Get your patient on Timolol Maleate - Timolol Maleate solution/ Drops (Timolol Maleate)
Timolol Maleate - Timolol Maleate solution/ Drops prescribing information
INDICATIONS AND USAGE
Timolol maleate ophthalmic solution is indicated in the treatment of elevated intraocular pressure in patients with ocular hypertension or open-angle glaucoma.
DOSAGE AND ADMINISTRATION
Timolol maleate ophthalmic solution is available in concentrations of 0.25 and 0.5%. The usual starting dose is one drop of timolol maleate ophthalmic solution 0.25% in the affected eye(s) twice a day. If the clinical response is not adequate, the dosage may be changed to one drop of 0.5% solution in the affected eye(s) twice a day.
Since in some patients the pressure-lowering response to timolol maleate ophthalmic solution may require a few weeks to stabilize, evaluation should include a determination of intraocular pressure after approximately 4 weeks of treatment with timolol maleate ophthalmic solution.
If the intraocular pressure is maintained at satisfactory levels, the dosage schedule may be changed to one drop once a day in the affected eye(s). Because of diurnal variations in intraocular pressure, satisfactory response to the once-a-day dose is best determined by measuring the intraocular pressure at different times during the day.
Dosages above one drop of timolol maleate ophthalmic solution 0.5% twice a day generally have not been shown to produce further reduction in intraocular pressure. If the patient's intraocular pressure is still not at a satisfactory level on this regimen, concomitant therapy with other agent(s) for lowering intraocular pressure can be instituted. The concomitant use of two topical beta-adrenergic blocking agents is not recommended [see PRECAUTIONS , Drug Interactions , Beta-adrenergic blocking agents ].
CONTRAINDICATIONS
Timolol maleate ophthalmic solution is contraindicated in patients with (1) bronchial asthma; (2) a history of bronchial asthma; (3) severe chronic obstructive pulmonary disease [see WARNINGS ]; (4) sinus bradycardia; (5) second or third degree atrioventricular block; (6) overt cardiac failure [see WARNINGS ]; (7) cardiogenic shock; or (8) hypersensitivity to any component of this product.
ADVERSE REACTIONS
The most frequently reported adverse experiences have been burning and stinging upon instillation (approximately one in eight patients).
The following additional adverse experiences have been reported less frequently with ocular administration of this or other timolol maleate formulations:
Body as a Whole
Headache, asthenia/fatigue, and chest pain.
Cardiovascular
Bradycardia, arrhythmia, hypotension, hypertension, syncope, heart block, cerebral vascular accident, cerebral ischemia, cardiac failure, worsening of angina pectoris, palpitation, cardiac arrest, pulmonary edema, edema, claudication, Raynaud's phenomenon, and cold hands and feet.
Digestive
Nausea, diarrhea, dyspepsia, anorexia, and dry mouth.
Immunologic
Systemic lupus erythematosus.
Nervous System/Psychiatric
Dizziness, increase in signs and symptoms of myasthenia gravis, paresthesia, somnolence, insomnia, nightmares, behavioral changes and psychic disturbances including depression, confusion, hallucinations, anxiety, disorientation, nervousness, and memory loss.
Skin
Alopecia and psoriasiform rash or exacerbation of psoriasis.
Hypersensitivity
Signs and symptoms of systemic allergic reactions, including anaphylaxis, angioedema, urticaria, and localized and generalized rash.
Respiratory
Bronchospasm (predominantly in patients with preexisting bronchospastic disease), respiratory failure, dyspnea, nasal congestion, cough and upper respiratory infections.
Endocrine
Masked symptoms of hypoglycemia in diabetic patients [see WARNINGS ] .
Special Senses
Signs and symptoms of ocular irritation including conjunctivitis, blepharitis, keratitis, ocular pain, discharge (e.g., crusting), foreign body sensation, itching and tearing, and dry eyes; ptosis; decreased corneal sensitivity; cystoid macular edema; visual disturbances including refractive changes and diplopia; pseudopemphigoid; choroidal detachment following filtration surgery [see PRECAUTIONS , General ]; and tinnitus.
Urogenital
Retroperitoneal fibrosis, decreased libido, impotence, and Peyronie's disease.
The following additional adverse effects have been reported in clinical experience with ORAL timolol maleate or other ORAL beta-blocking agents and may be considered potential effects of ophthalmic timolol maleate: Allergic: Erythematous rash, fever combined with aching and sore throat, laryngospasm with respiratory distress; Body as a Whole: Extremity pain, decreased exercise tolerance, weight loss; Cardiovascular: Worsening of arterial insufficiency, vasodilatation; Digestive: Gastrointestinal pain, hepatomegaly, vomiting, mesenteric arterial thrombosis, ischemic colitis; Hematologic: Nonthrombocytopenic purpura; thrombocytopenic purpura, agranulocytosis; Endocrine: Hyperglycemia, hypoglycemia; Skin: Pruritus, skin irritation, increased pigmentation, sweating; Musculoskeletal: Arthralgia; Nervous System/Psychiatric: Vertigo, local weakness, diminished concentration, reversible mental depression progressing to catatonia, an acute reversible syndrome characterized by disorientation for time and place, emotional lability, slightly clouded sensorium, and decreased performance on neuropsychometrics; Respiratory: Rales, bronchial obstruction; Urogenital: Urination difficulties.
