Tobramycin - Tobramycin Sulfate injection, Solution prescribing information
WARNINGS
Patients treated with Tobramycin Injection, USP and other aminoglycosides should be under close clinical observation, because these drugs have an inherent potential for causing ototoxicity and nephrotoxicity.
Neurotoxicity, manifested as both auditory and vestibular ototoxicity, can occur. The auditory changes are irreversible, are usually bilateral, and may be partial or total. Eighth-nerve impairment and nephrotoxicity may develop, primarily in patients having pre-existing renal damage and in those with normal renal function to whom aminoglycosides are administered for longer periods or in higher doses than those recommended. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching, and convulsions. The risk of aminoglycoside-induced hearing loss increases with the degree of exposure to either high peak or high trough serum concentrations. Patients who develop cochlear damage may not have symptoms during therapy to warn them of eighth-nerve toxicity, and partial or total irreversible bilateral deafness may continue to develop after the drug has been discontinued.
Rarely, nephrotoxicity may not become apparent until the first few days after cessation of therapy. Aminoglycoside-induced nephrotoxicity usually is reversible.
Renal and eighth-nerve function should be closely monitored in patients with known or suspected renal impairment and also in those whose renal function is initially normal but who develop signs of renal dysfunction during therapy. Peak and trough serum concentrations of aminoglycosides should be monitored periodically during therapy to assure adequate levels and to avoid potentially toxic levels. Prolonged serum concentrations above 12 mcg/mL should be avoided. Rising trough levels (above 2 mcg/mL) may indicate tissue accumulation. Such accumulation, excessive peak concentrations, advanced age, and cumulative dose may contribute to ototoxicity and nephrotoxicity (see PRECAUTIONS ). Urine should be examined for decreased specific gravity and increased excretion of protein, cells, and casts. Blood urea nitrogen, serum creatinine, and creatinine clearance should be measured periodically. When feasible, it is recommended that serial audiograms be obtained in patients old enough to be tested, particularly high-risk patients. Evidence of impairment of renal, vestibular, or auditory function requires discontinuation of the drug or dosage adjustment.
Tobramycin should be used with caution in premature and neonatal infants because of their renal immaturity and the resulting prolongation of serum half-life of the drug.
Concurrent and sequential use of other neurotoxic and/or nephrotoxic antibiotics, particularly other aminoglycosides (e.g., amikacin, streptomycin, neomycin, kanamycin, gentamicin, and paromomycin), cephaloridine, viomycin, polymyxin B, colistin, cisplatin, and vancomycin, should be avoided. Other factors that may increase patient risk are advanced age and dehydration.
Aminoglycosides should not be given concurrently with potent diuretics, such as ethacrynic acid and furosemide. Some diuretics themselves cause ototoxicity, and intravenously administered diuretics enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue.
Aminoglycosides can cause fetal harm when administered to a pregnant woman (see PRECAUTIONS ).
INDICATIONS AND USAGE:
Tobramycin is indicated for the treatment of serious bacterial infections caused by susceptible strains of the designated microorganisms in the diseases listed below:
Septicemia in the neonate, child, and adult caused by P. aeruginosa , E. coli , and Klebsiella sp.
Lower respiratory tract infections caused by P. aeruginos a, Klebsiella sp, Enterobacter sp, Serratia sp, E. coli , and S. aureus (penicillinase- and non-penicillinase-producing strains).
Serious central-nervous-system infections (meningitis) caused by susceptible organisms.
Intra-abdominal infections, including peritonitis, caused by E. coli , Klebsiella sp, and Enterobacter sp. Skin, bone, and skin-structure infections caused by P. aeruginosa , Proteus sp, E. coli , Klebsiella sp,
Enterobacter sp, and S. aureus.
Complicated and recurrent urinary tract infections caused by P. aeruginosa , Proteus sp (indole-positive and indole-negative), E. coli , Klebsiella sp, Enterobacter sp, Serratia sp, S. aureus , Providencia sp, and Citrobacter sp.
Aminoglycosides, including tobramycin sulfate, are not indicated in uncomplicated initial episodes of urinary tract infections unless the causative organisms are not susceptible to antibiotics having less potential toxicity. Tobramycin may be considered in serious staphylococcal infections when penicillin or other potentially less toxic drugs are contraindicated and when bacterial susceptibility testing and clinical judgment indicate its use.
