1. The most common adverse reactions reported in 10% or more patients treated with SIRTURO and occurred more frequently than the placebo arm were nausea, arthralgia, headache, hemoptysis and chest pain.
| Adverse Reactions | SIRTURO Treatment Group N=79 n (%) | Placebo Treatment Group N=81 n (%) |
|---|---|---|
| Nausea | 30 (38) | 26 (32) |
| Arthralgia | 26 (33) | 18 (22) |
| Headache | 22 (28) | 10 (12) |
| Hemoptysis | 14 (18) | 9 (11) |
| Chest Pain | 9 (11) | 6 (7) |
| Anorexia | 7 (9) | 3 (4) |
| Transaminases Increased Terms represented by 'transaminases increased' included transaminases increased, AST increased, ALT increased, hepatic enzyme increased, and hepatic function abnormal. | 7 (9) | 1 (1) |
| Rash | 6 (8) | 3 (4) |
| Blood Amylase Increased | 2 (3) | 1 (1) |
No additional unique adverse reactions were identified from the uncontrolled Study 3.
Transaminase Elevations
In both Studies 1 and 2, transaminase elevations of at least 3 times the upper limit of normal developed more frequently in the SIRTURO treatment group (11/102 [10.8%] vs 6/105 [5.7%]) than in the placebo treatment group. In Study 3, 22/230 (9.6%) patients had alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than or equal to 3 times the upper limit of normal during the overall treatment period.
In Study 4 during the treatment phase, transaminase elevations of at least 3 times the upper limit of normal developed in 45/211 (21%) in the 40-week SIRTURO treatment group, 45/202 (22%) in the 40-week active comparator group, and 23 /143 (16%) in the 28-week SIRTURO-containing group.
Table 4 presents select adverse reactions occurring in 5% or greater of patients receiving SIRTURO in combination with other antimycobacterial drugs in the 40-week SIRTURO arm in Study 4. The most common adverse reactions reported in 10% or more patients in the 40-week SIRTURO arm were QTc prolongation, nausea, vomiting, arthralgia, transaminases increased, abdominal pain, pruritus, dizziness, headache, chest pain, rash, insomnia, dry skin, and palpitations.
| Adverse Reactions | SIRTURO 40-week, bedaquiline, levofloxacin, clofazimine, ethambutol, and pyrazinamide, supplemented by high-dose isoniazid and prothionamide in the first 16 weeks (intensive phase). N=211 n (%) | Active Control 40-week control treatment of moxifloxacin or levofloxacin, clofazimine, ethambutol, pyrazinamide, supplemented by injectable kanamycin, high dose isoniazid and prothionamide in the first 16 weeks (intensive phase). , Study 4 was not designed to evaluate meaningful comparisons of the incidence of adverse reactions in the SIRTURO and the active control treatment groups. N=202 n (%) |
|---|---|---|
| QTc prolongation | 128 (61) | 113 (56) |
| Nausea | 114 (54) | 126 (62) |
| Vomiting | 112 (53) | 125 (62) |
| Arthralgia | 93 (44) | 67 (33) |
| Transaminases increased Terms represented by 'transaminases increased' included AST increased, ALT increased, hepatic enzyme increased, hepatic function abnormal, hypertransaminasemia, and transaminases increased. | 63 (30) | 59 (29) |
| Abdominal pain Terms represented by 'abdominal pain' included abdominal pain, abdominal pain upper, abdominal pain lower, and abdominal tenderness. | 60 (28) | 48 (24) |
| Pruritus Terms represented by 'pruritus' included pruritus, pruritus generalized, and rash pruritic. | 58 (27) | 44 (22) |
| Dizziness | 37 (18) | 42 (21) |
| Headache | 36 (17) | 36 (18) |
| Chest pain | 33 (16) | 24 (12) |
| Rash Terms represented by 'rash' included rash, rash papular and rash maculopapular. | 30 (14) | 17 (8) |
| Insomnia | 30 (14) | 19 (9) |
| Dry skin | 25 (12) | 16 (8) |
| Palpitations | 21 (10) | 13 (6) |
| Myalgia | 19 (9) | 6 (3) |
| Paresthesia | 16 (8) | 8 (4) |
| Diarrhea | 12 (6) | 17 (8) |
In the 28-week SIRTURO-containing arm (N=143), in which SIRTURO was used in combination with other antimycobacterial drugs, the most common selected adverse reactions (greater than 10%) were QTc prolongation (56%), arthralgia (55%), nausea (43%), vomiting (29%), pruritus (25%), transaminases increased (21%), dizziness (21%), chest pain (17%), abdominal pain (17%), headache (16%), rash (12%), and hemoptysis (11%).
Mortality Imbalance in Clinical Studies
In Study 1, there was a statistically significant increased mortality risk by Week 120 in the SIRTURO treatment group compared to the placebo treatment group (9/79 (11.4%) versus 2/81 (2.5%), p-value=0.03, an exact 95% confidence interval of the difference [1.1%, 18.2%]). Five of the 9 SIRTURO deaths and the 2 placebo deaths were TB-related. One death occurred during the 24-week SIRTURO treatment period. The median time to death for the remaining eight patients in the SIRTURO treatment group was 329 days after last intake of SIRTURO. The imbalance in deaths is unexplained; no discernible pattern between death and sputum conversion, relapse, sensitivity to other drugs used to treat TB, HIV status, and severity of disease was observed.
In the open-label Study 3, 16/233 (6.9%) patients died. The most common cause of death as reported by the investigator was TB (nine patients).