To report SUSPECTED ADVERSE REACTIONS, contact First Nation Group, LLC at l-855-221-5332 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Timolol maleate ophthalmic solution, USP is a non-selective beta-adrenergic receptor blocking agent. Its chemical name is (-)-1-( tert -butylamino)-3-[(4-morpholino-1,2,5-thiadiazol-3-yl)oxy]-2-propanol maleate (1:1) (salt). Timolol maleate, USP possesses an asymmetric carbon atom in its structure and is provided as the levo-isomer. The optical rotation of timolol maleate is:
25°
[α] in 1.0N HCl (C = 5%) = -12.2° (-11.7° to -12.5°)
405 nm
Its molecular formula is C 13 H 24 N 4 O 3 S•C 4 H 4 O 4 and its structural formula is:
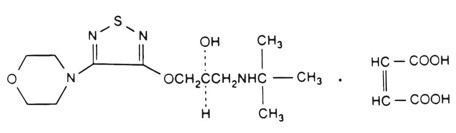
Timolol maleate has a molecular weight of 432.50. It is a white to almost white crystalline powder which is soluble in water; sparingly soluble in ethanol; slightly soluble in chloroform; practically insoluble in ether. Timolol maleate ophthalmic solution is stable at room temperature.
Timolol maleate ophthalmic solution, USP is supplied as a sterile, isotonic, buffered, aqueous solution of timolol maleate in two dosage strengths. Each mL of timolol maleate ophthalmic solution 0.25% contains 2.5 mg of timolol (3.4 mg of timolol maleate). The pH of the solution is approximately 7.0, and the osmolality is 270-330 mOsm/kg. Each mL of timolol maleate ophthalmic solution 0.5% contains 5 mg of timolol (6.8 mg of timolol maleate). Inactive ingredients: monobasic and dibasic sodium phosphate, sodium chloride, sodium hydroxide to adjust pH, and water for injection. Benzalkonium chloride 0.01% is added as preservative.
CLINICAL PHARMACOLOGY
Mechanism of Action
Timolol maleate is a beta 1 and beta 2 (non-selective) adrenergic receptor blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anesthetic (membrane-stabilizing) activity.
Beta-adrenergic receptor blockade reduces cardiac output in both healthy subjects and patients with heart disease. In patients with severe impairment of myocardial function, beta-adrenergic receptor blockade may inhibit the stimulatory effect of the sympathetic nervous system necessary to maintain adequate cardiac function.
Beta-adrenergic receptor blockade in the bronchi and bronchioles results in increased airway resistance from unopposed parasympathetic activity. Such an effect in patients with asthma or other bronchospastic conditions is potentially dangerous.
Timolol maleate ophthalmic solution, when applied topically on the eye, has the action of reducing elevated as well as normal intraocular pressure, whether or not accompanied by glaucoma.
Elevated intraocular pressure is a major risk factor in the pathogenesis of glaucomatous visual field loss. The higher the level of intraocular pressure, the greater the likelihood of glaucomatous visual field loss and optic nerve damage.
The onset of reduction in intraocular pressure following administration of timolol maleate ophthalmic solution can usually be detected within one-half hour after a single dose. The maximum effect usually occurs in one to two hours and significant lowering of intraocular pressure can be maintained for periods as long as 24 hours with a single dose. Repeated observations over a period of one year indicate that the intraocular pressure-lowering effect of timolol maleate ophthalmic solution is well maintained.
The precise mechanism of the ocular hypotensive action of timolol maleate ophthalmic solution is not clearly established at this time. Tonography and fluorophotometry studies in man suggest that its predominant action may be related to reduced aqueous formation. However, in some studies a slight increase in outflow facility was also observed.
Pharmacokinetics
In a study of plasma drug concentration in six subjects, the systemic exposure to timolol was determined following twice daily administration of timolol maleate ophthalmic solution 0.5%. The mean peak plasma concentration following morning dosing was 0.46 ng/mL and following afternoon dosing was 0.35 ng/mL.
Clinical Studies
In controlled multiclinic studies in patients with untreated intraocular pressures of 22 mmHg or greater, timolol maleate ophthalmic solution 0.25% or 0.5% administered twice a day produced a greater reduction in intraocular pressure than 1, 2, 3, or 4% pilocarpine solution administered four times a day or 0.5, 1, or 2% epinephrine hydrochloride solution administered twice a day.
In these studies, timolol maleate ophthalmic solution was generally well tolerated and produced fewer and less severe side effects than either pilocarpine or epinephrine. A slight reduction of resting heart rate in some patients receiving timolol maleate ophthalmic solution (mean reduction 2.9 beats/minute standard deviation 10.2) was observed.