Bacterial cultures should be obtained prior to and during treatment to isolate and identify etiologic organisms and to test their susceptibility to tobramycin. If susceptibility tests show that the causative organisms are resistant to tobramycin, other appropriate therapy should be instituted. In patients in whom a serious life-threatening gram-negative infection is suspected, including those in whom concurrent therapy with a penicillin or cephalosporin and an aminoglycoside may be indicated, treatment with tobramycin sulfate may be initiated before the results of susceptibility studies are obtained. The decision to continue therapy with tobramycin should be based on the results of susceptibility studies, the severity of the infection, and the important additional concepts discussed in the WARNINGS box above.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of tobramycin and other antibacterial drugs, tobramycin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antimicrobial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION:
This package insert labeling is for a Pharmacy Bulk Package and is intended for preparing intravenous admixtures only. Dosage recommendations and/or references for the intramuscular route of administration are for informational purposes only.
Tobramycin Injection, USP may be given intramuscularly or intravenously. Recommended dosages are the same for both routes. The patient's pretreatment body weight should be obtained for calculation of correct dosage. It is desirable to measure both peak and trough serum concentrations (see WARNINGS box and PRECAUTIONS ).
Administration for Patients with Normal Renal Function
Adults with Serious Infections: 3 mg/kg/day in 3 equal doses every 8 hours (see Table 3 ).
Adults with Life-Threatening Infections: Up to 5 mg/kg/day may be administered in 3 or 4 equal doses (see Table 3 ). The dosage should be reduced to 3 mg/kg/day as soon as clinically indicated. To prevent
increased toxicity due to excessive blood levels, dosage should not exceed 5 mg/kg/day unless serum levels are monitored (see WARNINGS box and PRECAUTIONS ).
| •Applicable to all product forms except Tobramycin Injection, USP, 10 mg/mL (Pediatric ). | |||
For Patient Weighing | Usual Dose for Serious Infections | ||
kg | lb | 1 mg/kg q8h (Total, 3 mg/kg/day) | |
mg/dose | mL/dose• | ||
q8h | |||
120 | 264 | 120 mg | 3 mL |
115 | 253 | 115 mg | 2.9 mL |
110 | 242 | 110 mg | 2.75 mL |
105 | 231 | 105 mg | 2.6 mL |
100 | 220 | 100 mg | 2.5 mL |
95 | 209 | 95 mg | 2.4 mL |
90 | 198 | 90 mg | 2.25 mL |
85 | 187 | 85 mg | 2.1 mL |
80 | 176 | 80 mg | 2 mL |
75 | 165 | 75 mg | 1.9 mL |
70 | 154 | 70 mg | 1.75 mL |
65 | 143 | 65 mg | 1.6 mL |
60 | 132 | 60 mg | 1.5 mL |
55 | 121 | 55 mg | 1.4 mL |
50 | 110 | 50 mg | 1.25 mL |
45 | 99 | 45 mg | 1.1 mL |
40 | 88 | 40 mg | 1 mL |
For Patient Weighing | Maximum Dose for Life- Threatening Infections (Reduce as soon as possible) | ||
kg | lb | 1.66 mg/kg q8h (Total, 5 mg/kg/day) | |
mg/dose | mL/dose• | ||
q8h | |||
120 | 264 | 200 mg | 5 mL |
115 | 253 | 191 mg | 4.75 mL |
110 | 242 | 183 mg | 4.5 mL |
105 | 231 | 175 mg | 4.4 mL |
100 | 220 | 166 mg | 4.2 mL |
95 | 209 | 158 mg | 4 mL |
90 | 198 | 150 mg | 3.75 mL |
85 | 187 | 141 mg | 3.5 mL |
80 | 176 | 133 mg | 3.3 mL |
75 | 165 | 125 mg | 3.1 mL |
70 | 154 | 116 mg | 2.9 mL |
65 | 143 | 108 mg | 2.7 mL |
60 | 132 | 100 mg | 2.5 mL |
55 | 121 | 91 mg | 2.25 mL |
50 | 110 | 83 mg | 2.1 mL |
45 | 99 | 75 mg | 1.9 mL |
40 | 88 | 66 mg | 1.6 mL |
Pediatric Patients (Greater Than 1 Week of Age): 6 to 7.5 mg/kg/day in 3 or 4 equally divided doses (2 to 2.5 mg/kg every 8 hours or 1.5 to 1.89 mg/kg every 6 hours).
Premature or Full-Term Neonates 1 Week of Age or Less: Up to 4 mg/kg/day may be administered in 2 equal doses every 12 hours.