In Study 4, patients originally assigned to non-SIRTURO containing regimens could receive SIRTURO as salvage when the original therapy was not tolerated or ineffective. At Week 132, deaths were observed in 11/211 (5.2%) patients in the 40-week SIRTURO arm versus 8/202 (4.0%) patients in the 40-week active control arm. For the 40-week SIRTURO arm, the most common cause of death was related to TB (five patients). In the 40-week active control arm, which included four of 29 patients who received SIRTURO as part of a salvage treatment, the most common cause of death was related to respiratory disease (e.g., TB, lobar pneumonia, respiratory distress in an HIV-positive patient). The adjusted difference in proportion of fatal adverse reactions between the 40-week SIRTURO arm and the 40-week active control arm was 1.2% [95% CI (-2.8%; 5.2%)]. In the 28-week SIRTURO arm at Week 132, 2/143 (1.4%) patients died; one of the two deaths was also related to TB. The overall mortality for patients treated with SIRTURO was 17/383 (4.4%).
Clinical Studies Experience in Pediatric Patients
The safety assessment of SIRTURO in pediatric patients is based on data from 45 pediatric patients in an ongoing, single-arm, open-label, multi-cohort trial.
Pediatric Patients (12 years to less than 18 years of age)
The first cohort was designed to enroll patients 12 years to less than 18 years of age (fifteen patients 14 years to less than 18 years of age were enrolled) with confirmed or probable pulmonary TB due to M. tuberculosis resistant to at least rifampin who received SIRTURO (400 mg once daily for the first 2 weeks and 200 mg 3 times per week for the following 22 weeks) in combination with a background regimen [see Clinical Studies (14.2) ] .
The most common adverse reactions were arthralgia in 6/15 (40%) patients, nausea in 2/15 (13%) patients, and abdominal pain in 2/15 (13%) patients. Among the 15 patients, no deaths were reported during the study (Week 120 analysis). Observed laboratory abnormalities were comparable to those in adults.
Pediatric Patients (5 years to less than 12 years of age)
The second cohort was designed to enroll patients 5 years to less than 12 years of age (fifteen patients aged 5 years to less than 11 years of age were enrolled) with confirmed or probable pulmonary TB due to M. tuberculosis resistant to at least rifampin who received SIRTURO (200 mg once daily for the first 2 weeks and 100 mg 3 times per week for the following 22 weeks) in combination with a background regimen [see Clinical Studies (14.2) ] .
The most common adverse reactions were related to elevations in liver enzymes (5/15, 33%), and led to discontinuation of SIRTURO in three patients. Elevations in liver enzymes were reversible upon discontinuation of SIRTURO and some of the background regimen drugs. Among these 15 pediatric patients, no deaths were reported during the study (Week 120 analysis).
Pediatric Patients (2 years to less than 5 years of age)
The third cohort enrolled 15 patients aged 2 years to less than 5 years of age with confirmed or probable pulmonary TB due to M. tuberculosis resistant to at least rifampin who received SIRTURO (80 to 120 mg once daily for the first 2 weeks and 40 to 60 mg 3 times per week for the following 22 weeks based on weight), in combination with a background regimen [see Clinical Studies (14.2) ] .
The most common adverse reaction was vomiting in 3/15 (20%) patients. Among these 15 pediatric patients, no deaths were reported during treatment with SIRTURO (Week 24 analysis).
DRUG INTERACTIONS
Effect of Other Drugs on SIRTURO
Strong and Moderate CYP3A4 Inducers
Coadministration of SIRTURO with moderate or strong CYP3A4 inducers may decrease systemic exposure of bedaquiline. Avoid coadministration of SIRTURO with strong or moderate CYP3A4 inducers [see Clinical Pharmacology (12.3) ] .
CYP3A4 Inhibitors
Coadministration of SIRTURO with CYP3A4 inhibitors increases the systemic exposure of bedaquiline which may increase the risk of adverse reactions. Closely monitor patient safety (e.g., liver function) when SIRTURO is coadministered with CYP3A4 inhibitors. No dose adjustment of SIRTURO is needed when coadministered with CYP3A4 inhibitors [see Clinical Pharmacology (12.3) ] .
Other Antimicrobial Medications
No dose adjustment of isoniazid or pyrazinamide is required during coadministration with SIRTURO.
In a placebo-controlled clinical trial in adult patients, no major impact of coadministration of SIRTURO on the pharmacokinetics of ethambutol, kanamycin, pyrazinamide, ofloxacin or cycloserine was observed.
QTc Interval Prolonging Drugs
In clinical trials of adult patients, additional QTc interval prolongation was observed during combination treatment with SIRTURO and other QTc prolonging drugs.
In Study 3, concurrent use of clofazimine with SIRTURO resulted in QTc prolongation: mean increases in QTc were larger in the 17 adult patients who were taking clofazimine with bedaquiline at Week 24 (mean change from Day-1 of 32 ms) than in patients who were not taking clofazimine with bedaquiline at Week 24 (mean change from Day-1 of 12 ms). Monitor ECGs if SIRTURO is coadministered to patients receiving other drugs that prolong the QTc interval, and discontinue SIRTURO if there is evidence of serious ventricular arrhythmia or QTc interval greater than 500 ms [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2) ] . ECG monitoring should be performed prior to initiation and at the expected time of maximum increase in the QTc interval of the concomitantly administered QTc prolonging drugs.
DESCRIPTION
SIRTURO ® contains bedaquiline fumarate, a diarylquinoline antimycobacterial drug for oral administration. Each SIRTURO 20 mg tablet contains 20 mg of bedaquiline (equivalent to 24.18 mg of bedaquiline fumarate). Each SIRTURO 100 mg tablet contains 100 mg of bedaquiline (equivalent to 120.89 mg of bedaquiline fumarate).
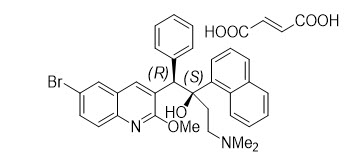
Bedaquiline fumarate is a white to almost white powder and is practically insoluble in aqueous media. The chemical name of bedaquiline fumarate is (1 R , 2 S )-1-(6-bromo-2-methoxy-3-quinolinyl)-4-(dimethylamino)-2-(1-naphthalenyl)-1-phenyl-2-butanol compound with fumaric acid (1:1). It has a molecular formula of C 32 H 31 BrN 2 O 2 ∙C 4 H 4 O 4 and a molecular weight of 671.58 (555.50 + 116.07). The molecular structure of bedaquiline fumarate is the following:
SIRTURO 20 mg tablet contains the following inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose 2910 5 mPa.s, polysorbate 20, purified water (removed during processing), silicified microcrystalline cellulose and sodium stearyl fumarate.
