Get your patient on Susvimo (Ranibizumab)
Susvimo prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Susvimo patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
- For intravitreal use via SUSVIMO ocular implant. (2.1 )
- Neovascular (wet) Age-related Macular Degeneration (AMD) and Diabetic Macular Edema (DME)
The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO implant with refills every 24 weeks (approximately 6 months). (2.2 ) - Diabetic Retinopathy (DR)
The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO implant with refills every 36 weeks (approximately 9 months). (2.3 ) - Supplemental treatment with 0.5 mg intravitreal ranibizumab injection may be administered in the affected eye if clinically necessary. (2.4 )
- Perform the initial implantation, refill-exchange, and implant removal (if necessary) procedures under strict aseptic conditions. (2.5 , 2.6 , 2.7 , 2.8 )
General Information
For Intravitreal Use via SUSVIMO ocular implant.
The SUSVIMO initial fill and ocular implant insertion and implant removal procedures must be performed under aseptic conditions by a physician experienced in vitreoretinal surgery. The SUSVIMO ocular implant must be surgically implanted in the eye or removed from the eye (if medically necessary) in an operating room using aseptic technique. See SUSVIMO Instructions for Use and the standardized steps to optimize surgical outcomes.
SUSVIMO refill-exchange procedures must be performed under aseptic conditions by a physician experienced in ophthalmic surgery [see Dosage and Administration (2.7) ] .
Do not administer SUSVIMO (ranibizumab injection) as a bolus intravitreal injection. Do not substitute SUSVIMO (ranibizumab injection) with other ranibizumab products.
Initial Fill : One SUSVIMO initial fill needle (34-gauge, with integrated 5 μm filter and blue cap) is included. A 5-micron sterile filter needle (19-gauge × 1½ inch), and a 1 mL Luer lock syringe are needed but not included .
Refill-Exchange : One SUSVIMO refill needle (34-gauge with integrated 5 μm filter and clear cap) is included. A 5-micron sterile filter needle (19-gauge × 1½ inch), and a 1 mL Luer lock syringe are needed but not included .
Neovascular (Wet) Age-Related Macular Degeneration (AMD) and Diabetic Macular Edema (DME)
The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO ocular implant with refills administered every 24 weeks (approximately 6 months).
Diabetic Retinopathy (DR)
The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO ocular implant with refills administered every 36 weeks (approximately 9 months).
Supplemental Treatment with Intravitreal Ranibizumab Injection
Supplemental treatment with 0.5 mg (0.05 mL of 10 mg/mL) intravitreal ranibizumab injection may be administered in the affected eye while the SUSVIMO implant is in place and if clinically necessary [see Clinical Studies (14) ] .
Ocular Implant Initial Fill
The implant initial fill procedure must be performed by a physician experienced in vitreoretinal surgery [see Dosage and Administration (2.1) ]. The implant will be filled using aseptic technique with 0.02 mL of SUSVIMO (ranibizumab injection) prior to insertion of the implant into the patient's eye [see Dosage and Administration (2.6) ].
Refer to the complete SUSVIMO Instructions for Use for the initial fill and implant procedure included in the insertion tool assembly carton for further details.
Use aseptic technique to carry out the following preparation steps prior to insertion of the ocular implant into the patient's eye:
Step 1: Gather the supplies needed.
| ||
| Step 2: Transfer Dose from Vial to Syringe | ||
| Note: | Use the filter needle (not included) to withdraw SUSVIMO (ranibizumab injection) from the vial. | 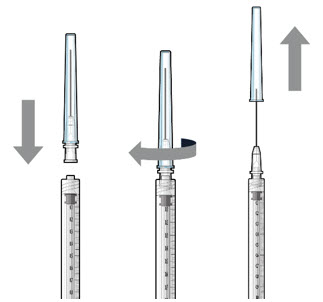 Figure 1 |
Do notuse the SUSVIMO initial fill needle for this step.
| ||
Step 3: Remove Air from Syringe
| 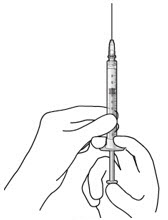 Figure2 | |
| Step 4: Attach SUSVIMO Initial Fill Needle Do notuse the filter needle to fill the implant.
| 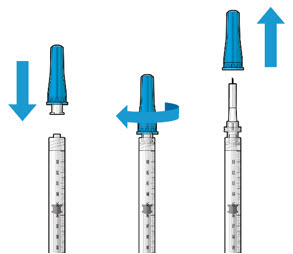 Figure 3 | |
Step 5: Remove Any Remaining Air from Syringe
| 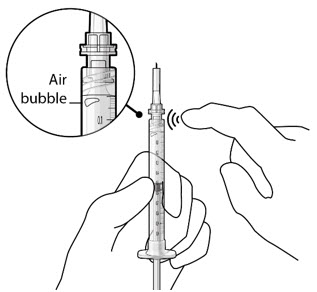 Figure 4 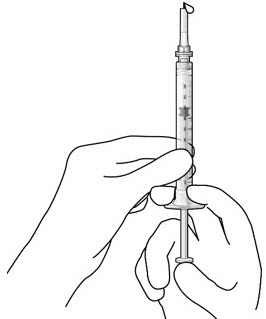 Figure 5 | |
| Note: | It is important to preserve as much drug as possible in order to completely fill the implant. | |
Step 6: Inspect the Syringe for Air Bubbles
| 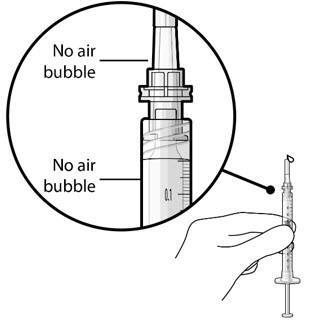 Figure 6 | |
| Note: | Use the syringe within 15 minutesof removing all air to avoid ranibizumab drying in the needle and impeding fluid flow. Do not use the initial fill needle if the needle is clogged. | |
| Step 7: Load Syringe into the Carrier Do not hold or push on the plunger rod of the syringe while inserting the needle into the implant septum.
| 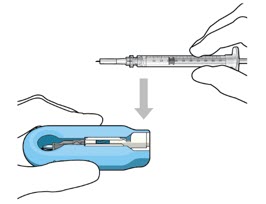 Figure 7: Align and lower the syringe into the carrier 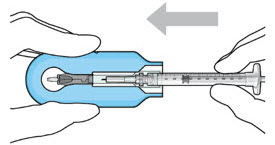 Figure 8: Push the syringe into the carrier 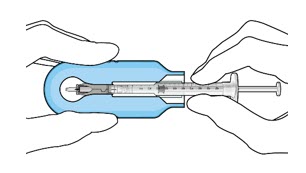 Figure 9: Syringe with initial fill needle inserted through the implant septum | |
Step 8: Fill Ocular Implant with SUSVIMO (ranibizumab injection) Under Microscope
| 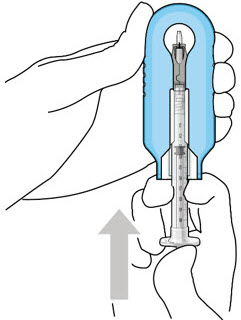 Figure 10: Administer ranibizumab into the implant 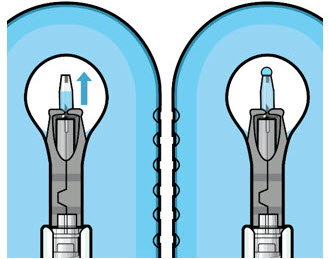 Figure 11: Dome of drug solution forms at tip of implant as viewed under magnification | |
| Note: | When filling the ocular implant, drug solution should only exit the ocular implant from the release control element. If drug solution is leaking from the implant at a different location, such as the side of the implant, do not use the ocular implant. If fluid is leaking from the septum at the needle insertion site, the needle may not be fully penetrating the implant septum. Fully push the syringe forward before continuing to fill the ocular implant. | |
| ||
Step 9: Inspect the Filled Ocular Implant Under the Microscope
| 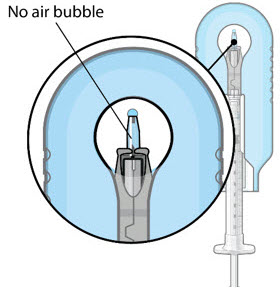 Figure 12: Proper appearance of implant after initial filling with ranibizumab | |
| Note: | Minimize air bubbles within the implant reservoir as they may cause slower drug release. If an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed, do not use the ocular implant. | |
| Note: | No more than 30 minutes should pass between the initial fill of the implant and the insertion into the patient's eye to ensure that the release control element remains saturated with SUSVIMO (ranibizumab injection). If SUSVIMO (ranibizumab injection) dries in the release control element, the implant may not release the drug properly into the vitreous after insertion. | |
Step 10: Remove the Syringe and Guide Sleeve from the Carrier
| 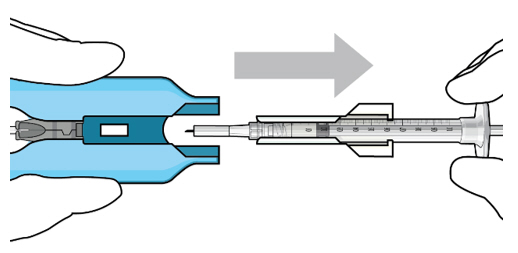 Figure 13: Remove the syringe and guide sleeve from the insertion tool carrier | |
| Step 11: Slide the Insertion Tool Handle into the Carrier | 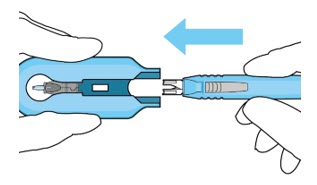 Figure 14: Insert the handle into the insertion tool carrier 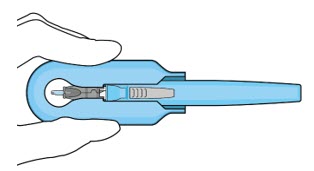 Figure 15: Fully inserted handle | |
| Note: | Do not withdraw the handle and implant until the eye is ready for insertion. Contact between the implant and any surface or object – even within the sterile field – may result in the introduction of a foreign body into the vitreous. | |
Ocular Implant Insertion
SUSVIMO ocular implant insertion is a surgical procedure that is performed in an operating room. The procedure must be performed under aseptic conditions by a physician experienced in vitreoretinal surgery [see Dosage and Administration (2.1) ] .
The ocular implant is filled with SUSVIMO (ranibizumab injection) immediately prior to insertion. No more than 30 minutes should pass between the initial fill of the ocular implant and the insertion into the patient's eye.
After placing an infusion line in the eye, create at least a 6×6 mm peritomy of the conjunctiva and Tenon's capsule centered around the selected SUSVIMO implant location in the supero-temporal quadrant. Perform careful conjunctival incision, hemostasis of the underlying sclera, and generous undermining of Tenon's capsule. Using aseptic technique, fill the ocular implant [see Dosage and Administration (2.5) ] . Using an MVR blade, create a full thickness dissection of the sclera 4 mm from the limbus until the pars plana is fully visible, with final target scleral incision length of 3.5 mm. Using a 532 nm laser endoprobe, apply contiguous, overlapping laser spots starting at 300 mW 1000 ms along the full length of the exposed pars plana and repeat until complete ablation is achieved. Pass a 3.2 mm slit knife perpendicularly through the center of the scleral dissection to open the underlying pars plana. Use the insertion tool to slowly insert the SUSVIMO implant into the sclero-pars plana incision perpendicular to the globe, ensuring that the long axis of the implant flange is properly aligned with the sclero-pars plana incision. Using the closed gripper tips of the insertion tool, seat the implant flush against the sclera. Clean any residual vitreous around the implant flange using a vitrector. Suture both Tenon's capsule and conjunctiva, using scleral anchoring at the apex of the peritomy, ensuring complete coverage of the implant flange. Refer to the complete SUSVIMO Instructions for Use for the initial fill and implant procedure included in the insertion tool assembly carton for further details.
Ocular Implant Removal
Removal of the SUSVIMO ocular implant is a surgical procedure that is performed in an operating room. The procedure must be performed under aseptic conditions by a physician experienced in vitreoretinal surgery [see Dosage and Administration (2.1) ] .
After placing an infusion line in the eye, create at least a 6×6 mm peritomy of the conjunctiva and Tenon's capsule around the SUSVIMO ocular implant flange. Remove any fibrous capsule or scar tissue that may have formed over the implant flange and septum using scalpel and forceps. With the explant tool oriented perpendicular to the globe, align the contoured tips with the long axis of the implant flange and grasp underneath the implant flange. Once the implant is secured in the explant tool, pull the implant from the eye in a perpendicular motion. Clear any vitreous prolapse present within or around the scleral wound using a vitrector. Completely close the scleral incision with multiple non-absorbable sutures. Close the Tenon's capsule and conjunctiva to completely cover the scleral incision. Refer to the complete Instructions for Use for the implant removal procedure included in the explant tool carton for further details.
Ocular Implant Refill-Exchange Procedure
The SUSVIMO ocular implant refill-exchange procedure must be performed under strict aseptic conditions by a physician experienced in ophthalmic surgery [see Dosage and Administration (2.1) ] . This includes the use of a surgical mask, sterile gloves, and a lid speculum.
Prior to and after the refill-exchange procedure, perform a dilated slit lamp exam and/or dilated indirect ophthalmoscopy to inspect the implant in the vitreous cavity through the pupil to identify if dislodgement of the implant septum has occurred [ see Figure 33 and Warnings and Precautions (5.4) ]. If the septum has dislodged, any further refill-exchange procedures should not be performed because normal device functioning cannot be assured. Discontinue treatment with SUSVIMO (ranibizumab injection) following septum dislodgement and consider implant removal should the benefit of the removal procedure outweigh the risk.
