Get your patient on Tetracycline Hydrochloride - Tetracycline Hydrochloride tablet, Film Coated (Tetracycline Hydrochloride)
Tetracycline Hydrochloride - Tetracycline Hydrochloride tablet, Film Coated prescribing information
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of tetracycline hydrochloride and other antibacterial drugs, tetracycline hydrochloride should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Tetracycline hydrochloride tablets are indicated in the treatment of infections caused by susceptible strains of the designated organisms in the conditions listed below:
- Upper respiratory tract infections caused by Streptococcus pyogenes , Streptococcus pneumoniae and Hemophilus influenzae . Note: Tetracycline hydrochloride tablets should not be used for streptococcal disease unless the organism has been demonstrated to be susceptible.
- Lower respiratory tract infections caused by Streptococcus pyogenes , Streptococcus pneumoniae , Mycoplasma pneumoniae (Eaton agent, and Klebsiella sp .)
- Skin and soft tissue infections caused by Streptococcus pyogenes , Staphylococcus aureaus . (Tetracyclines are not the drugs of choice in the treatment of any type of staphylococcal infections.)
- Infections caused by rickettsia including Rocky Mountain spotted fever, typhus group infections, Q fever, rickettsialpox.
- Psittacosis caused by Chlamydophila psittaci .
- Infections caused by Chlamydia trachomatis such as uncomplicated urethral, endocervical or rectal infections, inclusion conjunctivitis, trachoma, and lymphogranuloma venereum.
- Granuloma inquinale caused by Klebsiella granulomatis .
- Relapsing fever caused by Borrelia sp.
- Bartonellosis caused by Bartonella bacilliformis .
- Chancroid caused by Hemophilus ducreyi .
- Tularemia caused by Francisella tularensis .
- Plaque caused by Yersinia pestis .
- Cholera caused by Vibrio cholerae .
- Brucellosis caused by Brucella species (tetracycline hydrochloride tablets may be used in conjunction with an aminoglycoside).
- Infections due to Campylobacter fetus .
- As adjunctive therapy in intestinal amebiasis caused by Entamoeba histolytica .
- Urinary tract infections caused by susceptible strains of Escherichia coli , Klebsiella , etc.
- Other infections caused by susceptible gram-negative organisms such as E. coli , Enterobacter aerogenes , Shigella sp., Acinetobacter sp ., Klebsiella sp ., and Bacteroides sp.
- In severe acne, adjunctive therapy with tetracycline hydrochloride tablets may be useful.
When penicillin is contraindicated, tetracyclines are alternative drugs in the treatment of the following infections:
- Syphilis and yaws caused by Treponema pallidum and pertenue , respectively,
- Vincent's infection caused by Fusobacterium fusiforme ,
- Infections caused by Neisseria gonorrhoeae,
- Anthrax caused by Bacillus anthracis,
- Infections due to Listeria monocytogenes,
- Actinomycosis caused by Actinomyces species,
- Infections due to Clostridium species.
DOSAGE AND ADMINISTRATION
Adults: Usual daily dose, 1 gram as 500 mg twice a day or 250 mg four times a day. Higher doses such as 500 mg four times a day may be required for severe infections or for those infections which do not respond to the smaller doses.
For pediatric patients above eight years of age: Usual daily dose, 10 mg/lb to 20 mg/lb (25 mg/kg to 50 mg/kg) body weight divided in four equal doses.
Administration of adequate amounts of fluid with the tablet formulation of tetracycline is recommended to wash down the drug and reduce the risk of esophageal irritation and ulceration (see ADVERSE REACTIONS ).
Absorption of tetracycline is impaired by antacids containing aluminum, calcium or magnesium and preparations containing iron, zinc or sodium bicarbonate. Food and some dairy products also interfere with absorption.
When used in streptococcal infections, therapy should be continued for 10 days.
For treatment of brucellosis, 500 mg tetracycline four times a day for three weeks accompanied by streptomycin, 1 gram intramuscularly twice daily the first week and once daily the second week.