SIRTURO 100 mg tablet contains the following inactive ingredients: colloidal silicon dioxide, corn starch, croscarmellose sodium, hypromellose 2910 15 mPa.s, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polysorbate 20, purified water (removed during processing).
CLINICAL PHARMACOLOGY
Mechanism of Action
Bedaquiline is a diarylquinoline antimycobacterial drug [see Microbiology (12.4) ] .
Pharmacodynamics
Bedaquiline has activity against M. tuberculosis . The major metabolite, M2 is not thought to contribute significantly to clinical efficacy given its lower average exposure (23% to 31%) in humans and lower antimycobacterial activity (3-fold to 6-fold lower) compared to the parent compound. However, M2 plasma concentrations are correlated with QTc prolongation.
Cardiac Electrophysiology
In Study 1, the largest mean increase in QTc during the 24 weeks of SIRTURO treatment was 16 ms compared to 6 ms with placebo treatment (at Week 18). After SIRTURO treatment ended, the QTc gradually decreased, and the mean value was similar to that in the placebo group by study Week 60.
In Study 4, the mean change from baseline in QTc for the SIRTURO-containing arm was 22 ms (95% CI: 19; 24) at Week 2 (i.e., the end of the 2-week SIRTURO loading period), and 8 ms (95% CI: 2; 13) in the levofloxacin-containing control arm subgroup.
In Study 4, the mean QTc increased from baseline over the first 12 weeks for both SIRTURO and the non-SIRTURO containing control arm, when a plateau was reached. The largest mean QTc increase from baseline was 35 ms (95% CI: 32 to 38 ms) for the SIRTURO-containing arm and 28 ms (95% CI: 21 to 34 ms) for the levofloxacin-containing control arm subgroup [see Warnings and Precautions (5.1) and Drug Interactions (7.3) ].
Pharmacokinetics
Pharmacokinetics (PK) parameters
The pharmacokinetic properties of bedaquiline are summarized in Table 5 as mean (SD) in adult patients.
| Pharmacokinetic Parameters | Bedaquiline |
|---|---|
| SD=Standard Deviation | |
| Absorption | |
| Food effect | High fat meal (22 grams of fat, 558 total Kcal) increased C max and AUC by 2-fold. SIRTURO should be taken with food to enhance its oral bioavailability. |
| T max | Around 5 hours after single oral dose administration of SIRTURO |
| C max of bedaquiline | Week 2: 3060 (1124) ng/mL Week 24: 1838 (684) ng/mL Week 40: 1787 (666) ng/mL |
| C max of M2 | Week 2: 326 (135) ng/mL Week 24: 234 (85) ng/mL Week 40: 246 (103) ng/mL |
| Exposure | |
| AUC 168h of bedaquiline (AUC 24h for Week 2) | Week 2: 41510 (15064) ng.h/mL Week 24: 163924 (55710) (ng.h/mL) Week 40: 168376 (74476) ng.h/mL |
| AUC 168h of M2 (AUC 24h for Week 2) | Week 2: 7267 (3029) ng.h/mL Week 24: 37255 (13998) ng.h/mL Week 40: 39540 (17220) ng.h/mL |
| Distribution | |
| Percent bound to human plasma protein | greater than 99.9%. |
| apparent central volume of distribution | approximately 117 Liters. |
| Proportionality | C max and AUC increased proportionally up 700 mg (1.75 times the 400 mg loading dose). |
| Elimination | After reaching C max , bedaquiline concentrations decline tri-exponentially. |
| Apparent clearance of bedaquiline | 2.62 L/h |
| Apparent clearance of M2 | 4.95 L/h |
| Terminal half-life | ~ 5.5 months for both bedaquiline and N monodesmethyl metabolite (M2) |
| Metabolism | Metabolized to the M2 by CYP3A4; Relative exposure M2 versus bedaquiline: 23%~31% |
| Excretion | |
| Major route of excretion | Fecal excretion is the major route of elimination |
| %Excreted unchanged in urine | less than or equal to 0.001% of the dose in clinical studies |
Specific Populations
Patients with Hepatic Impairment: After single-dose administration of 400 mg SIRTURO to 8 adult patients with moderate hepatic impairment (Child-Pugh B), mean exposure to bedaquiline and M2 (AUC 672h ) was approximately 20% lower compared to healthy adults. SIRTURO has not been studied in patients with severe hepatic impairment [see Warnings and Precautions (5.4) and Use in Specific Populations (8.6) ].
Patients with Renal Impairment: SIRTURO has mainly been studied in adult patients with normal renal function. Renal excretion of unchanged bedaquiline is not substantial (less than or equal to 0.001%).
In a population pharmacokinetic analysis of adult TB patients treated with SIRTURO 200 mg three times per week, creatinine clearance was not found to influence the pharmacokinetic parameters of bedaquiline. It is therefore not expected that mild or moderate renal impairment will have a clinically relevant effect on the exposure to bedaquiline. However, in patients with severe renal impairment or end-stage renal disease requiring hemodialysis or peritoneal dialysis bedaquiline concentrations may be increased due to alteration of drug absorption, distribution, and metabolism secondary to renal dysfunction. As bedaquiline is highly bound to plasma proteins, it is unlikely that it will be significantly removed from plasma by hemodialysis or peritoneal dialysis [see Use in Specific Populations (8.7) ] .
Male and Female Patients: In a population pharmacokinetic analysis of adult TB patients treated with SIRTURO no clinically relevant difference in exposure between men and women were observed.
Race or Ethnicity: In a population pharmacokinetic analysis of adult TB patients treated with SIRTURO, systemic exposure (AUC) to bedaquiline was found to be 34% lower in Black patients than in patients from other race categories. This lower bedaquiline exposure in Black patients was not associated with lower efficacy in clinical trials.