Step 1: Gather the supplies needed.
| ||
Step 2: Inspect Packaging and Components
|  Figure 16 Figure 16 | |
Step 3: Inspect SUSVIMO (ranibizumab injection)
| ||
Step 4: Patient Preparation
| ||
| Step 5: Transfer Dose from Vial to Syringe | 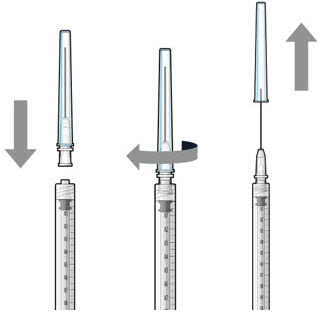 Figure 17 | |
| Note: | Use the filter needle to withdraw SUSVIMO (ranibizumab injection) from the vial. | |
Do not use the SUSVIMO refill needle for this step.
| ||
Step 6: Remove Air from Syringe
| 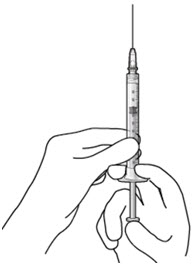 Figure 18 | |
| Step 7: Attach SUSVIMO Refill Needle Do not use the filter needle to fill the implant.
| 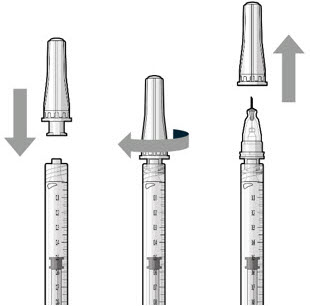 Figure 19 Figure 19 | |
Step 8: Remove Any Remaining Air from Syringe and Adjust Drug Dose
| 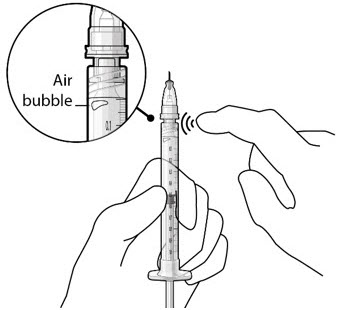 Figure 20 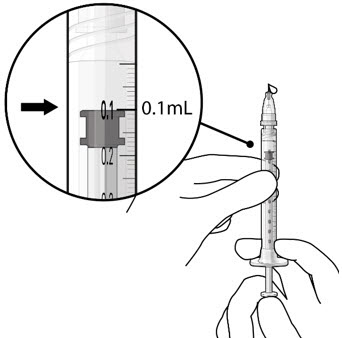 Figure 21 | |
| Step 9: Inspect the Syringe for Air Bubbles |  Figure 22 | |
| Note: | Ensure no air bubbles are present in the syringe and needle hub. Air injected into the implant could result in slower drug release. | |
| ||
| Note: | Use the syringe within 15 minutesof removing all air and adjusting the drug dose to avoid drug solution drying in the needle and impeding fluid flow. Do not use the refill needle or syringe if the needle is clogged. | |
| Step 10: Stabilize the globe and orient the refill needle | 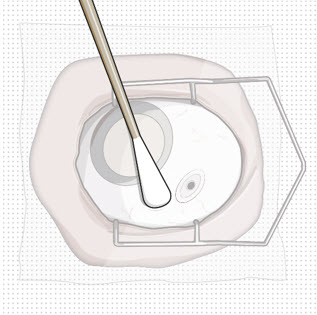 Figure 23 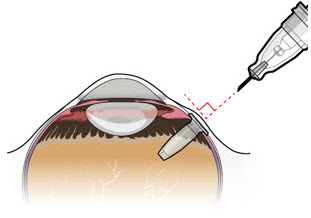 Figure 24 Figure 24 | |
| Note: | Perform the refill-exchange procedure using magnification (e.g., loupes, reading glasses, magnifiers) for visual assistance. | |
| ||
| Step 11: Insert the Refill Needle | 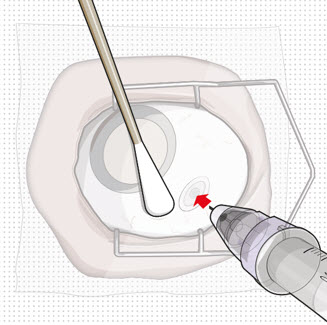 Figure 25 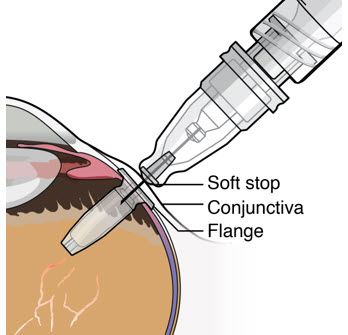 Figure 26 Figure 26 | |
| Note: | Insert needle at the very center of the implant septum and perpendicular to the implant to ensure the needle inserts fully. Do not maneuver if there is resistance as it will bend the needle. | |
Do not use a bent refill needle; replace if bent or if damage is suspected.
| ||
Step 12: Refill the SUSVIMO Implant
| 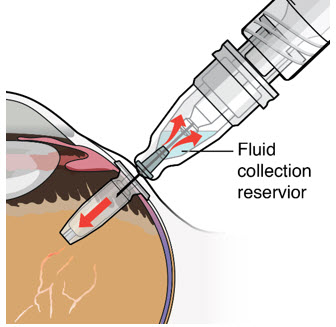 Figure 27 | |
Step 13: Withdraw the Syringe
| 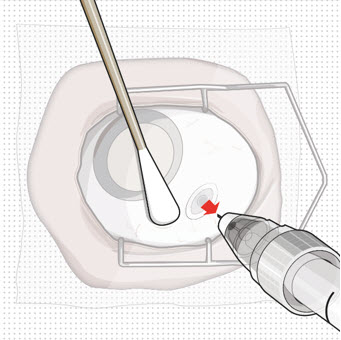 Figure 28 | |
Step 14: Dispose of the Used Components
| ||
Step 15: Perform Indirect Ophthalmoscopy
| ||
Delayed or Missed Doses
For patients with AMD or DME, if a planned dose (refill-exchange) of SUSVIMO (ranibizumab injection) is missed, it should be administered as soon as possible and the subsequent refill-exchange procedures should be performed 24 weeks (approximately 6 months) thereafter.
For patients with DR, if a planned dose (refill-exchange) of SUSVIMO (ranibizumab injection) is missed, it should be administered as soon as possible and the subsequent refill-exchange procedures should be performed 36 weeks (approximately 9 months) thereafter.
Dosage (Refill-Exchange) Modifications for Adverse Reactions
Table 1 describes dosage modifications for specific adverse Reactions [see Warnings and Precautions (5) ] . No dosage reductions for SUSVIMO are recommended.
| Adverse Reactions | Dosage Modification |
|---|---|
| Intraocular inflammation ≥ 1 + cells or flare | Withhold dose (refill-exchange) |
| Sight threatening events (e.g., rhegmatogenous retinal detachment, vitreous hemorrhage, unexplained vision loss, etc.) | Withhold dose (refill-exchange) |
| Local infections of either eye | Withhold dose (refill-exchange) |
| Infectious endophthalmitis | Withhold dose (refill-exchange) |
| Severe systemic infection | Withhold dose (refill-exchange) |
| Observed damage to the implant | Withhold dose (refill-exchange) and consider SUSVIMO implant removal [see Dosage and Administration (2.8 , 2.9) ] . |

Susvimo prescribing information
WARNING: ENDOPHTHALMITIS
The SUSVIMO implant has been associated with an up to 3-fold higher rate of endophthalmitis than monthly intravitreal injections of ranibizumab. Many of these events were associated with conjunctival retractions or erosions. Appropriate conjunctiva management and early detection with surgical repair of conjunctival retractions or erosions may reduce the risk of endophthalmitis. [see Contraindications (4.1) , Warnings and Precautions (5.1) ] .
INDICATIONS AND USAGE
SUSVIMO (ranibizumab injection) is a vascular endothelial growth factor (VEGF) inhibitor indicated for the treatment of patients with:
- Neovascular (wet) Age-related Macular Degeneration (AMD) who have previously responded to at least two intravitreal injections of a VEGF inhibitor (1.1 ).
- Diabetic Macular Edema (DME) who have previously responded to at least two intravitreal injections of a VEGF inhibitor (1.2 ).
- Diabetic Retinopathy (DR) who have previously responded to at least two intravitreal injections of a VEGF inhibitor (1.3 ).
Neovascular (wet) Age-related Macular Degeneration (AMD)
SUSVIMO (ranibizumab injection) is indicated for the treatment of patients with Neovascular (wet) Age-related Macular Degeneration (AMD) who have previously responded to at least two intravitreal injections of a Vascular Endothelial Growth Factor (VEGF) inhibitor medication.
Diabetic Macular Edema (DME)
SUSVIMO (ranibizumab injection) is indicated for the treatment of patients with Diabetic Macular Edema (DME) who have previously responded to at least two intravitreal injections of a Vascular Endothelial Growth Factor (VEGF) inhibitor medication.
Diabetic Retinopathy (DR)
SUSVIMO (ranibizumab injection) is indicated for the treatment of patients with Diabetic Retinopathy (DR) who have previously responded to at least two intravitreal injections of a Vascular Endothelial Growth Factor (VEGF) inhibitor medication.
DOSAGE AND ADMINISTRATION
- For intravitreal use via SUSVIMO ocular implant. (2.1 )
- Neovascular (wet) Age-related Macular Degeneration (AMD) and Diabetic Macular Edema (DME) The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO implant with refills every 24 weeks (approximately 6 months). (2.2 )
- Diabetic Retinopathy (DR) The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO implant with refills every 36 weeks (approximately 9 months). (2.3 )
- Supplemental treatment with 0.5 mg intravitreal ranibizumab injection may be administered in the affected eye if clinically necessary. (2.4 )
- Perform the initial implantation, refill-exchange, and implant removal (if necessary) procedures under strict aseptic conditions. (2.5 , 2.6 , 2.7 , 2.8 )
General Information
For Intravitreal Use via SUSVIMO ocular implant.
The SUSVIMO initial fill and ocular implant insertion and implant removal procedures must be performed under aseptic conditions by a physician experienced in vitreoretinal surgery. The SUSVIMO ocular implant must be surgically implanted in the eye or removed from the eye (if medically necessary) in an operating room using aseptic technique. See SUSVIMO Instructions for Use and the standardized steps to optimize surgical outcomes.
SUSVIMO refill-exchange procedures must be performed under aseptic conditions by a physician experienced in ophthalmic surgery [see Dosage and Administration (2.7) ] .
Do not administer SUSVIMO (ranibizumab injection) as a bolus intravitreal injection. Do not substitute SUSVIMO (ranibizumab injection) with other ranibizumab products.
Initial Fill : One SUSVIMO initial fill needle (34-gauge, with integrated 5 μm filter and blue cap) is included. A 5-micron sterile filter needle (19-gauge × 1½ inch), and a 1 mL Luer lock syringe are needed but not included .
Refill-Exchange : One SUSVIMO refill needle (34-gauge with integrated 5 μm filter and clear cap) is included. A 5-micron sterile filter needle (19-gauge × 1½ inch), and a 1 mL Luer lock syringe are needed but not included .
Neovascular (Wet) Age-Related Macular Degeneration (AMD) and Diabetic Macular Edema (DME)
The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO ocular implant with refills administered every 24 weeks (approximately 6 months).
Diabetic Retinopathy (DR)
The recommended dose of SUSVIMO (ranibizumab injection) is 2 mg (0.02 mL of 100 mg/mL solution) continuously delivered via the SUSVIMO ocular implant with refills administered every 36 weeks (approximately 9 months).
Supplemental Treatment with Intravitreal Ranibizumab Injection
Supplemental treatment with 0.5 mg (0.05 mL of 10 mg/mL) intravitreal ranibizumab injection may be administered in the affected eye while the SUSVIMO implant is in place and if clinically necessary [see Clinical Studies (14) ] .
Ocular Implant Initial Fill
The implant initial fill procedure must be performed by a physician experienced in vitreoretinal surgery [see Dosage and Administration (2.1) ]. The implant will be filled using aseptic technique with 0.02 mL of SUSVIMO (ranibizumab injection) prior to insertion of the implant into the patient's eye [see Dosage and Administration (2.6) ].
Refer to the complete SUSVIMO Instructions for Use for the initial fill and implant procedure included in the insertion tool assembly carton for further details.
