Temazepam - Temazepam capsule prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death ( see WARNINGS and Drug Interactions ).
• Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. • Limit dosages and durations to the minimum required. • Follow patients for signs and symptoms of respiratory depression and sedation
INDICATIONS AND USAGE
Temazepam capsules are indicated for the short-term treatment of insomnia (generally 7 to 10 days).
For patients with short-term insomnia, instructions in the prescription should indicate that Temazepam capsules should be used for short periods of time (7 to 10 days).
The clinical trials performed in support of efficacy were 2 weeks in duration with the final formal assessment of sleep latency performed at the end of treatment.
DOSAGE AND ADMINISTRATION
While the recommended usual adult dose is 15 mg before retiring, 7.5 mg may be sufficient for some patients, and others may need 30 mg. In transient insomnia, a 7.5 mg dose may be sufficient to improve sleep latency. In elderly or debilitated patients, it is recommended that therapy be initiated with 7.5 mg until individual responses are determined.
CONTRAINDICATIONS
Benzodiazepines may cause fetal harm when administered to a pregnant woman. An increased risk of congenital malformations associated with the use of diazepam and chlordiazepoxide during the first trimester of pregnancy has been suggested in several studies. Transplacental distribution has resulted in neonatal CNS depression following the ingestion of therapeutic doses of a benzodiazepine hypnotic during the last weeks of pregnancy.
Reproduction studies in animals with temazepam were performed in rats and rabbits. In a perinatal-postnatal study in rats, oral doses of 60 mg/kg/day resulted in increasing nursling mortality. Teratology studies in rats demonstrated increased fetal resorptions at doses of 30 and 120 mg/kg in one study and increased occurrence of rudimentary ribs, which are considered skeletal variants, in a second study at doses of 240 mg/kg or higher. In rabbits, occasional abnormalities such as exencephaly and fusion or asymmetry of ribs were reported without dose relationship. Although these abnormalities were not found in the concurrent control group, they have been reported to occur randomly in historical controls. At doses of 40 mg/kg or higher, there was an increased incidence of the 13th rib variant when compared to the incidence in concurrent and historical controls.
Temazepam capsules are contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus. Patients should be instructed to discontinue the drug prior to becoming pregnant. The possibility that a woman of childbearing potential may be pregnant at the time of institution of therapy should be considered.
ADVERSE REACTIONS
During controlled clinical studies in which 1076 patients received temazepam capsules at bedtime, the drug was well tolerated. Side effects were usually mild and transient. Adverse reactions occurring in 1% or more of patients are presented in the following table.
| Temazepam Capsules % Incidence (n=1076) | Placebo %Incidence (n=783) | |
| Drowsiness | 9.1 | 5.6 |
| Headache | 8.5 | 9.1 |
| Fatigue | 4.8 | 4.7 |
| Nervousness | 4.6 | 8.2 |
| Lethargy | 4.5 | 3.4 |
| Dizziness | 4.5 | 3.3 |
| Nausea | 3.1 | 3.8 |
| Hangover | 2.5 | 1.1 |
| Anxiety | 2 | 1.5 |
| Depression | 1.7 | 1.8 |
| Dry Mouth | 1.7 | 2.2 |
| Diarrhea | 1.7 | 1.1 |
| Abdominal Discomfort | 1.5 | 1.9 |
| Euphoria | 1.5 | 0.4 |
| Weakness | 1.4 | 0.9 |
| Confusion | 1.3 | 0.5 |
| Blurred Vision | 1.3 | 1.3 |
| Nightmares | 1.2 | 1.7 |
| Vertigo | 1.2 | 0.8 |
The following adverse events have been reported less frequently (0.5% to 0.9%):
Central Nervous System – anorexia, ataxia, equilibrium loss, tremor, increased dreaming
Cardiovascular – dyspnea, palpitations
Gastrointestinal – vomiting
Musculoskeletal – backache
Special Senses – hyperhidrosis, burning eyes
Amnesia, hallucinations, horizontal nystagmus, and paradoxical reactions including restlessness, overstimulation and agitation were rare (less than 0.5%).
Drug Interactions
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and monitor patients closely for respiratory depression and sedation.
The pharmacokinetic profile of temazepam does not appear to be altered by orally administered cimetidine dosed according to labeling.