HIV Coinfection: Bedaquiline exposure in patients coinfected with HIV was similar to that of patients not infected with HIV.
Geriatric Patients: There are limited data on the use of SIRTURO in TB patients 65 years of age and older.
In a population pharmacokinetic analysis of adult TB patients treated with SIRTURO, age was not found to influence the pharmacokinetics of bedaquiline. In five patients 65 to 69 years of age, the systemic bedaquiline exposure was similar to that in other adults.
Pediatric Patients:
Pediatric patients 12 years to less than 18 years of age with TB due to M. tuberculosis resistant to at least rifampin
The pharmacokinetic parameters of bedaquiline in 15 pediatric patients (body weight at baseline: 38 to 75 kg) who received the same adult dosage regimen of SIRTURO (400 mg once daily for the first two weeks and 200 mg three times per week for the following 22 weeks) in combination with a background regimen were comparable to those in adults. There was no impact of body weight on bedaquiline pharmacokinetics in this cohort.
Pediatric patients 5 years to less than 12 years of age with TB due to M. tuberculosis resistant to at least rifampin
Fifteen pediatric patients (body weight at baseline: 14 to 36 kg) received SIRTURO (200 mg once daily for the first two weeks and 100 mg three times per week for the following 22 weeks) in combination with a background regimen. Of these 15 pediatric patients, complete pharmacokinetic data were obtained for 10 patients at the aforementioned dosage regimen of SIRTURO. In nine of these 10 pediatric patients who weighed at least 15 kg at baseline, the mean bedaquiline C max and AUC 24h were similar to those of adult patients receiving the recommended adult dosage regimen. In one of these 10 pediatric patients who weighed 14 kg at baseline, the bedaquiline mean C max and AUC 24h were 3.8-fold and 2.6-fold, respectively, higher than the mean C max and AUC 24h in adult patients administered the recommended adult dosage regimen. The clinical significance of this higher pharmacokinetic plasma exposure in this one pediatric patient is not known.
Pediatric patients 2 years to less than 5 years of age with TB due to M. tuberculosis resistant to at least rifampin
Fifteen pediatric patients (body weight at baseline: 10 to 16 kg) received SIRTURO at a dose of 8 mg/kg once daily for 2 weeks (dose range: 80 to 120 mg; not an approved dosing regimen) followed by 4 mg/kg three times per week (dose range: 40 to 60 mg; not an approved dosing regimen) in combination with a background regimen. The results showed that the pediatric patients receiving the dosing regimens used in this pediatric study had mean bedaquiline C max and AUC 24h comparable to those of adult patients receiving the recommended adult dosage regimen [see Use in Specific Populations (8.4) ] .
The recommended dosing regimens for pediatric patients weighing 8 kg to less than 15 kg differ from those used in the pediatric clinical study. For pediatric patients weighing 8 kg to less than 15 kg, the recommended dosing regimens [see Dosage and Administration (2.4) ] were determined using a pharmacokinetic modeling and simulation approach based on clinical pharmacokinetic data, to achieve exposures comparable to those observed in adults taking SIRTURO tablets [see Use in Specific Populations (8.4) ] .
See Table 6 for a summary of the pharmacokinetic parameters at Week 12 in pediatric patients 2 years to less than 18 years of age.
| Pharmacokinetic Parameter | Bedaquiline Mean (SD) | ||
|---|---|---|---|
| 14 years to less than 18 years (N=15) | 5 years to less than 12 years (N=10) | 2 years to less than 5 years (N=15) | |
| SD=Standard Deviation | |||
| AUC 24h (ng∙h/mL) | 26,300 (10,300) | 32,200 (16,300) | 26,700 (7,270) N=10 |
| C max (ng/mL) | 1,800 (736) | 2,430 (1,670) | 1,810 (872) |
| T max (h) Median (range) | 4 (2 to 8) | 4 (2 to 8) | 6 (2 to 24) |
| C min (ng/mL) | 544 (263) | 461 (173) | 486 (161) |
Drug Interactions Studies
In vitro, bedaquiline did not significantly inhibit the activity of the following CYP450 enzymes that were tested: CYP1A2, CYP2A6, CYP2C8/9/10, CYP2C19, CYP2D6, CYP2E1, CYP3A4, CYP3A4/5 and CYP4A, and it does not induce CYP1A2, CYP2C9, CYP2C19, or CYP3A4 activities.
Bedaquiline is an in vitro substrate of CYP3A4, and because of this, the following clinical drug interaction studies were performed.
Clinical Studies
Ketoconazole: Coadministration of multiple-dose bedaquiline (400 mg once daily for 14 days) and multiple-dose ketoconazole (once daily 400 mg for 4 days) in healthy adult subjects increased the AUC 24h , C max and C min of bedaquiline by 22% [90% CI (12; 32)], 9% [90% CI (-2, 21)] and 33% [90% CI (24, 43)] respectively [see Drug Interactions (7.1) and (7.3) ] .
Clarithromycin: Coadministration of a single 100 mg dose of SIRTURO with clarithromycin at steady-state in healthy adults increased the mean [90% CI] bedaquiline exposure AUC 240h by 14% [9%;19%].
Clofazimine: In Study 3, long-term coadministration of clofazimine and SIRTURO, as part of a combination therapy for up to 24 weeks, did not affect bedaquiline exposure.
Rifampin: In a drug interaction study of single-dose 300 mg bedaquiline and multiple-dose rifampin (once daily 600 mg for 21 days) in healthy adult subjects, the exposure (AUC) to bedaquiline was reduced by 52% [90% CI (-57; -46)] [see Drug Interactions (7.1) ] .
Antimicrobial agents: The combination of multiple-dose bedaquiline 400 mg once daily with multiple-dose isoniazid/pyrazinamide (300 mg/2000 mg once daily) in healthy adult subjects did not result in clinically relevant changes in the exposure (AUC) to bedaquiline, isoniazid or pyrazinamide [see Drug Interactions (7.2) ] .