For the treatment of syphilis in patients allergic to penicillin, the following dosage of tetracycline is recommended: early syphilis (less than one year's duration), 500 mg four times a day for 15 days. Syphilis of more than one year's duration (except neurosyphilis), 500 mg four times a day for 30 days.
For treatment of gonorrhea, the recommended dose is 500 mg by mouth four times a day for seven days.
Uncomplicated urethral, endocervical or rectal infections in adults caused by Chlamydiatrachomatis : 500 mg, by mouth, four times a day for at least seven days.
In cases of moderate to severe acne which, in the judgement of the clinician, require long-term treatment, the recommended initial dosage is 1 gram daily in divided doses. When improvement is noted, reduce dosage gradually to maintenance levels ranging from 125 mg to 500 mg daily. In some patients it may be possible to maintain adequate remission of lesions with alternate day or intermittent therapy. Tetracycline hydrochloride tablets therapy of acne should augment the other standard measures known to be of value. Duration of long-term treatment which can safely be recommended has not been established (see WARNINGS and Carcinogenesis, Mutagenesis, Impairment of Fertility ).
Use in Specific Population
In patients with renal impairment (see WARNINGS ): decrease total dosage by reduction of recommended individual doses and/or by extending time intervals between doses.
CONTRAINDICATIONS
This drug is contraindicated in persons who have shown hypersensitivity to any of the tetracyclines.
ADVERSE REACTIONS
Gastrointestinal: anorexia, nausea, epigastric distress, vomiting, diarrhea, glossitis, black hairy tongue, dysphagia, enterocolitis, and inflammatory lesions (with Candida overgrowth) in the anogenital region.
Esophagitis and esophageal ulceration have been reported in patients receiving particularly the capsule and also the tablet forms of tetracyclines.
Most of the patients were reported to have taken medication immediately before going to bed (see DOSAGE AND ADMINISTRATION ).
Teeth: permanent discoloration of teeth may be caused during tooth development. Enamel hypoplasia has been reported (see WARNINGS ).
Skin: maculopapular and erythrematous rashes. Exfoliative dermatitis has been reported. Onycholysis and discoloration of the nails have been reported. Photosensitivity is discussed in WARNINGS .
Renal Toxicity: an increase in BUN has been reported and is dose related.
Liver: hepatotoxicity and liver failure have been observed in patients receiving tetracycline and in tetracycline-treated patients with renal impairment.
Hypersensitivity Reactions: urticaria, angioneurotic edema, anaphylaxis, anaphylactoid purpura, pericarditis, exacerbation of systemic lupus erythematosus, and serum sickness-like reactions, as fever, rash, and arthralgia.
Blood: hemolytic anemia, thrombocytopenia, thrombocytopenic purpura, neutropenia and eosinophilia have been reported.
When given over prolonged periods, tetracyclines have been reported to produce brown-black microscopic discoloration of thyroid glands. No abnormalities of thyroid function studies are known to occur.
Drug Interactions
Since bacteriostatic drugs may interfere with the bactericidal action of penicillin, it is advisable to avoid giving tetracycline in conjunction with penicillin or other bactericidal antibacterials.
Because the tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage.
The concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity.
Absorption of tetracyclines is impaired by antacids containing aluminum, calcium or magnesium and preparations containing iron, zinc, or sodium bicarbonate.
Concurrent use of tetracycline may render oral contraceptives less effective.
DESCRIPTION
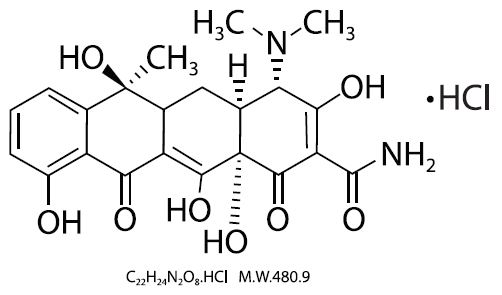
Tetracycline is a yellow, crystalline powder. Tetracycline is soluble in water, slightly soluble in ethanol (96%), practically insoluble in acetone. It dissolves in solutions of alkali hydroxides and carbonates. Solutions in water become turbid on standing, owing to the precipitation of tetracycline. The chemical name for tetracycline hydrochloride is 4-(Dimethylamino)-1,4,4a,5,5a,6,11,12a octahydro-3,6,10,12,-12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecar-boxamide monohydrochloride. Its structural formula is as follows:
Each tablet, for oral administration, contains 250 mg or 500 mg tetracycline hydrochloride.