Use aseptic technique to carry out the following preparation steps prior to insertion of the ocular implant into the patient's eye:
Step 1: Gather the supplies needed.
| ||
| Step 2: Transfer Dose from Vial to Syringe | ||
| Note: | Use the filter needle (not included) to withdraw SUSVIMO (ranibizumab injection) from the vial. |  Figure 1 Figure 1 |
Do notuse the SUSVIMO initial fill needle for this step.
| ||
Step 3: Remove Air from Syringe
|  Figure2 Figure2 | |
Step 4: Attach SUSVIMO Initial Fill Needle Do notuse the filter needle to fill the implant.
|  Figure 3 Figure 3 | |
Step 5: Remove Any Remaining Air from Syringe
|  Figure 4 Figure 4  Figure 5 Figure 5 | |
| Note: | It is important to preserve as much drug as possible in order to completely fill the implant. | |
Step 6: Inspect the Syringe for Air Bubbles
|  Figure 6 Figure 6 | |
| Note: | Use the syringe within 15 minutesof removing all air to avoid ranibizumab drying in the needle and impeding fluid flow. Do not use the initial fill needle if the needle is clogged. | |
Step 7: Load Syringe into the Carrier Do not hold or push on the plunger rod of the syringe while inserting the needle into the implant septum.
|  Figure 7: Align and lower the syringe into the carrier Figure 7: Align and lower the syringe into the carrier  Figure 8: Push the syringe into the carrier Figure 8: Push the syringe into the carrier  Figure 9: Syringe with initial fill needle inserted through the implant septum Figure 9: Syringe with initial fill needle inserted through the implant septum | |
Step 8: Fill Ocular Implant with SUSVIMO (ranibizumab injection) Under Microscope
|  Figure 10: Administer ranibizumab into the implant Figure 10: Administer ranibizumab into the implant  Figure 11: Dome of drug solution forms at tip of implant as viewed under magnification Figure 11: Dome of drug solution forms at tip of implant as viewed under magnification | |
| Note: | When filling the ocular implant, drug solution should only exit the ocular implant from the release control element. If drug solution is leaking from the implant at a different location, such as the side of the implant, do not use the ocular implant. If fluid is leaking from the septum at the needle insertion site, the needle may not be fully penetrating the implant septum. Fully push the syringe forward before continuing to fill the ocular implant. | |
| ||
Step 9: Inspect the Filled Ocular Implant Under the Microscope
|  Figure 12: Proper appearance of implant after initial filling with ranibizumab Figure 12: Proper appearance of implant after initial filling with ranibizumab | |
| Note: | Minimize air bubbles within the implant reservoir as they may cause slower drug release. If an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed, do not use the ocular implant. | |
| Note: | No more than 30 minutes should pass between the initial fill of the implant and the insertion into the patient's eye to ensure that the release control element remains saturated with SUSVIMO (ranibizumab injection). If SUSVIMO (ranibizumab injection) dries in the release control element, the implant may not release the drug properly into the vitreous after insertion. | |
Step 10: Remove the Syringe and Guide Sleeve from the Carrier
|  Figure 13: Remove the syringe and guide sleeve from the insertion tool carrier Figure 13: Remove the syringe and guide sleeve from the insertion tool carrier | |
| Step 11: Slide the Insertion Tool Handle into the Carrier |  Figure 14: Insert the handle into the insertion tool carrier Figure 14: Insert the handle into the insertion tool carrier  Figure 15: Fully inserted handle Figure 15: Fully inserted handle | |
| Note: | Do not withdraw the handle and implant until the eye is ready for insertion. Contact between the implant and any surface or object – even within the sterile field – may result in the introduction of a foreign body into the vitreous. | |
Ocular Implant Insertion
SUSVIMO ocular implant insertion is a surgical procedure that is performed in an operating room. The procedure must be performed under aseptic conditions by a physician experienced in vitreoretinal surgery [see Dosage and Administration (2.1) ] .
The ocular implant is filled with SUSVIMO (ranibizumab injection) immediately prior to insertion. No more than 30 minutes should pass between the initial fill of the ocular implant and the insertion into the patient's eye.
After placing an infusion line in the eye, create at least a 6×6 mm peritomy of the conjunctiva and Tenon's capsule centered around the selected SUSVIMO implant location in the supero-temporal quadrant. Perform careful conjunctival incision, hemostasis of the underlying sclera, and generous undermining of Tenon's capsule. Using aseptic technique, fill the ocular implant [see Dosage and Administration (2.5) ] . Using an MVR blade, create a full thickness dissection of the sclera 4 mm from the limbus until the pars plana is fully visible, with final target scleral incision length of 3.5 mm. Using a 532 nm laser endoprobe, apply contiguous, overlapping laser spots starting at 300 mW 1000 ms along the full length of the exposed pars plana and repeat until complete ablation is achieved. Pass a 3.2 mm slit knife perpendicularly through the center of the scleral dissection to open the underlying pars plana. Use the insertion tool to slowly insert the SUSVIMO implant into the sclero-pars plana incision perpendicular to the globe, ensuring that the long axis of the implant flange is properly aligned with the sclero-pars plana incision. Using the closed gripper tips of the insertion tool, seat the implant flush against the sclera. Clean any residual vitreous around the implant flange using a vitrector. Suture both Tenon's capsule and conjunctiva, using scleral anchoring at the apex of the peritomy, ensuring complete coverage of the implant flange. Refer to the complete SUSVIMO Instructions for Use for the initial fill and implant procedure included in the insertion tool assembly carton for further details.
Ocular Implant Removal
Removal of the SUSVIMO ocular implant is a surgical procedure that is performed in an operating room. The procedure must be performed under aseptic conditions by a physician experienced in vitreoretinal surgery [see Dosage and Administration (2.1) ] .
After placing an infusion line in the eye, create at least a 6×6 mm peritomy of the conjunctiva and Tenon's capsule around the SUSVIMO ocular implant flange. Remove any fibrous capsule or scar tissue that may have formed over the implant flange and septum using scalpel and forceps. With the explant tool oriented perpendicular to the globe, align the contoured tips with the long axis of the implant flange and grasp underneath the implant flange. Once the implant is secured in the explant tool, pull the implant from the eye in a perpendicular motion. Clear any vitreous prolapse present within or around the scleral wound using a vitrector. Completely close the scleral incision with multiple non-absorbable sutures. Close the Tenon's capsule and conjunctiva to completely cover the scleral incision. Refer to the complete Instructions for Use for the implant removal procedure included in the explant tool carton for further details.
Ocular Implant Refill-Exchange Procedure
The SUSVIMO ocular implant refill-exchange procedure must be performed under strict aseptic conditions by a physician experienced in ophthalmic surgery [see Dosage and Administration (2.1) ] . This includes the use of a surgical mask, sterile gloves, and a lid speculum.
Prior to and after the refill-exchange procedure, perform a dilated slit lamp exam and/or dilated indirect ophthalmoscopy to inspect the implant in the vitreous cavity through the pupil to identify if dislodgement of the implant septum has occurred [ see Figure 33 and Warnings and Precautions (5.4) ]. If the septum has dislodged, any further refill-exchange procedures should not be performed because normal device functioning cannot be assured. Discontinue treatment with SUSVIMO (ranibizumab injection) following septum dislodgement and consider implant removal should the benefit of the removal procedure outweigh the risk.
Step 1: Gather the supplies needed.
| ||
Step 2: Inspect Packaging and Components
|  Figure 16 Figure 16 | |
Step 3: Inspect SUSVIMO (ranibizumab injection)
| ||
Step 4: Patient Preparation
| ||
| Step 5: Transfer Dose from Vial to Syringe |  Figure 17 Figure 17 | |
| Note: | Use the filter needle to withdraw SUSVIMO (ranibizumab injection) from the vial. | |
Do not use the SUSVIMO refill needle for this step.
| ||
Step 6: Remove Air from Syringe
|  Figure 18 Figure 18 | |
Step 7: Attach SUSVIMO Refill Needle Do not use the filter needle to fill the implant.
|  Figure 19 Figure 19 | |
Step 8: Remove Any Remaining Air from Syringe and Adjust Drug Dose
|  Figure 20 Figure 20  Figure 21 Figure 21 | |
| Step 9: Inspect the Syringe for Air Bubbles |  Figure 22 Figure 22 | |
| Note: | Ensure no air bubbles are present in the syringe and needle hub. Air injected into the implant could result in slower drug release. | |
| ||
| Note: | Use the syringe within 15 minutesof removing all air and adjusting the drug dose to avoid drug solution drying in the needle and impeding fluid flow. Do not use the refill needle or syringe if the needle is clogged. | |
| Step 10: Stabilize the globe and orient the refill needle |  Figure 23 Figure 23  Figure 24 Figure 24 | |
| Note: | Perform the refill-exchange procedure using magnification (e.g., loupes, reading glasses, magnifiers) for visual assistance. | |
| ||
| Step 11: Insert the Refill Needle |  Figure 25 Figure 25  Figure 26 Figure 26 | |
| Note: | Insert needle at the very center of the implant septum and perpendicular to the implant to ensure the needle inserts fully. Do not maneuver if there is resistance as it will bend the needle. | |
Do not use a bent refill needle; replace if bent or if damage is suspected.
| ||
Step 12: Refill the SUSVIMO Implant
|  Figure 27 Figure 27 | |
Step 13: Withdraw the Syringe
|  Figure 28 Figure 28 | |
Step 14: Dispose of the Used Components
| ||
Step 15: Perform Indirect Ophthalmoscopy
| ||
Delayed or Missed Doses
For patients with AMD or DME, if a planned dose (refill-exchange) of SUSVIMO (ranibizumab injection) is missed, it should be administered as soon as possible and the subsequent refill-exchange procedures should be performed 24 weeks (approximately 6 months) thereafter.
For patients with DR, if a planned dose (refill-exchange) of SUSVIMO (ranibizumab injection) is missed, it should be administered as soon as possible and the subsequent refill-exchange procedures should be performed 36 weeks (approximately 9 months) thereafter.
Dosage (Refill-Exchange) Modifications for Adverse Reactions
Table 1 describes dosage modifications for specific adverse Reactions [see Warnings and Precautions (5) ] . No dosage reductions for SUSVIMO are recommended.
| Adverse Reactions | Dosage Modification |
|---|---|
| Intraocular inflammation ≥ 1 + cells or flare | Withhold dose (refill-exchange) |
| Sight threatening events (e.g., rhegmatogenous retinal detachment, vitreous hemorrhage, unexplained vision loss, etc.) | Withhold dose (refill-exchange) |
| Local infections of either eye | Withhold dose (refill-exchange) |
| Infectious endophthalmitis | Withhold dose (refill-exchange) |
| Severe systemic infection | Withhold dose (refill-exchange) |
| Observed damage to the implant | Withhold dose (refill-exchange) and consider SUSVIMO implant removal [see Dosage and Administration (2.8 , 2.9) ] . |
DOSAGE FORMS AND STRENGTHS
Injection: 100 mg/mL, clear to slightly opalescent, colorless to slightly brownish solution in a single-dose vial
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no adequate and well-controlled studies of SUSVIMO (ranibizumab injection) administration in pregnant women. Administration of ranibizumab to pregnant monkeys throughout the period of organogenesis resulted in a low incidence of skeletal abnormalities at intravitreal doses up to 41 times the human exposure (based on serum levels following the recommended clinical dose). No skeletal abnormalities were observed at serum trough levels similar to the human exposure after a single eye treatment at the recommended clinical dose ( see Data ) .
Animal reproduction studies are not always predictive of human response, and it is not known whether ranibizumab can cause fetal harm when administered to a pregnant woman. Based on the anti-VEGF mechanism of action for ranibizumab [see Clinical Pharmacology (12.1) ] , treatment with SUSVIMO (ranibizumab injection) may pose a risk to human embryofetal development.
All pregnancies have a background risk of birth defects, loss, and other adverse outcomes. The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects is 2% – 4% and of miscarriage is 15% – 20% of clinically recognized pregnancies.
Data
Animal Data
An embryo-fetal developmental toxicity study was performed on pregnant cynomolgus monkeys. Pregnant animals received intravitreal injections of ranibizumab every 14 days starting on Day 20 of gestation, until Day 62 at doses of 0, 0.125, and 1 mg/eye. Skeletal abnormalities including incomplete and/or irregular ossification of bones in the skull, vertebral column, and hindlimbs and shortened supernumerary ribs were seen at a low incidence in fetuses from animals treated with 1 mg/eye of ranibizumab. The 1 mg/eye dose resulted in trough serum ranibizumab levels up to 41 times higher than observed human C max levels of SUSVIMO (ranibizumab injection) after treatment of a single eye.
No skeletal abnormalities were seen at the lower dose of 0.125 mg/eye, a dose which resulted in trough exposures similar to single eye treatment with SUSVIMO (ranibizumab injection) in humans. No effect on the weight or structure of the placenta, maternal toxicity, or embryotoxicity was observed.
Lactation
Risk Summary
There are no data available on the presence of ranibizumab in human milk, the effects of ranibizumab on the breastfed infant or the effects of ranibizumab on milk production/excretion. Because many drugs are excreted in human milk, and because the potential for absorption and harm to infant growth and development exists, caution should be exercised when SUSVIMO is administered to a nursing woman.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for SUSVIMO (ranibizumab injection) and any potential adverse effects on the breastfed child from ranibizumab.
Females and Males of Reproductive Potential
Contraception
Females of reproductive potential should use effective contraception during treatment with SUSVIMO (ranibizumab injection) and for at least 12 months after the last dose of SUSVIMO (ranibizumab injection).
Infertility
No studies on the effects of ranibizumab on fertility have been conducted and it is not known whether ranibizumab can affect reproduction capacity. Based on the anti-VEGF mechanism of action for ranibizumab, treatment with SUSVIMO (ranibizumab injection) may pose a risk to reproductive capacity.
Pediatric Use
The safety and efficacy of SUSVIMO (ranibizumab injection) in pediatric patients have not been established.
Geriatric Use
In the Archway study, 90% (222 of 248) of the patients randomized to treatment with SUSVIMO were ≥ 65 years old and approximately 57% (141 of 248) were ≥ 75 years old. No notable difference in treatment effect or safety was seen with increasing age.
CONTRAINDICATIONS
Ocular or Periocular Infections
SUSVIMO (ranibizumab injection) is contraindicated in patients with ocular or periocular infections.
Active Intraocular Inflammation
SUSVIMO (ranibizumab injection) is contraindicated in patients with active intraocular inflammation.
Hypersensitivity
SUSVIMO (ranibizumab injection) is contraindicated in patients with known hypersensitivity to ranibizumab products or any of the excipients in SUSVIMO (ranibizumab injection).
WARNINGS AND PRECAUTIONS
The SUSVIMO implant and/or implant-related procedures have been associated with endophthalmitis, rhegmatogenous retinal detachment, implant dislocation, septum dislodgement, vitreous hemorrhage, conjunctival erosion, conjunctival retraction, and conjunctival blebs. Patients should be instructed to report any signs or symptoms that could be associated with these events without delay. In some cases, these events can present asymptomatically. The implant and the tissue overlying the implant flange should be monitored routinely following the implant insertion, and refill-exchange procedures to permit early medical or surgical intervention as necessary. Special precautions need to be taken when handling SUSVIMO components [see How Supplied/Storage and Handling (16.3) ].
Endophthalmitis
In the active comparator period of controlled clinical trials in AMD, the ranibizumab implant has been associated with a 3-fold higher rate of endophthalmitis than monthly intravitreal injections of ranibizumab (1.7% in the SUSVIMO arm vs 0.5% in the intravitreal arm). When including extension phases of clinical trials, 2% (11/555) of patients receiving the ranibizumab implant experienced an episode of endophthalmitis. Reports occurred between days 5 and 853, with a median of 173 days. Many, but not all, of the cases of endophthalmitis reported a preceding or concurrent conjunctival retraction or erosion event.