In a placebo-controlled study in adult patients with, no major impact of coadministration of bedaquiline on the pharmacokinetics of ethambutol, kanamycin, pyrazinamide, ofloxacin or cycloserine was observed.
Lopinavir/ritonavir: In a drug interaction study in healthy adults of single-dose bedaquiline (400 mg) and multiple-dose lopinavir (400 mg)/ritonavir (100 mg) given twice daily for 24 days, the mean AUC of bedaquiline was increased by 22% [90% CI (11; 34)] while the mean C max was not substantially affected. In Study 4, coadministration of SIRTURO with lopinavir/ritonavir in patients coinfected with HIV increased bedaquiline exposure, with an estimated 68% [90% CI (29%; 117%)] increase in mean AUC 168h at Week 24 and 72% [90% CI (32%; 123%)] at Week 40, in HIV-positive Black patients treated with lopinavir/ritonavir (N=16) compared to HIV-positive and -negative Black patients without lopinavir/ritonavir (N=67) treatment [see Drug Interactions (7.1) ] .
Nevirapine: Coadministration of multiple-dose nevirapine 200 mg twice daily for 4 weeks in HIV-infected adult patients with a single 400 mg dose of bedaquiline did not result in clinically relevant changes in the exposure to bedaquiline. In Study 4, coadministration of nevirapine and SIRTURO as part of combination therapy for up to 40 weeks in patients coinfected with HIV, resulted in a 29% [90% CI (0; 50%)] decrease in bedaquiline exposure (AUC 168h ).
Efavirenz: Coadministration of a single dose of bedaquiline 400 mg and efavirenz 600 mg daily for 27 days to healthy adult volunteers resulted in approximately a 20% decrease in the AUC inf of bedaquiline; the C max of bedaquiline was not altered. The AUC and C max of the primary metabolite of bedaquiline (M2) were increased by 70% and 80%, respectively. The effect of efavirenz on the pharmacokinetics of bedaquiline and M2 following steady-state administration of bedaquiline has not been evaluated [see Drug Interactions (7.3) ] .
Microbiology
Mechanism of Action
SIRTURO is a diarylquinoline antimycobacterial drug that inhibits mycobacterial ATP (adenosine 5'- triphosphate) synthase, by binding to subunit c of the enzyme that is essential for the generation of energy in M. tuberculosis .
Resistance
A potential for development of resistance to bedaquiline in M. tuberculosis exists. Modification of the atpE target gene, and/or upregulation of the MmpS5-MmpL5 efflux pump ( Rv0678 mutations) have been associated with increased bedaquiline MIC values in isolates of M. tuberculosis. Target-based mutations generated in preclinical studies lead to 8- to 133-fold increases in bedaquiline MIC, resulting in MICs ranging from 0.25 to 4 micrograms per mL. Efflux-based mutations have been seen in preclinical and clinical isolates. These lead to 2- to 8-fold increases in bedaquiline MICs, resulting in bedaquiline MICs ranging from 0.25 to 0.5 micrograms per mL.
M. tuberculosis isolates that had developed at least a 4-fold increase in bedaquiline MIC were associated with mutations in Rv0678 gene that lead to upregulation of the MmpS5-MmpL5 efflux pump. Isolates with these efflux-based mutations are less susceptible to clofazimine. Isolates that are phenotypically resistant to bedaquiline should be tested for cross-resistance to clofazimine, if clofazimine is being considered as part of the treatment regimen. In Studies 2, 3 and 4 there was no clear relationship between the presence of Rv0678 mutations at baseline and treatment outcome.
Antimicrobial Activity
SIRTURO has been shown to be active in vitro and in clinical infections against most isolates of M. tuberculosis [see Indications and Usage (1) and Clinical Studies (14) ] .
Susceptibility Testing
MICs for baseline M. tuberculosis isolates from patients in Studies 1 and 3 and their sputum culture conversion rates at Week 24 and from patients in the 40-week SIRTURO arm of Study 4 at Week 76 are shown in Table 7 below. Based on the available data, there was no relationship between poor microbiologic outcomes and baseline bedaquiline MIC.
| Baseline Bedaquiline MIC (micrograms/mL) | SIRTURO Treatment Group 24-Week Culture Conversion Rate (Study 1 and Study 3) n/N (%) | SIRTURO Treatment Group 76-Week Culture Conversion Rate (Study 4, 40-Week Arm) In addition, at Week 76 in the 40-week SIRTURO arm, there were 6 patients for whom no baseline MIC data were available. All 6 patients culture converted. n/N (%) |
|---|---|---|
| N=number of patients with data per baseline BDQ MIC category; n=number of patients with culture conversion; MIC=minimum inhibitory concentration | ||
| ≤ 0.008 | 2/2 (100) | 5/5 (100) |
| 0.015 | 13/15 (86.7) | 26/26 (100) |
| 0.03 | 36/46 (78.3) | 67/69 (97.1) |
| 0.06 | 82/107 (76.6) | 51/51 (100) |
| 0.12 | 36/42 (85.7) | 20/20 (100) |
| 0.25 | 3/4 (75.0) | 2/2 (100) |
| 0.5 | 5/6 (83.3) | 0 |
| ≥ 1 | 0/1 (0) | 0 |
Nineteen patients in the efficacy population of Study 3 had bedaquiline susceptibility testing results of paired (baseline and post-baseline, all of which were at Week 24 or later) genotypically identical M. tuberculosis isolates. Twelve of the 19 had a post-baseline ≥4-fold increase in bedaquiline MIC. Whole genome sequencing of nine of these 12 post-baseline isolates was done and no mutations were found in the ATP synthase operon. All nine were found to have a mutation in Rv0678 . Eleven of the twelve (11/12) increases in bedaquiline MIC were seen in patients with isolates that also demonstrated increased MICs to fluoroquinolones and/or second-line injectables.
Based on available data, response rate (culture conversion at Week 120 endpoint) was similar in patients with ≥4-fold increases in bedaquiline MIC (5/12) and patients with <4-fold increases (3/7).