It is desirable to limit treatment to a short term. The usual duration of treatment is 7 to 10 days. A longer course of therapy may be necessary in difficult and complicated infections. In such cases, monitoring of renal, auditory, and vestibular functions is advised, because neurotoxicity is more likely to occur when treatment is extended longer than 10 days.
Dosage in Patients with Cystic Fibrosis
In patients with cystic fibrosis, altered pharmacokinetics may result in reduced serum concentrations of aminoglycosides. Measurement of tobramycin serum concentration during treatment is especially important as a basis for determining appropriate dose. In patients with severe cystic fibrosis, an initial dosing regimen of 10 mg/kg/day in 4 equally divided doses is recommended. This dosing regimen is suggested only as a guide. The serum levels of tobramycin should be measured directly during treatment due to wide interpatient variability.
Administration for Patients with Impaired Renal Function
Whenever possible, serum tobramycin concentrations should be monitored during therapy.
Following a loading dose of 1 mg/kg, subsequent dosage in these patients must be adjusted, either with reduced doses administered at 8-hour intervals or with normal doses given at prolonged intervals. Both of these methods are suggested as guides to be used when serum levels of tobramycin cannot be measured directly. They are based on either the creatinine clearance or the serum creatinine of the patient, because these values correlate with the half-life of tobramycin. The dosage schedules derived from either method should be used in conjunction with careful clinical and laboratory observations of the patient and should be modified as necessary. Neither method should be used when dialysis is being performed.
Reduced Dosage at 8-hour Intervals — When the creatinine clearance rate is 70 mL or less per minute or when the serum creatinine value is known, the amount of the reduced dose can be determined by multiplying the normal dose from Table 3 by the percent of normal dose from the accompanying nomogram.
An alternate rough guide for determining reduced dosage at 8-hour intervals (for patients whose steady-state serum creatinine values are known) is to divide the normally recommended dose by the patient's serum creatinine.
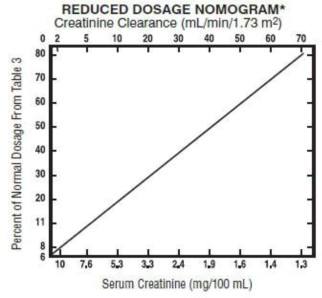
• Scales have been adjusted to facilitate dosage calculations.
Normal Dosage at Prolonged Intervals — If the creatinine clearance rate is not available and the patient's condition is stable, a dosage frequency in hours for the dosage given in Table 3 can be determined by multiplying the patient's serum creatinine by 6.
Dosage in Obese Patients
The appropriate dose may be calculated by using the patient's estimated lean body weight plus 40% of the excess as the basic weight on which to figure mg/kg.
Intravenous Administration
For intravenous administration, the usual volume of diluent (0.9% Sodium Chloride Injection or 5% Dextrose Injection) is 50 to 100 mL for adult doses. For pediatric patients, the volume of diluent should be proportionately less than for adults. The diluted solution usually should be infused over a period of 20 to 60 minutes. Infusion periods of less than 20 minutes are not recommended because peak serum levels may exceed 12 mcg/mL (see WARNINGS box).
Tobramycin Injection, USP should not be physically premixed with other drugs but should be administered separately according to the recommended dose and route.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Directions for Proper Use of Pharmacy Bulk Package Use Aseptic Technique - Not for Direct Infusion
The pharmacy bulk package is for use in a Pharmacy Admixture Service only.
- For hanger use, grasp portion of the bottle label marked "LIFT HERE". Peel the laminated film (sling) away from the printed portion of the pressure sensitive label. Invert bottle and pull sling over the base of the bottle. Hang bottle using sling portion of the label.
- During use, container must be stored and all manipulations performed in an appropriate laminar flow hood.
- Remove cover from container and cleanse closure with antiseptic.
- A single entry through the vial closure should be made with a sterile dispensing set which allows measured dispensing of the contents. Transfer individual dose(s) to appropriate intravenous infusion solutions without delay. Use of a syringe with needle is not recommended as it may cause leakage. Multiple entries will also increase the potential of microbial and particulate contamination. The above process should be carried out under a laminar flow hood using aseptic technique. ANY UNUSED PORTION MUST BE DISCARDED WITHIN 4 HOURS. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS:
A hypersensitivity to any aminoglycoside is a contraindication to the use of tobramycin. A history of hypersensitivity or serious toxic reactions to aminoglycosides may also contraindicate the use of any other aminoglycoside because of the known cross-sensitivity of patients to drugs in this class.
