Get your patient on Tobradex - Tobramycin And Dexamethasone ointment (Tobramycin And Dexamethasone)
Tobradex - Tobramycin And Dexamethasone ointment prescribing information
INDICATIONS AND USAGE: TOBRADEX (tobramycin and dexamethasone ophthalmic ointment) is indicated for steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where superficial bacterial ocular infection or a risk of bacterial ocular infection exists.
Ocular steroids are indicated in inflammatory conditions of the palpebral and bulbar conjunctiva, cornea and anterior segment of the globe where the inherent risk of steroid use in certain infective conjunctivitides is accepted to obtain a diminution in edema and inflammation. They are also indicated in chronic anterior uveitis and corneal injury from chemical, radiation or thermal burns, or penetration of foreign bodies.
The use of a combination drug with an anti-infective component is indicated where the risk of superficial ocular infection is high or where there is an expectation that potentially dangerous numbers of bacteria will be present in the eye.
The particular anti-infective drug in this product is active against the following common bacterial eye pathogens:
Staphylococci , including S. aureus and S. epidermidis (coagulase-positive and coagulase-negative), including penicillin-resistant strains.
Streptococci , including some of the Group A-beta-hemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae .
Pseudomonas aeruginosa , Escherichia coli , Klebsiella pneumoniae , Enterobacter aerogenes , Proteus mirabilis , Morganella morganii , most Proteus vulgaris strains, Haemophilus influenzae and H. aegyptius , Moraxella lacunata , Acinetobacter calcoaceticus and some Neisseria species.
DOSAGE AND ADMINISTRATION: Apply a small amount (approximately ½ inch ribbon) into the conjunctival sac(s) up to three or four times daily.
How to apply TOBRADEX ® (tobramycin and dexamethasone ophthalmic ointment):
1. Tilt your head back.
2. Place a finger on your cheek just under your eye and gently pull down until a "V" pocket is formed between your eyeball and your lower lid.
3. Place a small amount (about ½ inch) of TOBRADEX ® (tobramycin and dexamethasone ophthalmic ointment). Do not let the tip of the tube touch your eye.
4. Look downward before closing your eye.
Not more than 8 g should be prescribed initially and the prescription should not be refilled without further evaluation as outlined in PRECAUTIONS above.
CONTRAINDICATIONS: Epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, and many other viral diseases of the cornea and conjunctiva. Mycobacterial infection of the eye. Fungal diseases of ocular structures. Hypersensitivity to a component of the medication.
ADVERSE REACTIONS: Adverse reactions have occurred with steroid/anti-infective combination drugs which can be attributed to the steroid component, the anti-infective component, or the combination. Exact incidence figures are not available.
The most frequent adverse reactions to topical ocular tobramycin [TOBREX ® (tobramycin ophthalmic ointment) 0.3%] are hypersensitivity and localized ocular toxicity, including lid itching and swelling, and conjunctival erythema. These reactions occur in less than 4% of patients.
The reactions due to the steroid component are: elevation of intraocular pressure with possible development of glaucoma, and infrequent optic nerve damage; posterior subcapsular cataract formation; and delayed wound healing.
Secondary Infection . The development of secondary infection has occurred after use of combinations containing steroids and antimicrobials. Fungal infections of the cornea are particularly prone to develop coincidentally with long-term applications of steroids. The possibility of fungal invasion must be considered in any persistent corneal ulceration where steroid treatment has been used. Secondary bacterial ocular infection following suppression of host responses also occurs.
Postmarketing Experience: Additional adverse reactions identified from postmarketing use include, anaphylactic reaction, erythema multiforme.
The following additional adverse reactions have been reported with the individual components listed below:
Dexamethasone : Cushing’s syndrome and adrenal suppression may occur after use of dexamethasone in excess of the listed dosing instructions in predisposed patients, including children and patients treated with CYP3A4 inhibitors.
Aminoglycosides : Neurotoxicity, ototoxicity, and nephrotoxicity have occurred in patients receiving systemic aminoglycoside therapy. Aminoglycosides may aggravate muscle weakness in patients with known or suspected neuromuscular disorders, such as myasthenia gravis or Parkinson’s disease, because of their potential effect on neuromuscular function.
DESCRIPTION: TOBRADEX ® (tobramycin and dexamethasone ophthalmic ointment) is a sterile, multiple dose antibiotic and steroid combination for topical ophthalmic use. The chemical structures for tobramycin and dexamethasone are presented below:
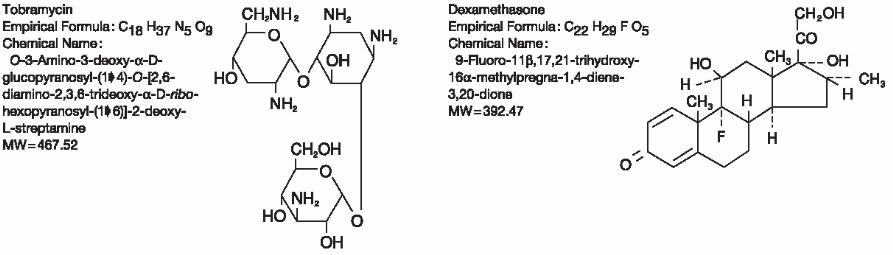
Each gram of TOBRADEX ® (tobramycin and dexamethasone ophthalmic ointment) contains: Actives: tobramycin 0.3% (3 mg) and dexamethasone 0.1% (1 mg). Preservative: chlorobutanol 0.5%. Inactives: mineral oil and white petrolatum.
CLINICAL PHARMACOLOGY: Corticoids suppress the inflammatory response to a variety of agents and they probably delay or slow healing. Since corticoids may inhibit the body's defense mechanism against infection, a concomitant antimicrobial drug may be used when this inhibition is considered to be clinically significant. Dexamethasone is a potent corticoid.
The antibiotic component in the combination (tobramycin) is included to provide action against susceptible organisms. In vitro studies have demonstrated that tobramycin is active against susceptible strains of the following microorganisms:
Staphylococci , including S. aureus and S. epidermidis (coagulase-positive and coagulase-negative), including penicillin-resistant strains.
Streptococci , including some of the Group A-beta-hemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae .
Pseudomonas aeruginosa , Escherichia coli , Klebsiella pneumoniae , Enterobacter aerogenes , Proteus mirabilis , Morganella morganii , most Proteus vulgaris strains, Haemophilus influenzae and H. aegyptius , Moraxella lacunata , Acinetobacter calcoaceticus and some Neisseria species.
No data are available on the extent of systemic absorption from TOBRADEX ® (tobramycin and dexamethasone ophthalmic ointment); however, it is known that some systemic absorption can occur with ocularly applied drugs.
HOW SUPPLIED: 3.5 g STERILE ointment supplied in an aluminum tube with a white polyethylene tip and white polyethylene cap (NDC 0078-0876-01)
STORAGE: Store at 2°C to 25°C (36°F to 77°F).
After opening, TOBRADEX (tobramycin and dexamethasone ophthalmic ointment) can be used until the expiration date on the tube.
Distributed by: Novartis Pharmaceuticals Corporation East Hanover, New Jersey 07936
© Novartis
T2021-62
May 2021