In the 40-week SIRTURO arm of Study 4, paired isolates (baseline and postbaseline, genotypically identical) susceptibility testing (MIC) results were available for 30 of 196 patients in the mITT population. At Week 132, three of five patients with a ≥4-fold increase in bedaquiline MIC and 25 of 25 patients with <4-fold increases had culture converted. Similarly, in the 28-week arm of Study 4, for 25 of 134 patients in the mITT population with paired isolates, two of four patients, with a ≥4-fold increase in bedaquiline MIC and 20 of 21 patients with <4-fold increases had culture converted. Based on the limited data, there was no clear relationship between bedaquiline MIC and microbiological outcomes.
For specific information regarding susceptibility test criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: www.fda.gov/STIC .
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Bedaquiline was not carcinogenic in rats up to the maximum tolerated dose of 10 mg/kg/day. Exposures at this dose in rats (AUCs) were within 1-fold to 2-fold of those observed in adult patients in the clinical trials.
No mutagenic or clastogenic effects were detected in the in vitro non-mammalian reverse mutation (Ames) test, in vitro mammalian (mouse lymphoma) forward mutation assay and an in vivo mouse bone marrow micronucleus assay.
SIRTURO did not affect fertility when evaluated in male and female rats at approximately twice the clinical exposure based on AUC comparisons. There was no effect of maternal treatment on sexual maturation, mating performance or fertility in F1 generation exposed to bedaquiline in utero at approximately twice the human exposure.
Animal Toxicology and/or Pharmacology
Bedaquiline is a cationic, amphiphilic drug that induced phospholipidosis (at almost all doses, even after very short exposures) in drug-treated animals, mainly in cells of the monocytic phagocytic system (MPS). All species tested showed drug-related increases in pigment-laden and/or foamy macrophages, mostly in the lymph nodes, spleen, lungs, liver, stomach, skeletal muscle, pancreas and/or uterus. After treatment ended, these findings were slowly reversible. Muscle degeneration was observed in several species at the highest doses tested. For example, the diaphragm, esophagus, quadriceps and tongue of rats were affected after 26 weeks of treatment at doses similar to clinical exposures based on AUC comparisons. These findings were not seen after a 12-week, treatment-free, recovery period and were not present in rats given the same dose biweekly. Degeneration of the fundic mucosa of the stomach, hepatocellular hypertrophy and pancreatitis were also seen.
CLINICAL STUDIES
SIRTURO Use in Adult Patients With Pulmonary Tuberculosis
Study 1 (NCT00449644, Stage 2) was a placebo-controlled, double-blind, randomized trial conducted in patients with newly diagnosed sputum smear-positive pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid. Patients were randomized to receive a combination of SIRTURO or placebo with five other antimycobacterial drugs (i.e., ethionamide, kanamycin, pyrazinamide, ofloxacin, and cycloserine/terizidone or available alternative) for a total duration of 18 to 24 months or at least 12 months after the first confirmed negative culture. Treatment was 24 weeks of treatment with SIRTURO 400 mg once daily for the first two weeks followed by 200 mg three times per week for 22 weeks or matching placebo for the same duration. Overall, 79 patients were randomized to the SIRTURO arm and 81 to the placebo arm. A final evaluation was conducted at Week 120.
Sixty-seven patients randomized to SIRTURO and 66 patients randomized to placebo had confirmed pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid, based on susceptibility tests (taken prior to randomization) or medical history if no susceptibility results were available, and were included in the efficacy analyses. Demographics were as follows: 64% of the study population was male, with a median age of 33 years, 38% were Black, 17% were Hispanic, 11% were White, 11% were Asian, and 24% were of another race; and 14% were HIV-positive (median CD4 cell count 446 cells/µL). Most patients had cavitation in one lung (62%); and 20% of patients had cavitation in both lungs.
Time to sputum culture conversion was defined as the interval in days between the first dose of study drug and the date of the first of two consecutive negative sputum cultures collected at least 25 days apart during treatment. In this trial, the SIRTURO treatment group had a decreased time to culture conversion and improved culture conversion rates compared to the placebo treatment group at Week 24. Median time to culture conversion was 83 days for the SIRTURO treatment group compared to 125 days for the placebo treatment group. Table 8 shows the proportion of patients with sputum culture conversion at Week 24 and Week 120.
| Microbiologic Status | SIRTURO (24 weeks) + combination of other antimycobacterial drugs N=67 | Placebo (24 weeks) + combination of other antimycobacterial drugs N=66 | Difference [95% CI] p-value |
|---|---|---|---|
| Week 24 | |||
| Sputum Culture Conversion | 78% | 58% | 20.0% [4.5%, 35.6%] 0.014 |
| Treatment failure A patient's reason for treatment failure was counted only in the first row for which a patient qualifies. | 22% | 42% | |
| Died | 1% | 0% | |
| Lack of conversion | 21% | 35% | |
| Discontinuation | 0% | 8% | |
| Week 120 Patients received 24 weeks of SIRTURO or placebo for the first 24 weeks and received a combination of other antimycobacterial drugs for up to 96 weeks. | |||
| Sputum Culture Conversion | 61% | 44% | 17.3% [0.5%, 34.0%] 0.046 |
| Treatment failure | 39% | 56% | |
| Died | 12% | 3% | |
| Lack of conversion/relapse | 16% | 35% | |
| Discontinuation | 10% | 18% |
Study 2 (NCT00449644, Stage 1) was a smaller placebo-controlled study designed similarly to Study 1 except that SIRTURO or placebo was given for only eight weeks instead of 24 weeks. Patients were randomized to either SIRTURO and other drugs used to treat pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid (SIRTURO treatment group) (n=23) or placebo and other drugs used to treat TB (placebo treatment group) (n=24). Twenty-one patients randomized to the SIRTURO treatment group and 23 patients randomized to the placebo treatment group had confirmed pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid based on patients' baseline M. tuberculosis isolate obtained prior to randomization. The SIRTURO treatment group had a decreased time to culture conversion and improved culture conversion rates compared to the placebo treatment group at Week 8. At Weeks 8 and 24, the differences in culture conversion proportions were 38.9% [95% CI: (12.3%; 63.1%) and p-value: 0.004], 15.7% [95% CI: (-11.9%; 41.9%) and p-value: 0.32], respectively.