In the active comparator period of the controlled clinical trial in DME, 0% of patients in the SUSVIMO arm compared to 0.3% in the intravitreal arm experienced an episode of endophthalmitis. When including the extension phase of the clinical trial, 0.7% (4/556) of patients receiving the ranibizumab implant experienced an episode of endophthalmitis. Reports occurred between days 625 and 1016, with a median of 824 days.
In the period with an observational comparator arm of the clinical trial in DR, there were no patients (0/105) in the SUSVIMO arm who experienced an episode of endophthalmitis [see Clinical Studies (14.3) ] . When including the extension phase of the clinical trial 0.8% (1/128) patients receiving the ranibizumab implant experienced an episode of endophthalmitis, with the event reported on day 695.
Endophthalmitis should be treated promptly in an effort to reduce the risk of vision loss and maximize recovery. The SUSVIMO (ranibizumab injection) dose (refill-exchange) should be delayed until resolution of endophthalmitis [see Dosage and Administration (2.10) and Adverse Reactions (6.1) ] .
Patients should not have an active or suspected ocular or periocular infection or severe systemic infection at the time of any SUSVIMO implant or refill procedure. Appropriate intraoperative handling followed by secure closure of the conjunctiva and Tenon's capsule, and early detection and surgical repair of conjunctival erosions or retractions and strict/controlled aseptic technique conditions throughout refill-exchange procedures may reduce the risk of endophthalmitis [see Dosage and Administration (2.1) and Warnings and Precautions (5.5) ] .
Rhegmatogenous Retinal Detachment
Rhegmatogenous retinal detachments have occurred in clinical trials of SUSVIMO and may result in vision loss. Rhegmatogenous retinal detachments should be promptly treated with an intervention (e.g., pneumatic retinopexy, vitrectomy, or laser photocoagulation). SUSVIMO (ranibizumab injection) dose (refill-exchange) should be delayed in the presence of a retinal detachment or retinal break [see Dosage and Administration (2.10) ] .
Careful evaluation of the retinal periphery is recommended to be performed, and any suspected areas of abnormal vitreo-retinal adhesion or retinal breaks should be treated before inserting the implant in the eye.
Implant Dislocation
In clinical trials, the device has dislocated/subluxated into the vitreous cavity or has extended outside the vitreous cavity into or beyond the subconjunctival space. Device dislocation requires urgent surgical intervention. Strict adherence to the scleral incision length and appropriate targeting of the pars plana during laser ablation may reduce the risk of implant dislocation.
Septum Dislodgement
In clinical trials, a type of implant damage where the septum has dislodged into the implant body has been reported. Perform a dilated slit lamp exam and/or dilated indirect ophthalmoscopy to inspect the implant in the vitreous cavity through the pupil prior to and after the refill-exchange procedure to identify if septum dislodgement has occurred. Discontinue treatment with SUSVIMO (ranibizumab injection) following septum dislodgement and consider implant removal should the benefit of the removal procedure outweigh the risk [ see Dosage and Administration (2.9) ]. The benefits and risks of retaining, removing, or removing and replacing an implant with a dislodged septum are not well characterized.
Appropriate handling and insertion of the refill needle into the septum (avoid twisting and/or rotation) is required to minimize the risk of septum dislodgement [ see Dosage and Administration (2.8) ].
Vitreous Hemorrhage
Vitreous hemorrhages may result in temporary vision loss. Vitrectomy may be needed in the case of a non-clearing vitreous hemorrhage [see Dosage and Administration (2.10) ] .
In clinical trials of SUSVIMO including the extension phases in patients with AMD, vitreous hemorrhages were reported in 5.2% (23/443) of patients receiving SUSVIMO.
In the clinical trial of SUSVIMO including the extension phases in patients with DME, vitreous hemorrhages were reported in 10.1% (56/556) of patients receiving SUSVIMO.
In the clinical trial of SUSVIMO including the extension phase in patients with DR, vitreous hemorrhages were reported in 9.4% (12/128) of patients receiving SUSVIMO.
The majority of these hemorrhages occurred within the first post-operative month following surgical implantation and the majority of vitreous hemorrhages resolved spontaneously.
Patients on antithrombotic medication (e.g., oral anticoagulants, aspirin, nonsteroidal anti-inflammatory drugs) may be at increased risk of vitreous hemorrhage. Antithrombotic medications are recommended to be temporarily interrupted prior to the implant insertion procedure. The SUSVIMO (ranibizumab injection) dose (refill-exchange) should be delayed in the event of sight-threatening vitreous hemorrhage.
The use of pars plana laser ablation and scleral cauterization should be performed to reduce the risk of vitreous hemorrhage.
Conjunctival Erosion or Retraction
A conjunctival erosion is a full thickness degradation or breakdown of the conjunctiva in the area of the implant flange. A conjunctival retraction is a recession or opening of the limbal and/or radial peritomy. Conjunctival erosions or retractions have been associated with an increased risk of endophthalmitis, especially if the implant becomes exposed. Surgical intervention (e.g., conjunctival/Tenon's capsule repair) is recommended to be performed in case of conjunctival erosion or retraction with or without exposure of the implant flange.
In clinical trials of SUSVIMO including the extension phases in patients with AMD, 3.6% (16/443) of patients receiving SUSVIMO reported conjunctival erosion and 1.6% (7/443) of patients receiving SUSVIMO reported conjunctival retraction in the study eye.
In the clinical trial of SUSVIMO including the extension phases of patients with DME, 2.2% (12/556) of patients receiving SUSVIMO reported conjunctival erosion and 1.3% (7/556) of patients receiving SUSVIMO reported conjunctival retraction in the study eye.
In the clinical trial of SUSVIMO including the extension phase in patients with DR, 2.3% (3/128) of patients receiving SUSVIMO reported conjunctival erosion and 1.6% (2/128) of patients receiving SUSVIMO reported conjunctival retraction in the study eye.
Appropriate intraoperative handling of conjunctiva and Tenon's capsule to preserve tissue integrity and secure closure of peritomy while ensuring placement of sutures away from implant edge may reduce the risk of conjunctival erosion or retraction. The implant and the tissue overlying the implant flange should be monitored routinely following the implant insertion.
Conjunctival Bleb
A conjunctival bleb is an encapsulated elevation of the conjunctiva above the implant flange, which may be secondary to subconjunctival thickening or fluid. Conjunctival blebs may require surgical management to avoid further complications, especially if the implant septum is no longer identifiable due to the conjunctival bleb.
In clinical trials of SUSVIMO including the extension phases in patients with AMD, 5.9% (26/443) of patients receiving SUSVIMO reported conjunctival bleb/conjunctival filtering bleb leak in the study eye.
In the clinical trial of SUSVIMO including the extension phases in patients with DME, 9% (50/556) of patients receiving SUSVIMO reported conjunctival bleb/conjunctival filtering bleb leak in the study eye.
In the clinical trial of SUSVIMO including the extension phase in patients with DR, 3.9% (5/128) of patients receiving SUSVIMO reported conjunctival bleb/conjunctival filtering bleb leak in the study eye.
Strict adherence to the scleral incision length, appropriate intraoperative handling of conjunctiva and Tenon's capsule to preserve tissue integrity and secure closure of peritomy, and proper seating of the refill needle during refill-exchange procedures may reduce the risk of conjunctival bleb.
Postoperative Decrease in Visual Acuity
Visual acuity was decreased by 4 letters on average in the first postoperative month and 2 letters on average in the second postoperative month following initial implantation of SUSVIMO in patients with AMD [see Clinical Studies (14.1) ] .
Visual acuity was decreased by 7 letters on average in the first postoperative month and 3 to 4 letters on average in the second postoperative month following initial implantation of SUSVIMO in patients with DME and DR [see Clinical Studies (14.2 and 14.3) ] .
Postoperative Intraocular Inflammation
Postoperative intraocular inflammation has occurred following SUSVIMO implantation. The majority of cases occurred during the first week following implantation and resolved within the first month.
Air Bubbles Causing Improper Filling of the Implant
Minimize air bubbles within the implant reservoir as they may cause slower drug release. During the initial fill procedure, if an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed after initial fill, do not use the implant. During the refill-exchange procedure, if excess air is present in the syringe and needle do not use the syringe and needle. If excess air bubbles are observed after the refill-exchange procedure, consider repeating the refill-exchange procedure.
Deflection or Movement of the Implant
Use caution when performing ophthalmic procedures (e.g., B-scan ophthalmic ultrasound, scleral depression, or gonioscopy) that may cause deflection or movement of the implant and subsequent injury.
ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the label:
- Endophthalmitis [see Warnings and Precautions (5.1) ]
- Rhegmatogenous Retinal Detachment [see Warnings and Precautions (5.2) ]
- Implant Dislocation [see Warnings and Precautions (5.3) ]
- Septum Dislodgement [see Warnings and Precautions (5.4) ]
- Vitreous Hemorrhage [see Warnings and Precautions (5.5) ]
- Conjunctival Erosion or Retraction [see Warnings and Precautions (5.6) ]
- Conjunctival Bleb [see Warnings and Precautions (5.7) ]
- Postoperative Decrease in Visual Acuity [see Warnings and Precautions (5.8) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in one clinical trial of a drug cannot be directly compared with rates in the clinical trials of the same or another drug and may not reflect the rates observed in practice.
The data below (Table 2 ) reflect exposure of 248 patients with AMD in the Archway study through Week 40, 320 patients with DME in the Pagoda study up to Week 64, and 105 patients with DR in the Pavillion study through Week 52 following the SUSVIMO initial fill and implant insertion, refill, and implant removal (if necessary) procedures.
In clinical trials of SUSVIMO in AMD patients, the most common (≥ 10%) adverse reactions up to Week 40 were conjunctival hemorrhage (72%), conjunctival hyperemia (26%), iritis (23%), and eye pain (10%). Septum dislodgement was reported in 0.4% of the AMD patient population.
In clinical trials of SUSVIMO in DME patients, patient population the most common (≥ 10%) adverse reactions up to Week 64 were conjunctival hemorrhage (62%), conjunctival hyperemia (15%), iritis (14%), eye pain (13%), cataract (11%), conjunctival disorder (10%) and vitreous hemorrhage (10%).
In clinical trials of SUSVIMO in DR patients, the most common (≥ 10%) adverse reactions up to Week 52 were conjunctival hemorrhage (73%), conjunctival disorder (14%), iritis (12%) and conjunctival hyperemia (11%).
| Adverse Reactions | AMD Week 40 | DME Week 64 | DR Week 52 | ||
|---|---|---|---|---|---|
| SUSVIMO n = 248 | Intravitreal ranibizumab n = 167 | SUSVIMO n = 320 | Intravitreal ranibizumab 0.5 mg n = 314 | SUSVIMO n = 105 | |
| Conjunctival hemorrhage | 72% | 6% | 62% | 18% | 73% |
| Conjunctival hyperemia | 26% | 2% | 15% | 0 | 11% |
| Iritis Iritis includes: iritis, anterior chamber flare, anterior chamber inflammation, and anterior chamber cell. | 23% | 0.6% | 14% | 2% | 12% |
| Eye pain | 10% | 5% | 13% | 6% | 9% |
| Conjunctival disorder Conjunctival disorder includes: conjunctival adhesion, conjunctival disorder, conjunctival edema, conjunctival erosion, conjunctival retraction, and subconjunctival fibrosis. | 9% | 0 | 10% | 0.3% | 14% |
| Vitreous floaters | 9% | 2% | 4% | 5% | 2% |
| Conjunctival bleb/ filtering bleb leak Conjunctival bleb/filtering bleb leak includes: conjunctival bleb, conjunctival filtering bleb leak, conjunctival cyst, subconjunctival cyst, and implant site cyst. | 8% | 0 | 8% | 0 | 2% |
| Foreign body sensation in eyes | 7% | 1% | 3% | 2% | 9% |
| Headache Headache includes: headache and procedural headache. | 6% | 2% | 6% | 4% | 9% |
| Hypotony of eye | 6% | 0 | 3% | 0 | 0 |
| Vitreous detachment | 6% | 5% | 8% | 5% | 9% |
| Vitreous hemorrhage | 5% | 2% | 10% | 2% | 6% |
| Cataract Cataract includes: cataract, cortical cataract, nuclear cataract, and subcapsular cataract. | 4% | 4% | 11% | 7% | 7% |
| Corneal disorder | 4% | 0 | 1% | 0 | 1% |
| Corneal abrasion Corneal abrasion includes: corneal abrasion and vital dye staining cornea present. | 4% | 0.6% | 4% | 1% | 4% |
| Corneal edema | 4% | 0 | 4% | 0.3% | 4% |
In clinical trials of SUSVIMO, hyphema was reported in 0.4% of AMD patients, 1.9% of DME patients, and 1.9% of DR patients.
DESCRIPTION
Ranibizumab is a recombinant humanized IgG1 kappa isotype monoclonal antibody fragment for intraocular use. Ranibizumab binds to and inhibits the biologic activity of human vascular endothelial growth factor-A (VEGF-A). Ranibizumab, which lacks an Fc region, has a molecular weight of approximately 48 kilodaltons and is produced by an E. coli expression system in a nutrient medium containing the antibiotic tetracycline. Tetracycline is not detectable in the final product.
SUSVIMO (ranibizumab injection) is supplied as a sterile, clear to slightly opalescent, colorless to slightly brownish solution for intravitreal use via the SUSVIMO implant. Each single-dose vial contains 10 mg of ranibizumab, histidine HCl (0.1 mg), polysorbate 20 (0.01 mg), sucrose (8.2 mg), and Water for Injection, in 0.1 mL of solution with a pH of 5.5. The SUSVIMO implant is designed to contain approximately 0.02 mL (2 mg) of ranibizumab solution when filled. SUSVIMO does not contain an antimicrobial preservative.