Inactive Ingredients: anhydrous lactose, magnesium stearate, microcrystalline cellulose, povidone, pregelatinized starch and stearic acid. The film coating for the 250 mg and 500 mg are made of D&C RED # 30 / helendon pink aluminium lake, hypromellose and titanium dioxide.
In addition to these, the 250 mg tablet film coating includes triacetin and 500 mg tablet film coating includes polyethylene glycol.
CLINICAL PHARMACOLOGY
Tetracyclines are readily absorbed and are bound to plasma protein in varying degrees. They are concentrated by the liver in the bile and excreted in the urine and feces at high concentrations in a biologically active form.
Microbiology
Tetracyclines are primarily bacteriostatic and exert their antimicrobial effect by the inhibition of protein synthesis by binding to the 30S ribosomal subunit. Tetracycline is active against a broad range of gram-negative and gram-positive organisms. The drugs in the tetracycline class have closely similar antimicrobial spectra, and cross-resistance among them is common.
Tetracycline has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section of the package insert.
Gram-negative Bacteria
Acinetobacter species
Bartonella bacilliformis
Brucella species
Campylobacter fetus
Enterobacter aerogenes
Escherichia coli
Francisella tularensis
Haemophilus ducreyi
Haemophilus influenzae
Klebsiella species
Klebsiella granulomatis
Neisseria gonorrhoeae
Shigella species
Vibrio cholerae
Yersinia pestis
Gram-positive Bacteria
Bacillus anthracis
Streptococcus pyogenes
Streptococcus pneumoniae
Staphylococcus aureus
Listeria monocytogenes
Anaerobes
Bacteroides species
Clostridium species
Fusobacterium fusiforme
Propionibacterium acnes
Other Bacteria
Actinomyces species
Borrelia recurrentis
Chlamydophila psittaci
Chlamydia trachomatis
Rickettsiae
Treponema pallidum
Treponema pallidum subspecies pertenue
Parasites
Entamoeba species
Balantidium coli
Susceptibility Test Methods
When available, the clinical microbiology laboratory should provide the results of in vitro susceptibility test results for antimicrobial drugs used in resident hospitals to the physician as periodic reports that describe the susceptibility profile of nosocomial and community-acquired pathogens. These reports should aid the physician in selecting the most effective antimicrobial.
Dilution Techniques
Quantitative methods are used to determine antimicrobial minimum inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized test method 1,2,4 (broth and/or agar). The MIC values should be interpreted according to the criteria provided in Table 1.
Diffusion Techniques
Quantitative methods that require measurement of zone diameters can also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. The zone size provides an estimate of the susceptibility of bacteria to antimicrobial compounds. The zone size should be determined using a standard test method 1,3,4 . This procedure uses paper disks impregnated with 30 mcg tetracycline to test the susceptibility of bacteria to tetracycline. The disk diffusion interpretive criteria are provided in Table 1.
Anaerobic Techniques
For anaerobic bacteria, the susceptibility to tetracycline can be determined by a standardized test method 5 . The MIC values obtained should be interpreted according to the criteria provided in Table 1.