Study 3 (NCT00910871) was a Phase 2b, uncontrolled study to evaluate the safety, tolerability, and efficacy of SIRTURO as part of an individualized treatment regimen in 233 patients with sputum smear positive (within 6 months prior to screening) pulmonary TB due to M. tuberculosis resistant to at least rifampin and isoniazid, including patients with isolates resistant to second-line injectables and/or fluoroquinolones. Patients received SIRTURO for 24 weeks in combination with antimycobacterial drugs. Upon completion of the 24-week treatment with SIRTURO, all patients continued to receive their background regimen in accordance with national TB program (NTP) treatment guidelines. A final evaluation was conducted at Week 120. Treatment responses to SIRTURO at Week 120 were generally consistent with those from Study 1.
Study 4 (NCT02409290) was a Phase 3, open-label, multicenter, active-controlled, randomized trial to evaluate the efficacy and safety of SIRTURO, coadministered with other oral anti-TB drugs for 40 weeks in patients with sputum smear-positive pulmonary TB caused by M. tuberculosis that was resistant to at least rifampin. Patients in whom the M. tuberculosis strain was known to be resistant at screening to second-line injectable agents or fluoroquinolones were excluded from enrollment. When phenotypic susceptibility testing of the baseline isolates became available post-randomization, patients infected with M. tuberculosis resistant to either second-line injectable agents or fluoroquinolones were kept in the study, however, those with M. tuberculosis resistant to both second-line injectables and fluoroquinolones were discontinued from the study.
Patients were randomized to one of four treatment arms:
- Arm A (N=32), the locally used treatment in accordance with 2011 WHO treatment guidelines with a recommended 20-month duration
- Arm B (N=202), a 40-week treatment of moxifloxacin (N=140) or levofloxacin (N=62), clofazimine, ethambutol, pyrazinamide, supplemented by injectable kanamycin, high-dose isoniazid and prothionamide in the first 16 weeks (intensive phase)
- Arm C (N=211), a 40-week, all-oral treatment of SIRTURO, levofloxacin, clofazimine, ethambutol, and pyrazinamide, supplemented by high-dose isoniazid and prothionamide in the first 16 weeks (intensive phase)
- Arm D (N=143), a 28-week treatment consisting of SIRTURO, levofloxacin, clofazimine, and pyrazinamide, supplemented by kanamycin injectable and a higher isoniazid dose for the first eight weeks (intensive phase)
SIRTURO was administered 400 mg once daily for the first two weeks and 200 mg three times a week for the following 38 weeks (in Arm C) or 26 weeks (in Arm D).
All patients were to be followed up until study completion at Week 132. During study conduct, enrollment in Arms A and D was stopped due to changes in the standard of care for TB treatment. Patients already randomized to these study arms were to complete their assigned treatment and follow-up.
The primary objective was to assess whether the proportion of patients with a favorable efficacy outcome in Arm C was noninferior to that in Arm B at Week 76.
The primary efficacy outcome measure was the proportion of patients with a favorable outcome at Week 76. A favorable outcome at Week 76 was defined as the last two consecutive cultures being negative, and with no unfavorable outcome. An unfavorable outcome at Week 76 was assessed as a composite endpoint, covering both clinical and microbiological aspects such as changes in TB treatment, all-cause mortality, at least one of the last two culture results positive, or no culture results within the Week 76 window. In case of treatment failure, recurrence or serious toxicity on the allocated treatment, salvage treatment that could include SIRTURO was provided, based on investigator judgment.
The modified intent-to-treat population (mITT) was the primary efficacy population and included all randomized patients with a positive sputum culture for M. tuberculosis that was resistant to at least rifampin and not resistant to both second-line injectables and fluoroquinolones, based on susceptibility results (taken prior to randomization). A total of 196 and 187 patients were included in the mITT population in Arm C and Arm B, respectively. Overall, in both treatment arms, 62% were male of median age 33 years, 47% were Asian, 34% were Black, 19% were White, and 14% were HIV-coinfected. Most patients had lung cavitation (74%), with multiple cavities in 63% and 47% of patients in Arm C and Arm B, respectively. The baseline drug resistance profile of M. tuberculosis for Arms C and B were as follows: 14% had resistance to rifampin while susceptible to isoniazid, 75% had resistance to rifampin and isoniazid, and 10% had resistance to rifampin, isoniazid, and either a second-line injectable or a fluoroquinolone.
For efficacy analyses beyond Week 76, data collection was stopped at the point when the last recruited patient was projected to reach Week 96. The long-term efficacy data therefore include data up to at least Week 96 for all patients, and up to Week 132 for 146/196 (74.5%) patients in Arm C and 145/187 (77.5%) patients in Arm B.
Table 9 shows results for favorable and unfavorable outcomes at Week 76 and Week 132 in Study 4.