CLINICAL PHARMACOLOGY
Mechanism of Action
Ranibizumab binds to the receptor binding site of multiple biologically active forms of VEGF-A, including VEGF 110 . VEGF-A has been shown to cause neovascularization and leakage in models of ocular angiogenesis and vascular occlusion and is thought to contribute to pathophysiology of neovascular AMD. The binding of ranibizumab to VEGF-A prevents the interaction of VEGF-A with its receptors (VEGFR1 and VEGFR2) on the surface of endothelial cells, reducing endothelial cell proliferation, vascular leakage, and new blood vessel formation.
Pharmacokinetics
The SUSVIMO implant provides a continuous release of ranibizumab where the release of ranibizumab into the vitreous decreases over time as the concentration in the implant decreases. The ranibizumab serum and aqueous humor concentrations with a SUSVIMO 24-week and 36-week treatment interval are maintained below the maximum and above the minimum concentrations experienced with monthly 0.5 mg intravitreal ranibizumab.
Distribution
Ranibizumab did not accumulate in serum when administered with refills every 24 weeks or every 36 weeks.
Following implant insertion of SUSVIMO in patients with AMD, the mean (±SD) maximum ranibizumab serum concentration (C max ) was 0.48 (±0.17) ng/mL and median (range) time to maximum serum concentration (T max ) was 26 (1 – 89) days. Following the initial fill and refill of SUSVIMO in patients, maximum serum concentrations of ranibizumab were below the ranibizumab concentration necessary to inhibit the biological activity of VEGF by 50%.
Elimination
Metabolism
The metabolism of SUSVIMO (ranibizumab injection) has not been studied. SUSVIMO (ranibizumab injection) is a monoclonal antibody fragment and antibodies are cleared principally by catabolism.
Excretion
The full excretion profile for ranibizumab following administration of SUSVIMO is unknown.
Specific Populations
Patients with renal impairment were included in the population pharmacokinetic analysis of SUSVIMO (ranibizumab injection). Across all indications, systemic clearance of ranibizumab was slightly lower in renally impaired patients but was not clinically significant. No clinically significant differences in the pharmacokinetics of ranibizumab were observed based on age in the patient population.
Immunogenicity
As with all therapeutic proteins, there is potential for immune response in patients treated with ranibizumab including SUSVIMO. The detection of an immune response is highly dependent on the sensitivity, specificity, and drug tolerance level of the assay. Additionally, the observed incidence of antibody positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the study described below with the incidence of antibodies in other studies or to other products may be misleading.
Prior to treatment with SUSVIMO, 2.1-3.6% of patients with AMD, DME, or DR tested positive for anti-ranibizumab antibodies. In patients with AMD, after the SUSVIMO implant insertion and treatment, 12% (29 of 247) patients tested positive for anti-ranibizumab antibodies. In patients with DME, after 4 doses of intravitreal ranibizumab, and SUSVIMO implant insertion and treatment, 13% (41 of 320) patients tested positive for anti-ranibizumab antibodies. In patients with DR, after 2 doses of intravitreal ranibizumab, and SUSVIMO implant insertion and treatment, 17% (17 of 99) patients tested positive for anti-ranibizumab antibodies. No clinically meaningful differences in the pharmacokinetics, efficacy, or safety in patients testing positive for anti-ranibizumab antibodies were observed.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been conducted to determine the carcinogenic or mutagenic potential of SUSVIMO (ranibizumab injection). Based on the anti-VEGF mechanism of action for ranibizumab, treatment with SUSVIMO (ranibizumab injection) may pose a risk to reproductive capacity [see Females and Males of Reproductive Potential (8.3) ] .
CLINICAL STUDIES
Neovascular (wet) Age-related Macular Degeneration (AMD)
The clinical efficacy and safety of SUSVIMO (ranibizumab injection) was assessed in a randomized, visual assessor-masked, active treatment-controlled study (Archway-NCT03677934) in patients with AMD. A total of 415 patients (248 in the SUSVIMO arm and 167 in the intravitreal ranibizumab arm) were enrolled and treated in this study. Patients were diagnosed with AMD within the 9 months prior to screening and received ≥ 3 doses of anti-VEGF intravitreal agents in the study eye within the last 6 months prior to screening. Each patient was required to have demonstrated a response to an anti-VEGF intravitreal agent prior to randomization. Patients were randomized in a 3:2 ratio to receive continuous delivery of SUSVIMO (ranibizumab injection) via the SUSVIMO implant every 24 weeks or 0.5 mg intravitreal ranibizumab injections every 4 weeks. For patients randomized to the SUSVIMO arm, supplemental treatment with 0.5 mg intravitreal ranibizumab injections was available at Weeks 16, 20, 40, 44, 64, 68, 88, and 92, if needed. In the first 24 weeks, 1.6% of patients assessed for supplemental treatment received 1 or more supplemental treatment(s) and in the following 24 weeks, 5.4% of patients assessed for supplemental treatment received 1 or more supplemental treatment(s).
The primary efficacy endpoint of change from baseline in distance Best Corrected Visual Acuity (BCVA) score averaged over Week 36 and Week 40 demonstrated that SUSVIMO was equivalent to intravitreal ranibizumab injections administered every 4 weeks. Detailed efficacy results are shown in Table 3 and Figure 29 below.
| Outcome Measure BCVA measured using the Early Treatment Diabetic Retinopathy Study (ETDRS) visual acuity chart at a starting distance of 4 meters. | SUSVIMO (100 mg/mL) n=248 | Intravitreal ranibizumab 0.5 mg (10 mg/mL) n=167 | Difference (95% CI) All estimates are adjusted estimates based on a mixed-effect model with repeated measures. SUSVIMO arm - intravitreal ranibizumab arm. 95% is a rounding of 95.03% CI; The type 1 error was adjusted for interim safety monitoring. |
|---|---|---|---|
| BCVA = Best corrected visual acuity | |||
| Adjusted Mean change from baseline in BCVA score averaged over Weeks 36 and 40 | 0.2 | 0.5 | -0.3 (-1.7, 1.1) Equivalence margins were ±4.5 letters. |
| Q24W = every 24 weeks; Q4W = every 4 weeks |
| Figure 29 Adjusted Mean change from Baseline in Best Corrected Visual Acuity in study eye through Week 48 in the Archway (GR40548) study Prior to study treatment, a median of 4 doses of anti-VEGF intravitreal agents were administered in the study eye of patients in the SUSVIMO and intravitreal ranibizumab arms. , Decrease in BCVA at Week 4 during post-operative recovery period. |
|
Consistent results were observed across patient subgroup analyses for mean change from baseline in BCVA score (age, gender, number of prior anti-VEGF intravitreal injections, and baseline BCVA score).
Diabetic Macular Edema (DME)
The clinical efficacy and safety of SUSVIMO were assessed in a randomized, visual assessor-masked, active treatment-controlled study (Pagoda-NCT04108156) in patients with DME. A total of 634 patients (381 in the SUSVIMO arm and 253 in the intravitreal ranibizumab 0.5 mg arm) were enrolled and treated in this study. Patients were randomized in a 3:2 ratio to receive continuous delivery of SUSVIMO via the implant every 24 weeks or 0.5 mg intravitreal ranibizumab injections every 4 weeks.
Prior to study treatment, a median of 4 doses of intravitreal ranibizumab 0.5 mg were administered in the study eye of patients in the SUSVIMO and intravitreal ranibizumab arms. Patient ages ranged from 29 to 89 years with a mean of 60.7 years. A total of 21% of patients were previously treated for DME. At baseline, the overall mean visual acuity was 65.3 letters (range: 25 to 89 letters).
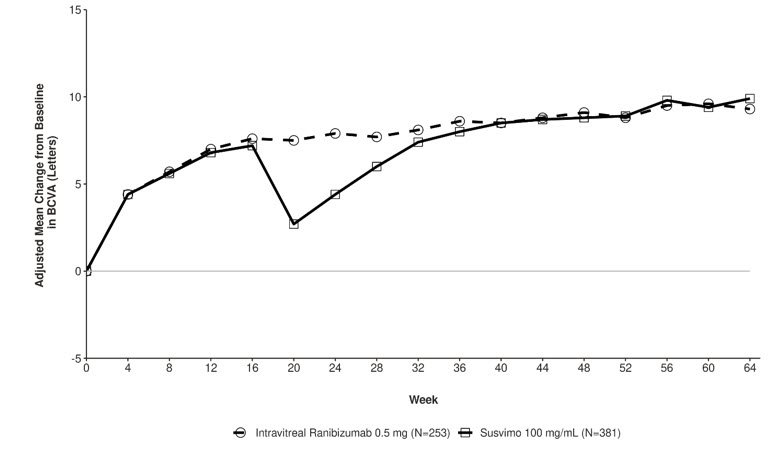
The primary efficacy endpoint of change from baseline in distance Best Corrected Visual Acuity (BCVA) score averaged over Week 60 and Week 64 demonstrated that SUSVIMO was non-inferior to intravitreal ranibizumab injections administered every 4 weeks. Detailed efficacy results are shown in Table 4 and Figure 30 below.
| Outcome Measure BCVA measured using the Early Treatment Diabetic Retinopathy Study (ETDRS) visual acuity chart at a starting distance of 4 meters. | SUSVIMO 100 mg/mL n=381 | Intravitreal ranibizumab 0.5 mg (10 mg/mL) n=253 | Difference (95% CI) All estimates are adjusted estimates based on a mixed-effect model with repeated measures. 95% is a rounding of 95.05% CI; The type 1 error was adjusted for interim safety monitoring. |
|---|---|---|---|
| BCVA = Best corrected visual acuity | |||
| Change in BCVA scores from baseline averaged over Week 60 and Week 64 Adjusted Mean | 9.6 | 9.4 | 0.2 (-1.2, 1.6) |
Figure 30 Adjusted Mean change from Baseline in Best Corrected Visual Acuity in study eye through Week 64 in the Pagoda (GR40550) study
Consistent results were observed across patient subgroup analyses for mean change from baseline in BCVA score (age, ethnicity, gender, baseline HbA1c score, focal/macular laser history, baseline BCVA score, prior intravitreal anti-VEGF treatment and DR severity).
Diabetic Retinopathy (DR)
The clinical efficacy and safety of SUSVIMO were assessed in a randomized, visual assessor and reading center-masked study (Pavilion-NCT04503551) in patients with moderately-severe to severe non-proliferative diabetic retinopathy (NPDR) [Early Treatment Diabetic Retinopathy Study Diabetic Retinopathy Severity Scale (ETDRS-DRSS) of 47 or 53], without center-involved DME (CI-DME), and who had not received prior treatment in the study eye for DR. A total of 174 patients (106 in the SUSVIMO arm and 68 in the observational comparator arm) were enrolled in this study.
Patients who had not received prior treatment in the study eye for DR were randomized in a 5:3 ratio to continuous delivery of SUSVIMO via the implant every 36 weeks or to clinical observation. Prior to the implant procedure, two loading doses of intravitreal ranibizumab 0.5 mg were administered in the study eye. The observational comparator arm did not receive loading doses of intravitreal ranibizumab. For patients who developed CI-DME or proliferative diabetic retinopathy/anterior segment neovascularization in either arm, supplemental treatment with intravitreal injections of 0.5 mg ranibizumab was available per investigator's clinical judgment at any non-refill-exchange study visit. Patient ages ranged from 24 to 83 years with a mean of 53.9 years. At baseline, the overall mean visual acuity was 82.4 letters (range: 69 to 97 letters).
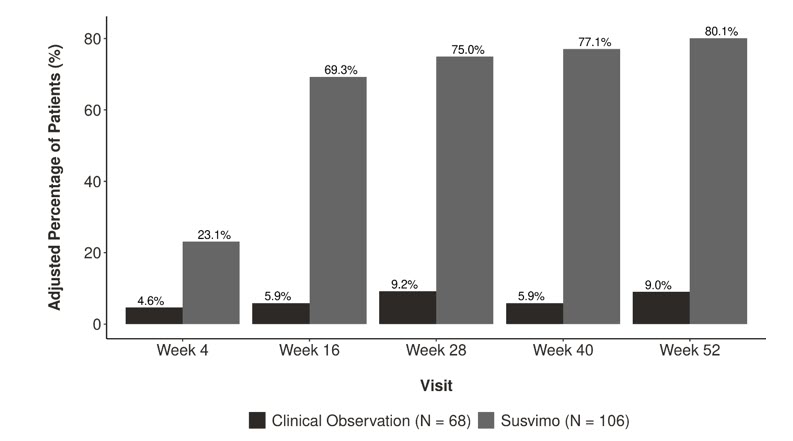
The primary efficacy endpoint was the proportion of patients with a ≥ 2-step improvement on the ETDRS-DRSS from baseline at Week 52 versus clinical observation. SUSVIMO with two loading doses of intravitreal ranibizumab was superior to clinical observation at Week 52. Detailed results are shown in Table 5 and Figure 31 below.
| Outcome Measure | SUSVIMO 100 mg/mL (n=106) | Clinical Observation (n=68) | Difference 95% CI All estimates are adjusted estimates based on the CMH method. 95% is a rounding of 95.04% CI; the type 1 error was adjusted for interim safety monitoring. p<0.01 compared with clinical observation. |
|---|---|---|---|
| ETDRS-DRSS = Early Treatment Diabetic Retinopathy Study Diabetic Retinopathy Severity Scores CMH = Cochran-Mantel-Haenszel test | |||
| Adjusted proportion of patients with ≥ 2-step improvement from baseline on the ETDRS-DRSS at Week 52 | 80% | 9% | 71% (61%, 81%) |
Figure 31 Adjusted Proportion of Patients with a ≥ 2-Step Improvement from Baseline on ETDRS-DRSS in Study Eye over Time through Week 52 in the Pavilion (GR41675) Study
In the SUSVIMO arm, none of the patients assessed for supplemental treatment received any supplemental injections of intravitreal ranibizumab and 40% of patients in the observational comparator arm received 1 or more supplemental treatments through Week 52.