| Table 1: Susceptibility Test Interpretive Criteria for Tetracycline | |||||||||
| Bacteria a | Minimal Inhibitory Concentration (mcg/mL) | Zone Diameter (mm) | Agar Dilution (mcg/mL) | ||||||
| S | I | R | S | I | R | S | I | R | |
| Acinetobacter spp. | <4 | 8 | >16 | >15 | 12-14 | <11 | - | - | - |
| Anaerobes | - | - | - | - | - | - | <4 | 8 | >16 |
| Bacillusanthracis a | <1 | - | - | - | - | - | - | - | - |
| Brucella species a | <1 | - | - | - | - | - | - | - | - |
| Enterobacteriaceae | <4 | 8 | >16 | >15 | 12-14 | <11 | - | - | - |
| Franciscellatularensis a | <4 | - | - | - | - | - | - | - | - |
| Haemophilus influenza e | <2 | 4 | >8 | >29 | 26-28 | <25 | - | - | - |
| Mycoplasma pneumoniae | - | - | - | - | - | - | < 2 | - | - |
| Neisseria gonorrhoeae b | - | - | - | >38 | 31-37 | <30 | <0.25 | 0.5-1 | >2 |
| Staphylococcus aureus | <4 | 8 | >16 | >19 | 15-18 | <14 | - | - | - |
| Streptococcus pneumoniae | <1 | 2 | >4 | >28 | 25-27 | <24 | - | - | - |
| Streptococcus pyogenes | <2 | 4 | >8 | >23 | 19-22 | <18 | - | - | - |
| Vibrio cholerae | <4 | 8 | >16 | >15 | 12-14 | <11 | - | - | - |
| Yersinia pestis | <4 | 8 | >16 | - | - | - | - | - | - |
| a The current absence of resistance isolates precludes defining any results other than "Susceptible". If isolates yielding MIC results other than susceptible, they should be submitted to a reference laboratory for further testing. b Gonococci with 30 mcg tetracycline disk zone diameters of less than 19 mm usually indicate a plasmid- mediated tetracycline resistant Neisseria gonorrhoeae isolate. Resistance in these strains should be confirmed by a dilution test (MIC greater than or equal to 16 mcg/mL). | |||||||||
A report of Susceptible (S) indicates that the antimicrobial is likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations usually achievable at the site of infection. A report of Intermediate (I) indicates that the result should be considered equivocal, and, if the bacteria is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone that prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of Resistant (R) indicates that the antimicrobial is not likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations usually achievable at the infection site; other therapy should be selected.
Quality Control
Standardized susceptibility test procedures require the use of laboratory controls to monitor and ensure the accuracy and precision of the supplies and reagents used in the assay, and the techniques of the individuals performing the test 1,2,3,4,5,6,7 . Standard tetracycline powders should provide the following range of MIC values noted in Table 2. For the diffusion technique using the 30 mcg tetracycline disk the criteria noted in Table 2 should be achieved.
| Table 2: Acceptable Quality Control Ranges for Susceptibility Testing for Tetracycline | |||
| QC Strain | Minimal Inhibitory Concentration (mcg/mL) | Zone Diameter (mm) | Agar Dilution (mcg/mL) |
| Enterococcus faecalis ATCC 29212 | 8 - 32 | - | - |
| Escherichia coli ATCC 25922 | 0.5 - 2 | 18 - 25 | - |
| Haemophilus influenzae ATCC 49247 | 4 - 32 | 14 - 22 | - |
| Mycoplasma pneumoniae ATCC 29342 | 0.06-0.5 | - | 0.06-0.5 |
| Neisseria gonorrhoeae ATCC 49226 | - | 30 - 42 | 0.25 - 1 |
| Staphylococcus aureus ATCC 25923 | - | 24 - 30 | - |
| Staphylococcus aureus ATCC 29213 | 0.12 - 1 | - | - |
| Streptococcus pneumoniae ATCC 49619 | 0.06 - 0.5 | 27 - 31 | - |
| Bacteroides fragilis ATCC 25285 | - | - | 0.12 - 0.5 |
| Bacteroides thetaiotaomicron ATCC 29741 | - | - | 8 - 32 |
HOW SUPPLIED
Tetracycline Hydrochloride Tablets, USP are available as:
250 mg: Light pink, ovaloid, coated tablet debossed with "250" on one side and plain on the other side.
Available in bottles of:
60 NDC 82644-087-60
500 mg: Pink, ovaloid, coated tablet debossed with "500" on one side and plain on the other side.
Available in bottles of:
60 NDC 82644-870-60
Dispense in a tight, light-resistant containers as defined in the USP. Use child-resistant closure (as required).
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.