HOW SUPPLIED
Timolol maleate ophthalmic solution, USP is a clear, colorless solution.
Timolol maleate ophthalmic solution 0.25% timolol equivalent is supplied in a Natural LDPE bottle with a Natural LDPE controlled drop tip and a Yellow HDPE cap as follows:
NDC 81469-209-05 5 mL in a 5 mL capacity bottle
NDC 81469-209-10 10 mL in a 10 mL capacity bottle
Timolol maleate ophthalmic solution 0.5% timolol equivalent is supplied in a Natural LDPE bottle with a Natural LDPE controlled drop tip and a Yellow HDPE cap as follows:
NDC 81469-210-05 5 mL in a 5 mL capacity bottle
NDC 81469-210-10 10 mL in a 10 mL capacity bottle
Storage
Store at 20°C to 25°C (68°F to 77°F) [See USP Controlled Room Temperature]. Protect from freezing. Protect from light. After opening, timolol maleate ophthalmic solution can be used until the expiration date on the bottle.
Manufactured for:
First Nation Group, LLC
4566 E Highway 20, Suite #208
Niceville, FL 32578-8839
Product of Germany
Revised: January 2024, V-01
Instructions for Use
Timolol Maleate Ophthalmic Solution, USP 0.25% and 0.5%
(tim oh lol mal ee ate)
Read this Instructions for Use that comes with timolol maleate ophthalmic solution before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment.
Important information about timolol maleate ophthalmic solution:
- Use timolol maleate ophthalmic solution exactly as your doctor tells you to use it. Your doctor will tell you how much timolol maleate ophthalmic solution to use and when to use it.
- If you use other medicines in your eye, wait at least 10 minutes between using timolol maleate ophthalmic solution and your other eye medicines.
- Do not touch your eye or areas around your eye with the tip of the timolol maleate ophthalmic solution bottle. You may get bacteria on the tip of the bottle that can cause you to get an eye infection that can lead to serious eye damage or vision loss.
How should I use timolol maleate ophthalmic solution?
Step 1. Wash your hands.
Step 2. Turn your closed bottle of timolol maleate ophthalmic solution upside down (invert) and shake once.
Step 3. Remove the timolol maleate ophthalmic solution cap by turning the cap in the direction of the arrows shown ( See Figure A ). Put the cap in a clean and dry area. Do not let the tip of the bottle touch your fingers or other surfaces.

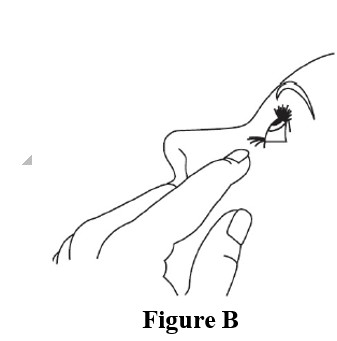
Step 4. Hold the bottle between your thumb and index finger with 1 hand. Use the index finger of the other hand to pull down the lower eyelid to form a pocket for the eye drop ( See Figure B ).Tilt your head backwards.
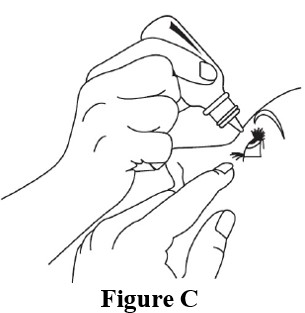
Step 5. Place the tip of the bottle close to your eye. Be careful not to touch your eye with the tip of the bottle . Gently squeeze the bottle and let 1 drop fall into the space between your lower eyelid and your eye ( See Figure C ). If a drop misses your eye, follow the instructions in steps 4 and 5 again.
Step 6. If your doctor has told you to use timolol maleate ophthalmic solution in both eyes, repeat steps 4 and 5 for your other eye.
Step 7. Put the cap back on the bottle and close.
- The timolol maleate ophthalmic solution bottle tip is made to give 1 drop at a time. Do not try to make the hole in the tip of your bottle bigger.
- Do not wash the bottle tip.
- After you have used all of your timolol maleate ophthalmic solution doses, there will be some timolol maleate ophthalmic solution left in the bottle. Do not try to remove the extra timolol maleate ophthalmic solution from the bottle. Throw it away.
How should I store timolol maleate ophthalmic solution?
- Store timolol maleate ophthalmic solution at room temperature between 20°C to 25°C (68°F to 77°F) in an upright position.
- Do not freeze timolol maleate ophthalmic solution.
- Keep timolol maleate ophthalmic solution out of light.
Keep timolol maleate ophthalmic solution and all medicines out of the reach of children.
If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for more information about timolol maleate ophthalmic solution that is written for health professionals.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Rx only
Manufactured for:
First Nation Group, LLC
4566 E Highway 20, Suite #208
Niceville, FL 32578-8839
Product of Germany
Revised: January 2024, V-01