Get your patient on Sterile Water - Sterile Water injection (Sterile Water)
Sterile Water - Sterile Water injection prescribing information
This parenteral preparation is indicated only for diluting or dissolving drugs for intravenous, intramuscular, or subcutaneous injection, according to instructions of the manufacturer of the drug to be administered.
The volume of the preparation to be used for diluting or dissolving any drug for injection is dependent on the vehicle concentration, dose, and route of administration as recommended by the manufacturer. This parenteral should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Please refer to INSTRUCTIONS FOR USE for administration instructions.
Sterile Water for Injection must be made approximately isotonic prior to use.
Reactions which may occur because of this solution, added drugs, or the technique of reconstitution or administration include febrile response, local tenderness, abscess, tissue necrosis or infection at the site of injection, venous thrombosis, or phlebitis extending from the site of injection and extravasation.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate countermeasures, and if possible, retrieve and save the remainder of the unused vehicle for examination.
Some drugs for injection may be incompatible in a given vehicle, when combined in the same vehicle, or in a vehicle containing benzyl alcohol. Consult with pharmacist, if available.
Use aseptic technique for withdrawal from the container. When diluting or dissolving drugs, mix thoroughly and use promptly.
Do not store reconstituted solutions of drugs for injection unless otherwise directed by the manufacturer of the solute. Do not use unless the solution is clear.
Do not reuse single-dose containers. Discard unused portion.
This preparation is designed solely for parenteral use, only after addition of drugs that require dilution or must be dissolved in an aqueous vehicle prior to injection.
Sterile Water for Injection, USP is a sterile, nonpyrogenic preparation of water for injection, which contains no bacteriostat, antimicrobial agent or added buffer, and is supplied only in single-dose containers to dilute or dissolve drugs for injection. For intravenous injection, dissolve sufficient solute to make an approximately isotonic solution. Water for Injection, USP is chemically designated H2O. pH 5.0 to 7.0.
The plastic single-dose vial is fabricated from polypropylene resin. The plastic vials feature a twist-off cap which, when removed, allows access to a luer-lock fitting for connection to a luer-lock syringe. The twist-off cap is a one-time use tamper evident feature, and the container vial cannot be reclosed once the cap is removed.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight.
Average adult daily requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water for distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
The small volume of fluid provided by Sterile Water for Injection when used only as a pharmaceutical aid for diluting or dissolving drugs for parenteral injection, is unlikely to exert a significant effect on fluid balance except possibly in neonates or very small infants.
Sterile Water for Injection, USP is supplied in 5 mL vials, packaged in cartons as follows: NDC 0487-6105-01, 30 individual 5 mL single-dose luer-lock vials.
Discard unused portion. Each vial is made from a polypropylene (PP) resin. Store at 2°C to 25°C (36°F to 77°F).
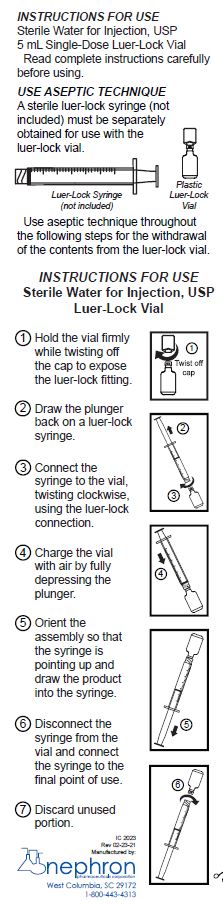
Sterile Water for Injection, USP 5 mL Single-Dose Luer-Lock Vial Read complete instructions carefully before using. USE ASEPTIC TECHNIQUE and instructions for use. A sterile luer-lock syringe (not included) must be separately obtained for use with the luer-lock vial. Use aseptic technique throughout the Infor the withdrawal of the contents from the luer-lock vial.