Consistent results were observed across patient subgroup analyses for ETDRS-DRSS score including age, race, ethnicity, baseline hemoglobin (HbA1c) and baseline ETDRS-DRSS score.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Each SUSVIMO initial fill needle kit (NDC 50242-078-55) contains:
- One SUSVIMO 100 mg/mL single-dose glass vial
- One SUSVIMO initial fill needle (34-gauge needle with a 5 μm integrated filter) with a blue cap
Each SUSVIMO (ranibizumab injection) carton (NDC 50242-078-12) contains one SUSVIMO (ranibizumab injection) 100 mg/mL that is clear to slightly opalescent, colorless to slightly brownish solution in a single-dose glass vial.
Each SUSVIMO initial fill needle carton contains a SUSVIMO initial fill needle (34-gauge needle with a 5 μm integrated filter) with a blue cap.
Each SUSVIMO refill needle carton contains a SUSVIMO refill needle (34-gauge vented needle with a 5 μm integrated filter) with a clear cap.
| Device and Materials Description | |
| Components | |
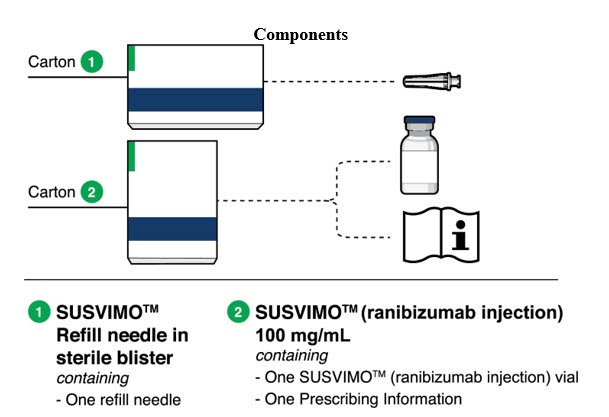 Figure 32 Figure 32 | |
SUSVIMO implant
|  Figure 33 Figure 33 |
SUSVIMO (ranibizumab injection) 100 mg/mL vial
| 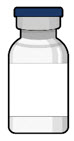 Figure 34 Figure 34 |
SUSVIMO refill needle
|  Figure 35 Figure 35 |
Materials List
Materials that are required and supplied to perform the procedure are:
- SUSVIMO refill needle, 34 G, with clear cap
- SUSVIMO (ranibizumab injection) 100 mg/mL
Additional materials required to perform the procedure but are not provided are:
- One sterile 1 mL Luer Lock syringe (not included)
- One sterile 5-micron filter needle (19-gauge × 1½ inch) (not included )
- Anesthetic ophthalmic solutions
- Ophthalmic broad-spectrum microbicide solution
- Cotton tips and gauze
- Sterile powder free gloves
- Face masks
- Lid speculum
- Magnification such as visor or loupes
- Task lighting
- Indirect ophthalmoscope and lens
- Sterile drape (optional for refill-exchange procedure)
Storage
Store SUSVIMO initial fill needle kit at 2°C to 8°C (36°F to 46°F). Do not freeze. Protect from light. Do not shake. The SUSVIMO initial fill needle has been sterilized with electron beam processing.
Store SUSVIMO (ranibizumab injection) 100 mg/ mL vial at 2°C to 8°C (36°F to 46°F). Do not freeze. Protect from light. Do not shake. Prior to use, the unopened vial may be kept at 9°C to 30°C (48°F to 86°F) for up to 24 hours provided it is protected from light.
Store the SUSVIMO implant and insertion tool assembly, refill needle and explant tool at room temperature 15°C to 25°C (59°F to 77°F). The SUSVIMO implant and insertion tool assembly has been sterilized with ethylene oxide gas. The SUSVIMO refill needle and explant tool has been sterilized with electron beam processing.
Store the SUSVIMO initial fill needle at 2°C to 25°C (36°F to 77°F). The SUSVIMO initial fill needle has been sterilized with electron beam processing.
Handling
SUSVIMO components are supplied sterile and are for single-use only. Do not reprocess, re-sterilize, or reuse SUSVIMO components. Do not use if the sterility has been compromised or the contents have been dropped, damaged or tampered with. Do not use past the expiration date printed on the label. Do not open sealed tray until time of use. Avoid contact between sharp surgical instruments and the SUSVIMO implant as the material of the septum and silicone encasing is soft and susceptible to damage.
Important Device Handling Information
- Use caution when performing ophthalmic procedures (e.g., B-scan ophthalmic ultrasound, scleral depression, or gonioscopy) that may cause deflection or movement of the implant and subsequent injury.
Ocular Implant Initial Fill Procedure
- Minimize air bubbles within the implant reservoir as they may cause slower drug release. If an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed after initial fill, do not use the implant.
Ocular Implant Insertion Procedure
- Perpendicular entry of the implant is important to avoid contact between the implant and intraocular structures such as the lens, as contact between the implant and the intraocular structures may cause adverse reactions such as traumatic cataract.
- Avoid excessive force on the globe by first ensuring that the tip of the implant has passed through the sclero-pars plana incision before slowly pushing the implant into place.
Ocular Implant Removal Procedure
- Do not grasp the implant by the short axis of the implant flange. Remove the implant in a gentle manner. Perpendicular exit of the implant is important to avoid contact between the implant and intraocular structures such as the lens.
Susvimo ® (ranibizumab injection) For Susvimo ocular implant use Instructions for Use
| Initial Fill and Implant Procedure |
Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician.
Susvimo procedures should be performed by an ophthalmologist experienced in vitreoretinal surgery.
Refer to the Susvimo (ranibizumab injection) 100 mg/mL prescribing information for a complete list of indications, contraindications, warnings, precautions, and adverse events.
Contents
Introduction
| 3 | Device Description |
| 4 | Components |
| 8 | Intended Use/Indications for Use |
| 8 | Contraindications |
| 8 | Warnings |
| 9 | Precautions |
| 10 | How Supplied, Handling, and Storage |
Instructions for Use
| 11 | Introduction and Materials |
| 13 | Preparatory Procedures |
| 14 | Infusion Line Placement Procedure |
| 15 | Conjunctival Dissection Procedure |
| 17 | Syringe Preparation and Initial Implant Fill |
| 25 | Implant Insertion |
| 35 | Disposal and Post-insertion Procedures |
| 36 | Explanation of symbols on product or package labeling |
Introduction
These instructions include only the procedure for filling and inserting the Susvimo implant. For more information, refer to the Susvimo prescribing information for the refill-exchange procedure and Susvimo Instructions for Use for the implant removal procedure.
Device Description
Susvimo is an intraocular drug delivery system designed to be used specifically with Susvimo (ranibizumab injection) 100 mg/mL. The system consists of an intraocular implant along with ancillary devices used to fill, insert, and explant (if needed) the implant.
The implant is a refillable drug reservoir that is inserted into the eye through the pars plana. The implant is secured within the scleral incision, with the extrascleral flange that remains visible through the conjunctiva following insertion. Once filled with ranibizumab, the implant is designed to provide continuous release of ranibizumab. The implant will be refilled with ranibizumab in an office-based setting via an administration through the conjunctiva and implant septum.
Components
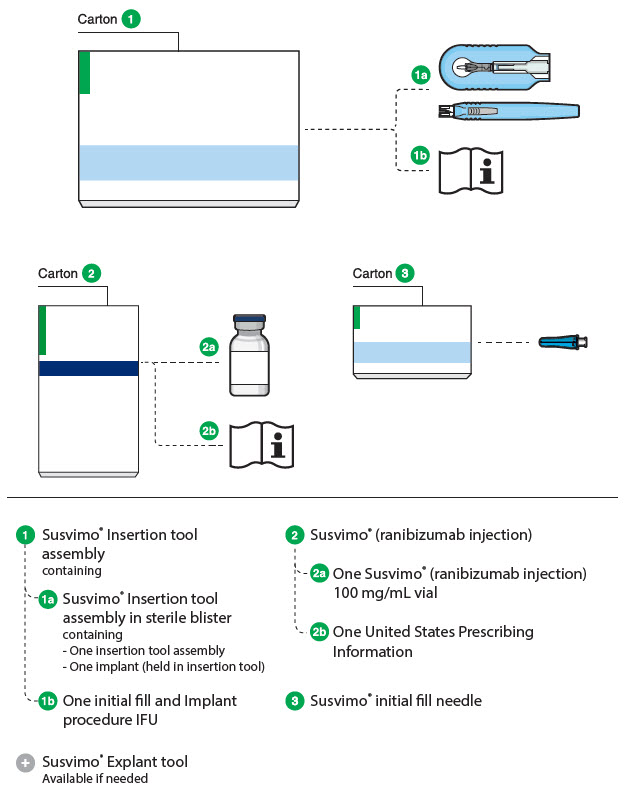
Figure 1
Susvimo components for initial fill and implant procedure
Susvimo implant (packaged with insertion tool assembly)
Susvimo implant (Figure 2 and Figure 3 ) is a refillable reservoir inserted into the eye through the pars plana. The body of the implant, which includes the release control element, extends into the vitreous cavity.
|
|
| Figure 2 Susvimo implant | Figure 3 Susvimo implant detail |
| Implant components | Description |
|---|---|
| Extrascleral flange | The extrascleral flange provides secure anchoring of the implant within the scleral incision and is encased in silicone. |
| Septum | The septum is a self-sealing interface through which ranibizumab is administered into the implant both prior to insertion and during subsequent refills in an office-based setting. |
| Body | The body of the implant contains a hollow drug reservoir, capable of holding 0.02 mL of drug. |
| Release control element | The titanium release control element controls the rate of ranibizumab diffusion from the drug reservoir into the vitreous. |
Susvimo insertion tool assembly
Susvimo insertion tool assembly is designed to facilitate handling of the implant during the initial fill and implant procedure. The insertion tool assembly is comprised of a carrier and handle (Figure 4 and Figure 5 ), described in more detail below:
|
|
| Figure 4 Susvimo insertion tool carrier | Figure 5 Susvimo insertion tool handle |
| Insertion tool assembly components | Description |
|---|---|
| Gripper tips | The implant is provided pre-positioned in the insertion tool assembly gripper tips. After initial fill of the implant with ranibizumab, the implant and gripper tips are transferred from the insertion tool carrier to the insertion tool handle. |
| Luer lock slot | The syringe is loaded into the insertion tool carrier by aligning the syringe Luer lock with the insertion tool carrier Luer lock slot. |
| Guide channel | The guide channel serves to direct the syringe and initial fill needle in the insertion tool carrier during the initial fill of the implant. |
| Release button | Pressing the release button on the insertion tool handle will open the gripper tips and release the implant. |
Susvimo (ranibizumab injection) 100 mg/mL vial
Susvimo (ranibizumab injection) (Figure 6 ) is used to fill the implant prior to insertion or during subsequent refill-exchange in an office-based setting.
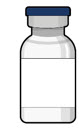
Figure 6 Susvimo (ranibizumab injection) 100 mg/mL vial
Susvimo initial fill needle
Susvimo initial fill needle (Figure 7 ) is designed to fill the implant with ranibizumab prior to implant insertion. The initial fill needle is distinguished by its blue cap.
Figure 7 Susvimo initial fill needle
| Initial fill needle components | Description |
|---|---|
| Needle | 34 G needle |
| Integrated filter | Integrated 5 µm filter within needle hub |
Intended Use/Indications for Use
Susvimo ocular implant is approved for use with Susvimo (ranibizumab injection). Refer to the Susvimo (ranibizumab injection) prescribing information for a complete list of indications, contraindications, warnings, precautions, and adverse events.
Contraindications
Susvimo is contraindicated in patients with ocular or periocular infections, with active intraocular inflammation, or with known hypersensitivity to ranibizumab or any of the excipients in Susvimo (ranibizumab injection) 100 mg/mL. Hypersensitivity reactions may manifest as severe intraocular inflammation.
Warnings
- Do not use if the sterility has been compromised or the contents have been dropped, damaged or tampered with.
- Minimize air bubbles within the implant reservoir as they may cause slower drug release. If an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed after initial fill, do not use the implant.
- Perpendicular entry of the implant is important to avoid contact between the implant and intraocular structures such as the lens, as contact between the implant and the intraocular structures may cause adverse events such as traumatic cataract.
- Avoid excessive force on the globe by first ensuring that the tip of the implant has passed through the sclero-pars plana incision before slowly pushing the implant into place.
Precautions
- Read and follow all instructions, warnings, and cautions prior to use.
- Susvimo procedures should be performed by an ophthalmologist experienced in vitreoretinal surgery.
- Use the Susvimo components and materials as specified in these instructions to perform the implant insertion procedure including initial fill.
- Avoid contact between sharp surgical instruments and the implant as the material of the septum and silicone encasing are soft and susceptible to damage.
- The implant is MR Conditional. The Patient Implant card is provided with instructions and must be completed and given to the patient after implant insertion. For further information please refer to the ' Post-insertion patient instructions ' section.
Use with Standard Procedures
Susvimo implant is compatible for use with the following standard procedures: A-scan ophthalmic ultrasound, slit lamp examination, indirect ophthalmoscopy, tonometry, optical coherence tomography (OCT), visual field (perimetry), standard lasers for ophthalmic treatments, radiography (x-ray), computed tomography (CT) scan, fluorescein/ indocyanine angiography, and fundus autofluorescence.
Use caution when performing ophthalmic procedures that may cause deflection of the implant and subsequent injury. For example, B-scan ophthalmic ultrasound, scleral depression, or gonioscopy.
Magnetic Resonance Imaging (MRI) Safety Information
 MR Conditional.
MR Conditional.
Non-clinical testing has demonstrated that the Susvimo implant is MR Conditional. A patient with this device can be safely scanned in an MR system meeting the following conditions:
- Static magnetic field of 1.5-Tesla (1.5 T) or 3-Tesla (3 T)
- Maximum spatial field gradient of 3,000 G/cm (30 T/m)
- Maximum MR system reported, whole body averaged specific absorption rate (SAR) of 4.0 W/kg (First Level Controlled Operating Mode)
Under the scan conditions defined above, the Susvimo implant is expected to produce a maximum temperature rise of less than 1°C after 15 minutes of continuous scanning.