| SIRTURO Arm C 40-week, all-oral regimen of SIRTURO, levofloxacin, clofazimine, ethambutol, and pyrazinamide, supplemented by high-dose isoniazid and prothionamide in the first 16 weeks (intensive phase). (N=196) | Active Control Arm B 40-week control treatment of moxifloxacin or levofloxacin, clofazimine, ethambutol, pyrazinamide, supplemented by injectable kanamycin, high dose isoniazid and prothionamide in the first 16 weeks (intensive phase). (N=187) | |
|---|---|---|
| mITT = modified intent-to-treat | ||
| Favorable outcome at Week 76 n (%) | 162 (82.7) | 133 (71.1) |
| Difference The adjusted difference in proportions was estimated using a stratified analysis of the risk difference from each stratum using Cochran Mantel-Haenszel weights. The analysis was stratified by randomization protocol and HIV and CD4 cell count status. SIRTUROvs Active Control (95% CI) | 11.0% (2.9%, 19.0%) | |
| Unfavorable outcome at Week 76 n (%) | 34 (17.3) | 54 (28.9) |
| Reasons for unfavorable outcome through Week 76 Patients were classified by the first event that made the patient unfavorable. Of the patients with an unfavorable outcome at Week 76 in the control arm, 29 patients had a treatment modification from their allocated treatment that included SIRTURO as part of a salvage regimen. | ||
| Treatment modified or extended | 16 (8.2) | 43 (23.0) |
| No culture results within Week 76 window | 12 (6.1) | 7 (3.7) |
| Death through Week 76 | 5 (2.6) | 2 (1.1) |
| At least one of last 2 cultures positive at Week 76 | 1 (0.5) | 2 (1.1) |
| Favorable outcome at Week 132 n (%) Week 132 outcome reflects efficacy follow up until the last patient reached Week 96. | 154 (78.6) | 129 (69.0) |
| DifferenceSIRTUROvs Active Control (95% CI) | 9.0% (0.6%,17.5%) | |
SIRTURO Use in Pediatric Patients (2 years to less than 18 years of age) With Pulmonary Tuberculosis
The pediatric trial, (NCT02354014), was designed as a single-arm, open-label, multi-cohort trial to evaluate the pharmacokinetics, safety and tolerability of SIRTURO in combination with a background regimenin patients 2 to less than 18 years of age with confirmed or probable pulmonary TB due to M. tuberculosis resistant to at least rifampin.
Pediatric Patients (12 years to less than 18 years of age)
Fifteen patients 14 years to less than 18 years of age were enrolled in the first cohort. The median age was 16 years, 80% were female, 53% were Black, 33% were White and 13% were Asian. No patient 12 years to less than 14 years of age was enrolled in this cohort. SIRTURO was administered as 400 mg once daily for the first two weeks and 200 mg three times per week for the following 22 weeks using the 100 mg tablet.
In the subset of patients with mycobacteria growth indicator tube (MGIT) culture positive pulmonary TB resistant to at least rifampin at baseline, treatment with SIRTURO resulted in conversion to a negative culture in 75% (6/8 patients) at Week 24, which was sustained at Week 120.
Pediatric Patients (5 years to less than 12 years of age)
Fifteen patients 5 years to 10 years of age were enrolled in the second cohort. The median age was seven years, 60% were female, 60% were Black, 33% were White and 7% were Asian. No patient older than 10 years to less than 12 years of age was enrolled in this cohort. The body weight range was 14 kg to 36 kg; only one patient weighing 14 kg was enrolled. SIRTURO was administered as 200 mg once daily for the first two weeks and 100 mg three times per week for the following 22 weeks using the 20 mg tablet.
In the subset of patients with MGIT culture positive pulmonary TB resistant to at least rifampin at baseline, treatment with SIRTURO resulted in conversion to a negative culture in 100% (3/3 patients) at Week 24, which was sustained at Week 120.
Pediatric Patients (2 years to less than 5 years of age)
Fifteen patients 2 years to less than 5 years of age were enrolled in the third cohort. The median age was 4 years, 47% were female, 27% were Black and 73% were Asian. At baseline, the body weight ranged from 10 kg to 16 kg. SIRTURO was administered as 8 mg/kg once daily for the first two weeks (dose range: 80 to 120 mg; not an approved dosing regimen) followed by 4 mg/kg three times per week (dose range: 40 to 60 mg; not an approved dosing regimen) for the following 22 weeks using the 20 mg tablet.
In the one patient with MGIT culture positive pulmonary TB resistant to at least rifampin at baseline, treatment with SIRTURO resulted in conversion to a negative culture (1/1 MGIT culture evaluable patient) at Week 24, which was sustained at Week 120.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
SIRTURO ® 20 mg tablets are supplied as uncoated white to almost white oblong functionally scored tablets with a score line on both sides, debossed with "2" and "0" on one side and plain on the other side.
SIRTURO ® 100 mg tablets are supplied as uncoated white to almost white round biconvex 100 mg tablets with debossing of "T" over "207" on one side and "100" on the other side.
SIRTURO tablets are packaged in white high-density polyethylene (HDPE) bottles with child-resistant polypropylene (PP) closure with induction seal liner in the following configurations:
- 20 mg tablets – bottles of 60 tablets. Each bottle contains silica gel desiccant (NDC 59676-702-60)
- 100 mg tablets – bottles of 188 tablets (NDC 59676-701-01).
Storage and Handling
SIRTURO 20 mg Tablet
Store in original container. Bottle contains desiccant. Do not discard desiccant. Protect from light and moisture. Keep the container tightly closed.
Store at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
SIRTURO 100 mg Tablet
Dispense in original container. Store tablets dispensed outside the original container in a tight light-resistant container with an expiration date not to exceed 3 months. Protect from light. Keep the container tightly closed.
Store at 25°C (77°F); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Bedaquiline is a diarylquinoline antimycobacterial drug [see Microbiology (12.4) ] .
Sirturo - Bedaquiline Fumarate tablet PubMed™ news
- Journal Article • 2026 MayAdverse events in bedaquiline- and pretomanid-based regimens for drug-resistant tuberculosis from trial, implementation and pharmacovigilance studies.
- Journal Article • 2026 MayBreaking barriers in extensively drug-resistant TB: pretomanid-centred long regimen achieves cure in a bedaquiline-resistant strain.
- Journal Article • 2026 MayQT prolongation in participants receiving bedaquiline-containing regimens: analysis of data from Phase 3 STREAM Stage 2.
- Journal Article • 2026 MayQuality of life outcomes and patient-reported experiences in a randomised controlled trial for rifampicin-resistant TB (PRACTECAL-PRO): a mixed-methods study.
- Journal Article • 2026 MayOutcomes in patients with multidrug- or rifampin-resistant tuberculosis treated with bedaquiline and/or delamanid under the South Korean nationwide approval process.