DESCRIPTION
Temazepam, USP is a benzodiazepine hypnotic agent. The chemical name is 7-chloro-1, 3-dihydro-3-hydroxy-1-methyl-5-phenyl-2H-1, 4-benzodiazepin-2-one, and the structural formula is: 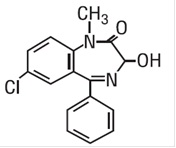 C 16 H 13 ClN 2 O 2 MW = 300.7 Temazepam, USP is a white or almost white crystalline powder, freely slightly soluble in methanol and methylene chloride.
C 16 H 13 ClN 2 O 2 MW = 300.7 Temazepam, USP is a white or almost white crystalline powder, freely slightly soluble in methanol and methylene chloride.
Temazepam Capsules USP, 7.5 mg, 15 mg, 22.5 mg, and 30 mg, are for oral administration.
7.5 mg, 15 mg, 22.5 mg, and 30 mg Capsules Active Ingredient: temazepam USP
7.5 mg Capsules Inactive Ingredients: lactose monohydrate, croscarmellose sodium, microcrystalline cellulose, sodium lauryl sulphate, magnesium stearate, FD&C Blue 1, D&C Red 28, FD&C Red 40, titanium dioxide, gelatin.
15 mg Capsules Inactive Ingredients: lactose monohydrate, croscarmellose sodium, microcrystalline cellulose, sodium lauryl sulphate, magnesium stearate, FD&C Blue 1, FD&C Red 40, titanium dioxide, gelatin.
22.5 mg Capsules Inactive Ingredients: lactose monohydrate, croscarmellose sodium, microcrystalline cellulose, sodium lauryl sulphate, magnesium stearate, FD&C Blue 1, D&C Red 28, titanium dioxide, gelatin.
30 mg Capsules Inactive Ingredients: lactose monohydrate, croscarmellose sodium, microcrystalline cellulose, sodium lauryl sulphate, magnesium stearate, FD&C Blue 1, FD&C Red 40, D&C Red 28, gelatin, titanium dioxide.
The printing ink contains shellac, propylene glycol, potassium hydroxide and iron oxide black.
CLINICAL PHARMACOLOGY
Pharmacokinetics
In a single and multiple dose absorption, distribution, metabolism, and excretion (ADME) study, using 3 H labeled drug, temazepam capsules was well absorbed and found to have minimal (8%) first pass metabolism. There were no active metabolites formed and the only significant metabolite present in blood was the O-conjugate. The unchanged drug was 96% bound to plasma proteins. The blood level decline of the parent drug was biphasic with the short half-life ranging from 0.4 to 0.6 hours and the terminal half-life from 3.5 to 18.4 hours (mean 8.8 hours), depending on the study population and method of determination. Metabolites were formed with a half-life of 10 hours and excreted with a half-life of approximately 2 hours. Thus, formation of the major metabolite is the rate limiting step in the biodisposition of temazepam. There is no accumulation of metabolites. A dose-proportional relationship has been established for the area under the plasma concentration/time curve over the 15 to 30 mg dose range.
Temazepam was completely metabolized through conjugation prior to excretion; 80% to 90% of the dose appeared in the urine. The major metabolite was the O-conjugate of temazepam (90%); the O-conjugate of N-desmethyl temazepam was a minor metabolite (7%).
Bioavailability, Induction, and Plasma Levels
Following ingestion of a 30 mg temazepam capsule, measurable plasma concentrations were achieved 10 to 20 minutes after dosing with peak plasma levels ranging from 666 to 982 ng/mL (mean 865 ng/mL) occurring approximately 1.2 to 1.6 hours (mean 1.5 hours) after dosing.
In a 7 day study, in which subjects were given a 30 mg temazepam capsule 1 hour before retiring, steady-state (as measured by the attainment of maximal trough concentrations) was achieved by the third dose. Mean plasma levels of temazepam (for days 2 to 7) were 260±210 ng/mL at 9 hours and 75±80 ng/mL at 24 hours after dosing. A slight trend toward declining 24 hour plasma levels was seen after day 4 in the study, however, the 24 hour plasma levels were quite variable.
At a dose of 30 mg once-a-day for 8 weeks, no evidence of enzyme induction was found in man.