In non-clinical testing, the image artifact caused by the device extends approximately 4 mm from the Susvimo implant when imaged with a gradient echo pulse sequence in a 3 T MRI system.
How Supplied, Handling, and Storage
- All Susvimo components are supplied sterile. Do not reprocess or resterilize.
- All Susvimo components are for single use only. Do not reuse Susvimo components.
- Do not open sealed tray until time of use.
- Do not use if the package is damaged or broken as sterility may be compromised.
- Do not use past the expiration date printed on the label.
Susvimo ocular implant and insertion tool assembly
- The sealed tray has been sterilized with ethylene oxide gas.
- Store the Susvimo implant and insertion tool assembly at room temperature 15°C to 25°C (59°F to 77°F).
Susvimo (ranibizumab injection)
- Susvimo should be stored at 2°C to 8°C (36°F to 46°F). Do not freeze. Protect from light. Do not shake. Prior to use, the unopened vial may be kept at 9°C to 30°C (48°F to 86°F) for up to 24 hours.
Initial fill needle
- Susvimo initial fill needle should be stored at 2°C to 25°C (36°F to 77°F). Susvimo initial fill needle has been sterilized with electron beam processing.
See Susvimo (ranibizumab injection) prescribing information for additional information.
Instructions for Use
Introduction and Materials
Implant insertion is a surgical procedure that is performed in an operating room. The implant is filled with Susvimo (ranibizumab injection) immediately prior to insertion.
Materials List
The materials that are required in the operating room on the day of the procedure are listed in Tables 4 and 5 .
| Item Description |
|---|
| Susvimo ocular implant with insertion tool assembly |
| Susvimo initial fill needle, 34 G, with blue cap |
| Susvimo (ranibizumab injection) 100 mg/mL |
| Susvimo explant tool (Refer to Susvimo implant removal Instructions for Use for information on implant removal.) |
| Item Description |
|---|
| One sterile 1 mL Luer Lock syringe (not included) |
| One sterile 5-micron filter needle (19-gauge × 1½ inch) (not included) |
| Surgical microscope |
| Vitrectomy surgical control system |
| Standard 25 G or 27 G vitrectomy set up |
| 23 G or 25 G 532 nm Endolaser probe and associated source |
| Standard vitrectomy tray (including adjustable caliper, 0.12 straight toothed forceps, blunt wescott scissors) |
| Cauterization equipment (including standard fine tip diathermy and eraser tip wet-field cautery) |
| Ophthalmic broad-spectrum microbicide solution |
| Marking pad |
| 3.5 mm and 4.0 mm fixed caliper or equivalent fixed tool |
| 3.5 mm fixed width gauge or equivalent fixed tool |
| 19 G or 20 G MVR Straight Knife |
| Slit Knife, 3.2 mm Straight |
| Gut or Vicryl sutures for conjunctival tissues (suggested 7-0 to 9-0: monofilament recommended) |
| Indirect ophthalmoscope and lens |
| Drapes |
Preparatory Procedures
1. Inspect packaging and components
|
|
2. Inspect Susvimo (ranibizumab injection)
| |
3. Patient Preparation
| |
Infusion Line Placement Procedure
1. Place infusion line
|
|
Conjunctival Dissection Procedure
| 1. Identify the site of insertion |
|
| 2. Create conjunctival peritomy | |
| |
|
|
| |
Syringe Preparation and Initial Implant Fill
Using aseptic technique, the implant will be filled with 0.02 mL of ranibizumab prior to insertion of the implant into the patient's eye.
1. Transfer dose from vial to syringe
|
|
2. Remove air from syringe
|
|
3. Attach initial fill needle
|
|
4. Remove any remaining air from syringe
|
|
5. Inspect the syringe for air bubbles
|
|
6. Load syringe into the carrier
|
|
7. Fill implant with ranibizumab under microscope
|
|
8. Inspect the filled implant under the microscope
|
|
| 9. Remove the syringe and guide sleeve from the carrier | |
|
|
| 10. Slide the insertion tool handle into the carrier | |
|
|
Implant Insertion
1. Verify hemostasis of the scleral surface
- Clear any excess blood from the scleral surface.
- Perform careful scleral wet field cauterization as needed, particularly in the area of the sclero-pars plana incision.
2. Mark incision site
|
|
3. Perform scleral incision
|
|
| 4. Perform laser treatment of the pars plana
|
|
- Using a 532 nm laser endoprobe, apply contiguous, overlapping laser spots starting at 300 mW 1000 ms along the full length of the exposed pars plana (Figure 32 ). Repeat until complete ablation is achieved.
- Maintain focus of laser on exposed pars plana.
- Keep foot pedal depressed to achieve full 1000 ms spots (expect smoke).
- Ensure the pars plana at the corners of scleral incision is adequately treated.
- Do not use painting strokes with the laser.
- Do not contact the pars plana directly with laser probe.
- Repeat application of laser along the full length of the pars plana until full or partial split of the pars plana, or other visual endpoints are achieved, as indicated by:
- Gray color change.
- Uniform perforated appearance.
- Domes of vitreous fluid percolating through pars plana.
- If visual endpoint is not achieved after several passes with the laser, increase laser power in 100 mW increments.
- Once laser ablation of pars plana is completed, remeasure the entire length (including corners) of the scleral incision with 3.5 mm gauge to confirm the final post-laser incision is 3.5 mm. If final incision is greater than 3.5 mm, as indicated by a loose fit/side to side motion of 3.5 mm gauge, place a suture through the wound opposite the relaxing incision to reduce incision down to 3.5 mm.
 Caution
Caution
A final post-laser incision length of 3.5 mm provides a secure fit for the implant.
Do not enlarge the scleral dissection beyond 3.5 mm as a final incision length greater than 3.5 mm may result in an improperly seated implant and will require additional suturing.
5. Perform pars plana incision
|
|
| 6. Withdraw the insertion tool handle with filled implant | |
|
|
7. Stabilize the globe
|
|
8. Orient the implant
|
|
9. Insert the implant
|
|
10. Release the implant
|
|
11. Seat the implant
|
|
12. Suture Tenon's capsule and conjunctiva
|
|
13. Remove infusion cannula
- If the infusion was previously turned on, then set the infusion pressure to 20 mmHg before removing the infusion cannula.
- Check for persistent leaks at the infusion cannula site and suture if necessary.
14. Check Intraocular Pressure (IOP)
- Using digital palpation, check the IOP. If necessary, inject additional fluid to restore IOP.
15. Check implant placement
- Perform indirect ophthalmoscopy to confirm implant position and to examine for the presence of any complications.
Disposal and Post-insertion Procedures
1. Dispose of used Susvimo components and tools
- Do not recap the needle or detach it from the syringe. Dispose of the used Susvimo components and tools in a sharps disposal container or in accordance with local requirements.
2. Perform post-insertion procedures
- Post-insertion procedures are consistent with standard post-surgical procedures.
3. Post-insertion patient instructions
Provide the patient with the following post-operative instructions:
- Positioning:
- Keep head above shoulder level for the rest of the day.
- Sleep with head elevated on 3 or more pillows if lying down during the day and night after implant insertion.
- Information on caring for the eye after the procedure, including but not limited to the following:
- Do not remove the eye shield until they are instructed to do so by their physician. At bedtime, continue to wear the eye shield for at least 7 nights following implant insertion.
- Administer all post-operative eye medications, as directed by their physician.
- Do not push on the eye, rub the eye, or touch the region of the eye where the implant is located (underneath the eyelid in the upper and outer part of the eye) for 30 days following implant insertion. Avoid rubbing the eye or touching the area where the implant is located as much as possible at all other times but if necessary to do so, make sure hands are cleaned prior to touching the eye.
- Do not participate in strenuous activities until 1 month after implant insertion or after discussion with their physician.
- Monitor for symptoms that may require immediate medical attention while the implant is in place.
- MR Conditional information:
- The surgeon should inform the patient that the implant is MR Conditional (as noted on their Susvimo implant card) and if patient needs to undergo an MRI, they should let their doctor know they have Susvimo implanted in their eye.
- After implant insertion, the surgeon should give the patient the implant card with the appropriate information filled in, and should advise the patient to keep the card in a safe place, e.g. his or her wallet, for future reference. The surgeon should advise the patient that this implant card contains important information related to the Susvimo implant and that the card should be shown to their current and future health care providers.
Explanation of symbols on product or package labeling
| Symbol | Title |
|---|---|
 | Manufacturer |
 | Prescription only |
 | Do not re-use |
 | Do not use if package is damaged |
 | Consult Instructions for Use |
 | Sterilized by irradiation |
 | Sterilized using ethylene oxide |
 | Temperature limit |
 | Expiration date/Use by date |
 | Lot/Batch number |
 | MR Conditional |
Manufactured by:
Genentech, Inc. A Member of the Roche Group 1 DNA Way South San Francisco, CA 94080-4990 U.S. License No.: 1048
SUSVIMO is a registered trademark of Genentech, Inc. ©2025 Genentech, Inc. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Approved: 9/2025
Mechanism of Action
Ranibizumab binds to the receptor binding site of multiple biologically active forms of VEGF-A, including VEGF 110 . VEGF-A has been shown to cause neovascularization and leakage in models of ocular angiogenesis and vascular occlusion and is thought to contribute to pathophysiology of neovascular AMD. The binding of ranibizumab to VEGF-A prevents the interaction of VEGF-A with its receptors (VEGFR1 and VEGFR2) on the surface of endothelial cells, reducing endothelial cell proliferation, vascular leakage, and new blood vessel formation.
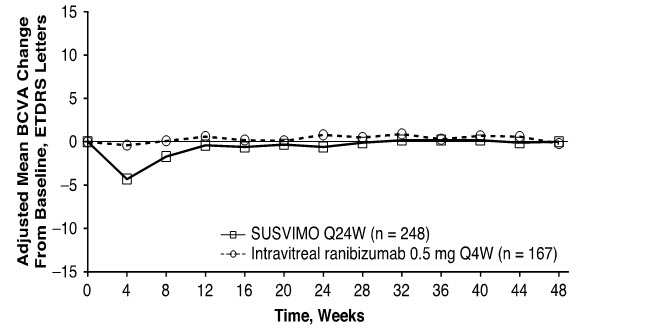
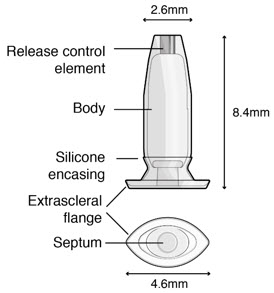
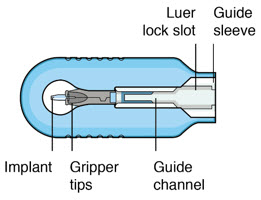
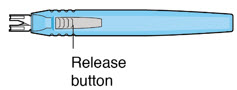
 Warning Do not use if the sterility has been compromised or the contents have been dropped, damaged, or tampered with.
Warning Do not use if the sterility has been compromised or the contents have been dropped, damaged, or tampered with.  Figure 8 Susvimo components on sterile field
Figure 8 Susvimo components on sterile field  Caution Do not use if particulate, cloudiness or discoloration are visible.
Caution Do not use if particulate, cloudiness or discoloration are visible.  Figure 9 Place infusion cannula in inferotemporal quadrant
Figure 9 Place infusion cannula in inferotemporal quadrant 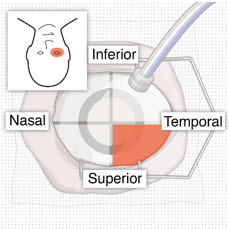 Figure 10 Superotemporal quadrant
Figure 10 Superotemporal quadrant 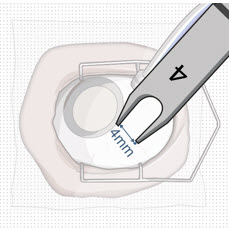 Figure 11 4 mm posterior to the limbus
Figure 11 4 mm posterior to the limbus  Caution The peritomy size should be at least 6 mm by 6 mm centered around the selected implant location to ensure the proper clearance of the conjunctiva and Tenon's capsule from the implant flange once the implant is inserted into the eye.
Caution The peritomy size should be at least 6 mm by 6 mm centered around the selected implant location to ensure the proper clearance of the conjunctiva and Tenon's capsule from the implant flange once the implant is inserted into the eye. 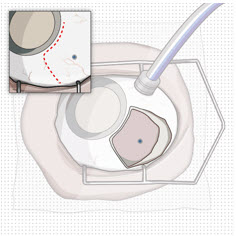 Figure 12 Conjunctival peritomy with blue dot representing selected implant location
Figure 12 Conjunctival peritomy with blue dot representing selected implant location  Caution Use the filter needle to withdraw ranibizumab from the vial. Do not use the initial fill needle for this step.
Caution Use the filter needle to withdraw ranibizumab from the vial. Do not use the initial fill needle for this step. 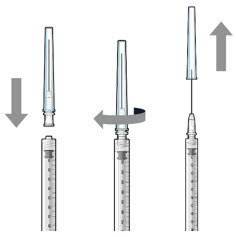 Figure 13 Filter needle attached to syringe and cap removal
Figure 13 Filter needle attached to syringe and cap removal  Figure 14 No air bubbles in syringe
Figure 14 No air bubbles in syringe  Caution Ensure that the initial fill needle is attached to the syringe. Do not use the filter needle to fill the implant.
Caution Ensure that the initial fill needle is attached to the syringe. Do not use the filter needle to fill the implant. 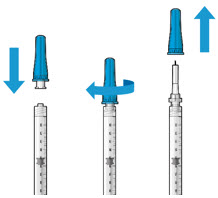 Figure 15 Initial fill needle attached to syringe and cap removal
Figure 15 Initial fill needle attached to syringe and cap removal 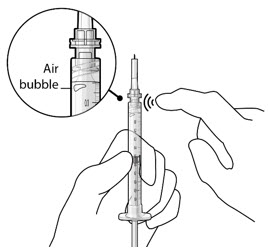 Figure 16 Gently tap the syringe to dislodge bubbles
Figure 16 Gently tap the syringe to dislodge bubbles 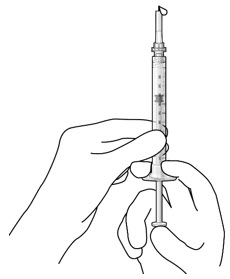 Figure 17 No air bubbles and a drop of drug at the needle tip
Figure 17 No air bubbles and a drop of drug at the needle tip  Caution Use the syringe within 15 minutesof removing all air to avoid ranibizumab drying in the needle and impeding fluid flow. Do not use the initial fill needle if the needle is clogged.