ADVERSE REACTIONS:
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Neurotoxicity
Adverse effects on both the vestibular and auditory branches of the eighth nerve have been noted, especially in patients receiving high doses or prolonged therapy, in those given previous courses of therapy with an ototoxin, and in cases of dehydration. Symptoms include dizziness, vertigo, tinnitus, roaring in the ears, and hearing loss. Hearing loss is usually irreversible and is manifested initially by diminution of high-tone acuity. Tobramycin and gentamicin sulfates closely parallel each other in regard to ototoxic potential.
Nephrotoxicity
Renal function changes, as shown by rising BUN, NPN, and serum creatinine and by oliguria, cylindruria, and increased proteinuria, have been reported, especially in patients with a history of renal impairment who are treated for longer periods or with higher doses than those recommended. Adverse renal effects can occur in patients with initially normal renal function.
Clinical studies and studies in experimental animals have been conducted to compare the nephrotoxic potential of tobramycin and gentamicin. In some of the clinical studies and in the animal studies, tobramycin caused nephrotoxicity significantly less frequently than gentamicin. In some other clinical studies, no significant difference in the incidence of nephrotoxicity between tobramycin and gentamicin was found.
Other reported adverse reactions possibly related to tobramycin sulfate include anemia, granulocytopenia, and thrombocytopenia; and fever, rash, exfoliative dermatitis, itching, urticaria, nausea, vomiting, diarrhea, headache, lethargy, pain at the injection site, mental confusion, and disorientation. Laboratory abnormalities possibly related to tobramycin include increased serum transaminases (SGOT, SGPT); increased serum LDH and bilirubin; decreased serum calcium, magnesium, sodium, and potassium; and leukopenia, leukocytosis, and eosinophilia.
DESCRIPTION:
Tobramycin sulfate, a water-soluble antibiotic of the aminoglycoside group, is derived from the actinomycete Streptomyces tenebrarius . Tobramycin Injection, USP is a clear and colorless sterile aqueous solution for parenteral administration.
Each mL contains tobramycin sulfate equivalent to 40 mg tobramycin; sodium metabisulfite added as an antioxidant, 3.2 mg; and edetate disodium added as a stabilizer, 0.1 mg. Contains sulfuric acid and may contain sodium hydroxide for pH adjustment (3.0 to 6.5).
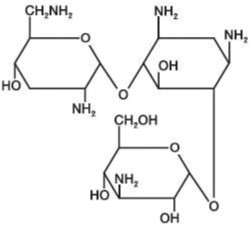
Tobramycin sulfate is, O -3-amino-3-deoxy-α-D-glucopyranosyl-(1→4)
- O -[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine (2:5)(salt) and has the chemical formula (C 18 H 37 N 5 O 9 ) 2 • 5H 2 SO 4 . The molecular weight is 1,425.42. The structural formula for tobramycin is as follows:
A pharmacy bulk package is a container of a sterile preparation for parenteral use that contains many single doses. The contents are intended for use in a pharmacy admixture service and are restricted to the preparation of admixtures for intravenous infusion or to the filling of empty sterile syringes for intravenous injection for patients with individualized dosing requirements. FURTHER DILUTION IS REQUIRED BEFORE USE (see DOSAGE AND ADMINISTRATION, Directions for proper use of Pharmacy Bulk Package ).
CLINICAL PHARMACOLOGY:
Tobramycin is rapidly absorbed following intramuscular administration. Peak serum concentrations of tobramycin occur between 30 and 90 minutes after intramuscular administration.
Following an intramuscular dose of 1 mg/kg of body weight, maximum serum concentrations reach about 4 mcg/mL, and measurable levels persist for as long as 8 hours. Therapeutic serum levels are generally considered to range from 4 to 6 mcg/mL. When tobramycin sulfate is administered by intravenous infusion over a 1-hour period, the serum concentrations are similar to those obtained by intramuscular administration. Tobramycin is poorly absorbed from the gastrointestinal tract.
In patients with normal renal function, except neonates, tobramycin administered every 8 hours does not accumulate in the serum. However, in those patients with reduced renal function and in neonates, the serum concentration of the antibiotic is usually higher and can be measured for longer periods of time than in normal adults. Dosage for such patients must, therefore, be adjusted accordingly (see DOSAGE AND ADMINISTRATION ).
Following parenteral administration, little, if any, metabolic transformation occurs, and tobramycin is eliminated almost exclusively by glomerular filtration. Renal clearance is similar to that of endogenous creatinine. Ultrafiltration studies demonstrate that practically no serum protein binding occurs. In patients with normal renal function, up to 84% of the dose is recoverable from the urine in 8 hours and up to 93% in 24 hours.