Elimination Rate of Benzodiazepine Hypnotics and Profile of Common Untoward Effects
The type and duration of hypnotic effects and the profile of unwanted effects during administration of benzodiazepine hypnotics may be influenced by the biologic half-life of the administered drug and for some hypnotics, the half-life of any active metabolites formed. Benzodiazepine hypnotics have a spectrum of half-lives from short (<4 hours) to long (>20 hours). When half-lives are long, drug (and for some drugs their active metabolites) may accumulate during periods of nightly administration and be associated with impairments of cognitive and/or motor performance during waking hours; the possibility of interaction with other psychoactive drugs or alcohol will be enhanced. In contrast, if half-lives are shorter, drug (and, where appropriate, its active metabolites) will be cleared before the next dose is ingested, and carry-over effects related to excessive sedation or CNS depression should be minimal or absent. However, during nightly use for an extended period, pharmacodynamic tolerance or adaptation to some effects of benzodiazepine hypnotics may develop. If the drug has a short elimination half-life, it is possible that a relative deficiency of the drug, or, if appropriate, its active metabolites (i.e., in relationship to the receptor site) may occur at some point in the interval between each night’s use. This sequence of events may account for 2 clinical findings reported to occur after several weeks of nightly use of rapidly eliminated benzodiazepine hypnotics, namely, increased wakefulness during the last third of the night, and the appearance of increased signs of daytime anxiety.
Controlled Trials Supporting Efficacy
Temazepam capsules improved sleep parameters in clinical studies. Residual medication effects (“hangover”) were essentially absent. Early morning awakening, a particular problem in the geriatric patient, was significantly reduced.
Patients with chronic insomnia were evaluated in 2 week, placebo controlled sleep laboratory studies with temazepam capsules at doses of 7.5 mg, 15 mg, and 30 mg, given 30 minutes prior to bedtime. There was a linear dose-response improvement in total sleep time and sleep latency, with significant drug-placebo differences at 2 weeks occurring only for total sleep time at the 2 higher doses, and for sleep latency only at the highest dose.
In these sleep laboratory studies, REM sleep was essentially unchanged and slow wave sleep was decreased. No measurable effects on daytime alertness or performance occurred following temazepam capsules treatment or during the withdrawal period, even though a transient sleep disturbance in some sleep parameters was observed following withdrawal of the higher doses. There was no evidence of tolerance development in the sleep laboratory parameters when patients were given temazepam capsules nightly for at least 2 weeks.
In addition, normal subjects with transient insomnia associated with first night adaptation to the sleep laboratory were evaluated in 24 hour, placebo controlled sleep laboratory studies with temazepam capsules at doses of 7.5 mg, 15 mg, and 30 mg, given 30 minutes prior to bedtime. There was a linear dose-response improvement in total sleep time, sleep latency and number of awakenings, with significant drug-placebo differences occurring for sleep latency at all doses, for total sleep time at the 2 higher doses and for number of awakenings only at the 30 mg dose.
HOW SUPPLIED
Temazepam Capsules, USP
7.5 mg
Opaque blue cap / Opaque pink body, size 3, hard gelatin capsules imprinted with “A222” on body in black ink, filled with white to off white powder.
NDC 46708-379-30 Bottle of 30 capsules
NDC 46708-379-31 Bottle of 100 capsules
15 mg
Maroon cap / Opaque pink body, size 3, hard gelatin capsules imprinted with “A223” on body in black ink, filled with white to off white powder.
NDC 46708-380-30 Bottle of 30 capsules
NDC 46708-380-90 Bottle of 90 capsules
NDC 46708-380-31 Bottle of 100 capsules
NDC 46708-380-71 Bottle of 500 capsules
22.5 mg
Opaque blue cap / Opaque blue body, size 3, hard gelatin capsules imprinted with “A224” on body in black ink, filled with white to off white powder.
NDC 46708-381-30 Bottle of 30 capsules
30 mg
Maroon cap / Opaque blue body, size 3, hard gelatin capsules imprinted with “A225” on body in black ink, filled with white to off white powder.
NDC 46708-382-30 Bottle of 30 capsules
NDC 46708-382-90 Bottle of 90 capsules
NDC 46708-382-31 Bottle of 100 capsules
NDC 46708-382-71 Bottle of 500 capsules
Dispense in a well-closed, light-resistant container with a child-resistant closure.
Storage: Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
The brands listed are trademarks of their respective owners.
Manufactured by: Alembic Pharmaceuticals Limited (Formulation Division), Village Panelav, P. O. Tajpura, Near Baska, Taluka-Halol, Panchmahal, Gujarat, India.
Revised: 09/2018