Caution Use the syringe within 15 minutesof removing all air to avoid ranibizumab drying in the needle and impeding fluid flow. Do not use the initial fill needle if the needle is clogged. 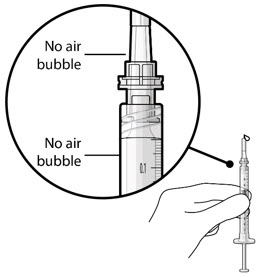 Figure 18 No air bubbles in the syringe and needle hub
Figure 18 No air bubbles in the syringe and needle hub  Caution Do not hold or push on the plunger rod of the syringe while inserting the needle into the implant septum.
Caution Do not hold or push on the plunger rod of the syringe while inserting the needle into the implant septum. 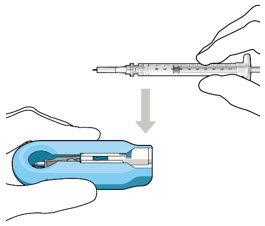 Figure 19 Align and lower the syringe into the carrier
Figure 19 Align and lower the syringe into the carrier 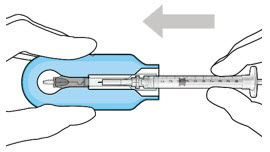 Figure 20 Push the syringe into the carrier
Figure 20 Push the syringe into the carrier 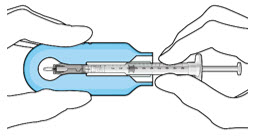 Figure 21 Syringe with initial fill needle inserted through the implant septum
Figure 21 Syringe with initial fill needle inserted through the implant septum  Caution When filling the implant, fluid should only exit the implant from the release control element. If fluid is leaking from the implant at a different location, such as the side of the implant, do not use the implant. If fluid is leaking from the septum at the needle insertion site, the needle may not be fully penetrating the implant septum. Fully push the syringe forward before continuing to fill the implant.
Caution When filling the implant, fluid should only exit the implant from the release control element. If fluid is leaking from the implant at a different location, such as the side of the implant, do not use the implant. If fluid is leaking from the septum at the needle insertion site, the needle may not be fully penetrating the implant septum. Fully push the syringe forward before continuing to fill the implant. 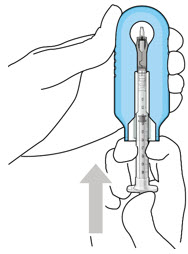 Figure 22 Administer ranibizumab into the implant
Figure 22 Administer ranibizumab into the implant 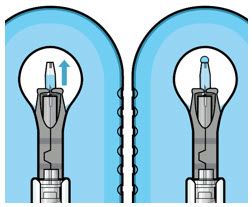 Figure 23 Dome of drug solution forms at tip of implant as viewed under magnification
Figure 23 Dome of drug solution forms at tip of implant as viewed under magnification  Warning Minimize air bubbles within the implant reservoir as they may cause slower drug release. If an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed, do not use the implant.
Warning Minimize air bubbles within the implant reservoir as they may cause slower drug release. If an air bubble is present, it must be no larger than 1/3 of the widest diameter of the implant. If excess air is observed, do not use the implant.  Caution No more than 30 minutesshould pass between the initial fill of the implant and the insertion into the patient's eye to ensure that the release control element remains saturated with ranibizumab. If ranibizumab dries in the release control element, the implant may not release the drug properly into the vitreous after insertion.
Caution No more than 30 minutesshould pass between the initial fill of the implant and the insertion into the patient's eye to ensure that the release control element remains saturated with ranibizumab. If ranibizumab dries in the release control element, the implant may not release the drug properly into the vitreous after insertion. 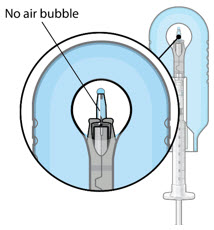 Figure 24 Proper appearance of implant after initial filling with ranibizumab
Figure 24 Proper appearance of implant after initial filling with ranibizumab 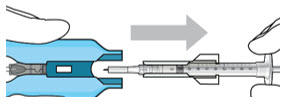 Figure 25 Remove the syringe and guide sleeve from the insertion tool carrier
Figure 25 Remove the syringe and guide sleeve from the insertion tool carrier  Caution Do not withdraw the handle and implant until the eye is ready for insertion. Contact between the implant and any surface or object – even within the sterile field – may result in the introduction of a foreign body into the vitreous.
Caution Do not withdraw the handle and implant until the eye is ready for insertion. Contact between the implant and any surface or object – even within the sterile field – may result in the introduction of a foreign body into the vitreous. 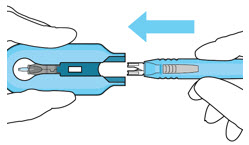 Figure 26 Insert the handle into the insertion tool carrier
Figure 26 Insert the handle into the insertion tool carrier  Figure 27 Fully inserted handle
Figure 27 Fully inserted handle 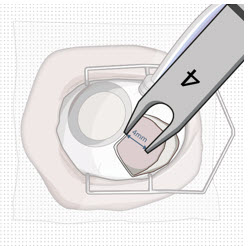 Figure 28 4 mm posterior to the limbus
Figure 28 4 mm posterior to the limbus 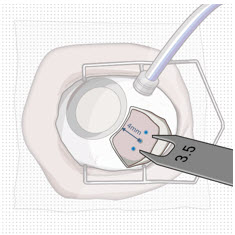 Figure 29 Fixed width caliper measurement
Figure 29 Fixed width caliper measurement  Caution The final target length for the scleral incision is 3.5 mm. Keep in mind that laser may further result in enlargement of incision and limit the secureness of the fit of the implant.
Caution The final target length for the scleral incision is 3.5 mm. Keep in mind that laser may further result in enlargement of incision and limit the secureness of the fit of the implant. 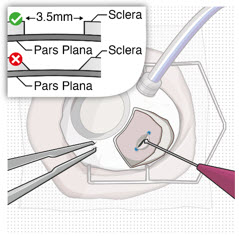 Figure 30 Stabilize the globe and perform full thickness scleral incision
Figure 30 Stabilize the globe and perform full thickness scleral incision 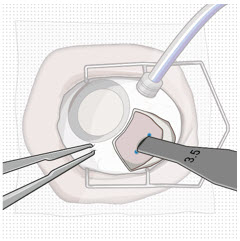 Figure 31 Confirm correct length of scleral incision
Figure 31 Confirm correct length of scleral incision  Caution Laser treatment may result in enlargement of the incision. Laser treatment should not be used to intentionally enlarge the length of scleral incision.
Caution Laser treatment may result in enlargement of the incision. Laser treatment should not be used to intentionally enlarge the length of scleral incision.  Caution Carefully apply the laser only to the choroidal tissue in the exposed pars plana. Minimize any laser application to the surrounding scleral tissue to avoid damaging tissue integrity.
Caution Carefully apply the laser only to the choroidal tissue in the exposed pars plana. Minimize any laser application to the surrounding scleral tissue to avoid damaging tissue integrity. 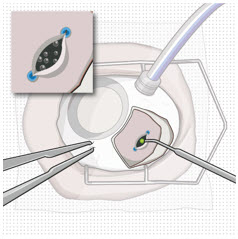 Figure 32 Laser treatment of the pars plana
Figure 32 Laser treatment of the pars plana  Caution Do not enlarge the pars plana incision with lateral movements. The incision width of 3.2 mm ensures that the pars plana incision is within the laser-treated area of the pars plana and reduces the risk of vitreous hemorrhage.
Caution Do not enlarge the pars plana incision with lateral movements. The incision width of 3.2 mm ensures that the pars plana incision is within the laser-treated area of the pars plana and reduces the risk of vitreous hemorrhage. 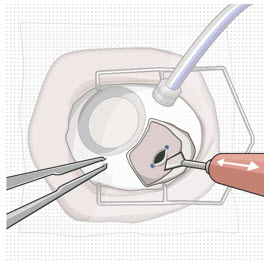 Figure 33 Pars plana incision
Figure 33 Pars plana incision 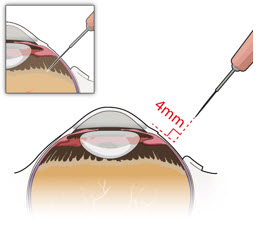 Figure 34 Perpendicular entry of slit knife
Figure 34 Perpendicular entry of slit knife  Caution When holding the insertion tool handle, take care not to touch the implant to any surface other than the sclero-pars plana incision during the insertion procedure. Do not use the implant if it is inadvertently released or is contaminated through contact with any surface other than the exposed sclera, including objects within the surgical field.
Caution When holding the insertion tool handle, take care not to touch the implant to any surface other than the sclero-pars plana incision during the insertion procedure. Do not use the implant if it is inadvertently released or is contaminated through contact with any surface other than the exposed sclera, including objects within the surgical field.  Figure 35 Withdraw the insertion tool handle with filled implant
Figure 35 Withdraw the insertion tool handle with filled implant 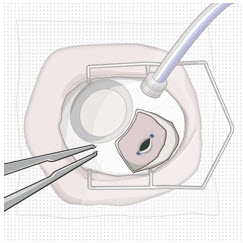 Figure 36 Stabilize the globe with forceps
Figure 36 Stabilize the globe with forceps 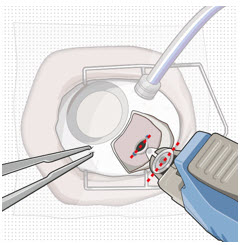 Figure 37 The long axis of the implant aligned with the length of the sclero-pars plana incision
Figure 37 The long axis of the implant aligned with the length of the sclero-pars plana incision  Warning Perpendicular entry of the implant is important to avoid contact between the implant and intraocular structures such as the lens, as contact between the implant and the intraocular structures may cause adverse events such as traumatic cataract.
Warning Perpendicular entry of the implant is important to avoid contact between the implant and intraocular structures such as the lens, as contact between the implant and the intraocular structures may cause adverse events such as traumatic cataract.  Warning Avoid excessive force on the globe by first ensuring that the tip of the implant has passed through the sclero-pars plana incision before slowly pushing the implant into place.
Warning Avoid excessive force on the globe by first ensuring that the tip of the implant has passed through the sclero-pars plana incision before slowly pushing the implant into place. 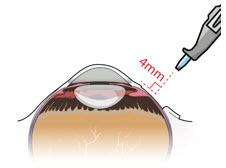 Figure 38 Perpendicular entry of the implant
Figure 38 Perpendicular entry of the implant 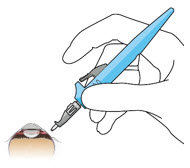 Figure 39 Implant insertion
Figure 39 Implant insertion 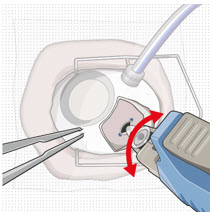 Figure 40 Implant insertion with a slight initial twisting motion
Figure 40 Implant insertion with a slight initial twisting motion  Caution If the implant flange is not parallel to the limbus after releasing, it needs to be repositioned solely using the gripper tips of the insertion tool handle. Reposition gently to avoid damage to the implant and to avoid contact between the implant and intraocular structures such as the lens. Avoid excessive manipulation of the implant flange. Do not use any other rigid instruments to reposition.
Caution If the implant flange is not parallel to the limbus after releasing, it needs to be repositioned solely using the gripper tips of the insertion tool handle. Reposition gently to avoid damage to the implant and to avoid contact between the implant and intraocular structures such as the lens. Avoid excessive manipulation of the implant flange. Do not use any other rigid instruments to reposition. 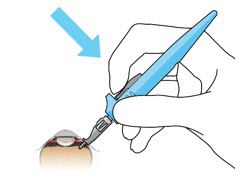 Figure 41 Release implant with the release button
Figure 41 Release implant with the release button 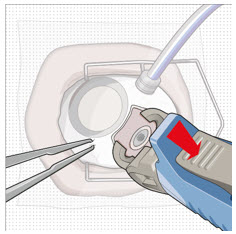 Figure 42 Implant release
Figure 42 Implant release  Caution Use the insertion tool gripper tips to seat the implant. Do not use any other rigid instruments as they may damage the implant.
Caution Use the insertion tool gripper tips to seat the implant. Do not use any other rigid instruments as they may damage the implant.  Caution Ensure that the long axis of the implant is aligned with the length of the sclero-pars plana incision and that the implant is seated flush against the sclera.
Caution Ensure that the long axis of the implant is aligned with the length of the sclero-pars plana incision and that the implant is seated flush against the sclera. 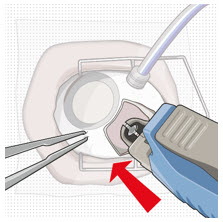 Figure 43 Seat the implant
Figure 43 Seat the implant  Caution Do not place a suture directly over the implant, otherwise adverse events including incomplete healing, infection, and discomfort can occur.
Caution Do not place a suture directly over the implant, otherwise adverse events including incomplete healing, infection, and discomfort can occur.  Caution Complete closure of both Tenon's capsule and conjunctiva across the surgical site is critical to minimize potential complications such as conjunctival retraction over the implant.
Caution Complete closure of both Tenon's capsule and conjunctiva across the surgical site is critical to minimize potential complications such as conjunctival retraction over the implant. 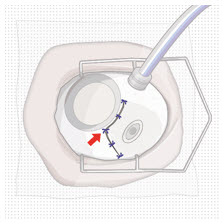 Figure 44 Suture conjunctiva
Figure 44 Suture conjunctiva