Peak urine concentrations ranging from 75 to 100 mcg/mL have been observed following the intramuscular injection of a single dose of 1 mg/kg. After several days of treatment, the amount of tobramycin excreted in the urine approaches the daily dose administered. When renal function is impaired, excretion of tobramycin sulfate is slowed, and accumulation of the drug may cause toxic blood levels.
The serum half-life in normal individuals is 2 hours. An inverse relationship exists between serum half- life and creatinine clearance, and the dosage schedule should be adjusted according to the degree of renal impairment (see DOSAGE AND ADMINISTRATION ). In patients undergoing dialysis, 25% to 70% of the administered dose may be removed, depending on the duration and type of dialysis.
Tobramycin can be detected in tissues and body fluids after parenteral administration. Concentrations in bile and stools ordinarily have been low, which suggests minimum biliary excretion. Tobramycin has appeared in low concentration in the cerebrospinal fluid following parenteral administration, and concentrations are dependent on dose, rate of penetration, and degree of meningeal inflammation. It has also been found in sputum, peritoneal fluid, synovial fluid, and abscess fluids, and it crosses the placental membranes. Concentrations in the renal cortex are several times higher than the usual serum levels.
Probenecid does not affect the renal tubular transport of tobramycin.
Microbiology
Tobramycin is an aminoglycoside antibiotic with activity against Gram-positive and Gram- negative bacteria.
Mechanism of Action
Tobramycin acts by inhibiting synthesis of protein in bacterial cells. In vitro tests demonstrate that tobramycin is bactericidal.
Interactions with Other Antibiotics
Although most strains of enterococci demonstrate in vitro resistance, some strains in this group are susceptible. In vitro studies have shown that an aminoglycoside combined with an antibiotic that interferes with cell-wall synthesis affects some enterococcal strains synergistically. The combination of penicillin G and tobramycin results in a synergistic bactericidal effect in vitro against certain strains of Enterococcus faecalis . However, this combination is not synergistic against other closely related organisms, e.g. Enterococcus faecium . Speciation of enterococci alone cannot be used to predict susceptibility. Susceptibility testing and tests for antibiotic synergism are emphasized.
Cross-Resistance
Cross-resistance between aminoglycosides may occur.
Antimicrobial Activity:
Tobramycin has been shown to be active against most strains of the following organisms both in vitro
and in clinical infections: (see INDICATIONS AND USAGE ). Aerobic and facultative Gram-positive microorganisms Staphylococcus aureus
Aerobic and facultative Gram-negative microorganisms
Citrobacter sp Enterobacter sp Escherichia coli Klebsiella sp Morganella morganii
Pseudomonas aeruginosa Proteus mirabilis
Proteus vulgaris Providencia sp Serratia sp
Aminoglycosides have a low order of activity against most Gram-positive organisms, including Streptococcus pyogenes, Streptococcus pneumoniae , and enterococci.
Susceptibility Tests
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
HOW SUPPLIED:
Tobramycin Injection USP, in the Pharmacy Bulk Package, is supplied as follows:
Product Code | Unit of Sale | Strength | Each |
300751 | NDC 63323-307-51 | 2 g per 50 mL (40 mg per mL) 50 mL fill, in a 60 mL vial | 60 mL Pharmacy Bulk Package vial, packaged individually |
Store at 20 to 25°C (68 to 77°F) [see USP Controlled Room Temperature]. The container closure is not made with natural rubber latex.
Tobramycin Injection, USP, is also available in multiple dose vials:
Product Code | Unit of Sale | Strength | Each |
300502 | NDC 63323-305-02 Unit of 25 | 20 mg per 2 mL (10 mg per mL) 2 mL fill, in a 2 mL vial. | NDC 63323-305-01 2 mL Multiple Dose Vial |
300602 | NDC 63323-306-02 Unit of 25 | 80 mg per 2 mL (40 mg per mL) 2 mL fill, in a 2 mL vial. | NDC 63323-306-01 2 mL Multiple Dose Vial |
300630 | NDC 63323-306-30 Unit of 10 | 1,200 mg per 30 mL (40 mg per mL) 30 mL fill, in a 30 mL vial. | NDC 63323-306-05 30 mL Multiple Dose Vial |

Lake Zurich, IL 60047 www.fresenius-kabi.com/us
45964H
Revised: October 2023
Mechanism of Action
Tobramycin acts by inhibiting synthesis of protein in bacterial cells. In vitro tests demonstrate that tobramycin is bactericidal.