Get your patient on Viberzi (Eluxadoline)
Viberzi prior authorization resources
Most recent state uniform prior authorization forms
Viberzi patient education
Patient toolkit
Dosage & administration
2 DOSAGE AND ADMINISTRATION
The recommended dosage of VIBERZI is 100 mg taken orally twice daily with food.
The recommended dosage of VIBERZI is 75 mg taken orally twice daily with food in patients:
- unable to tolerate the 100 mg dose of VIBERZI [see Adverse Reactions (6.1 ) ].
- receiving concomitant OATP1B1 inhibitors [see Drug Interactions (7 )] .
- with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment [see Use in Specific Population s (8.6 ) ] .
- with moderate or severe renal impairment (eGFR less than 60 mL/min/1.73 m 2 ); and in patients with end stage renal disease (ESRD) not yet on dialysis (eGFR less than 15 mL/min/1.73 m 2 ) [see Use in Specific Populations (8.7 )].
Discontinue VIBERZI in patients who develop severe constipation [see Warnings and Precautions (5.4 )] .
Instruct patients if they miss a dose, take the next dose at the regular time and not to take 2 doses at the same time to make up for a missed dose.

Viberzi prescribing information
1 INDICATIONS AND USAGE
VIBERZI is indicated in adults for the treatment of irritable bowel syndrome with diarrhea (IBS-D).
2 DOSAGE AND ADMINISTRATION
The recommended dosage of VIBERZI is 100 mg taken orally twice daily with food.
The recommended dosage of VIBERZI is 75 mg taken orally twice daily with food in patients:
- unable to tolerate the 100 mg dose of VIBERZI [see Adverse Reactions (6.1 ) ].
- receiving concomitant OATP1B1 inhibitors [see Drug Interactions (7 )] .
- with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment [see Use in Specific Population s (8.6 ) ] .
- with moderate or severe renal impairment (eGFR less than 60 mL/min/1.73 m 2 ); and in patients with end stage renal disease (ESRD) not yet on dialysis (eGFR less than 15 mL/min/1.73 m 2 ) [see Use in Specific Populations (8.7 )].
Discontinue VIBERZI in patients who develop severe constipation [see Warnings and Precautions (5.4 )] .
Instruct patients if they miss a dose, take the next dose at the regular time and not to take 2 doses at the same time to make up for a missed dose.
3 DOSAGE FORMS AND STRENGTHS
- 75 mg tablets: capsule-shaped tablets are coated in pale-yellow to light tan color debossed with “FX75” on one side. Each tablet contains 75 mg eluxadoline.
- 100 mg tablets: capsule-shaped tablets are coated in pink-orange to peach color debossed with “FX100” on one side. Each tablet contains 100 mg eluxadoline.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no studies with VIBERZI in pregnant women that inform any drug-associated risks. The background risk of major birth defects and miscarriage for the indicated population is unknown. However, the background risk in the U.S. general population of major birth defects is 2 to 4% and of miscarriage is 15 to 20% of clinically recognized pregnancies. In animal reproduction studies, oral and subcutaneous administration of eluxadoline to rats and rabbits during organogenesis at doses approximately 51 and 115 times the human exposure after a single oral dose of 100 mg, respectively, demonstrated no teratogenic effects. In a pre- and postnatal development study in rats, no adverse effects were observed in offspring with oral administration of eluxadoline at doses approximately 10 times the human exposure ( see Data ) .
Data
Animal Data
Eluxadoline administered as combined oral (1000 mg/kg/day) and subcutaneous (5 mg/kg/day) doses during the period of organogenesis to rats and rabbits (exposures about 51 and 115 times, respectively, the human AUC of 24 ng.h/mL after a single oral dose of 100 mg) did not cause any adverse effects on embryofetal development. A pre- and postnatal development study in rats showed no evidence of any adverse effect on pre- and postnatal development at oral doses of eluxadoline up to 1000 mg/kg/day (with exposures about 10 times the human AUC of 24 ng.h/mL after a single oral dose of 100 mg). In the same study, eluxadoline was detected in the milk of lactating rats administered oral doses of 100, 300 and 1000 mg/kg/day (with exposures about 1.8, 3 and 10 times, respectively, the human AUC of 24 ng.h/mL after a single oral dose of 100 mg). Milk samples were collected from six lactating females per group on lactation day 12. Mean concentrations of eluxadoline in the milk of lactating rats on lactation day 12 were 2.78, 5.49 and 44.02 ng/mL at 100, 300 and 1000 mg/kg/day, respectively.
8.2 Lactation
Risk Summary
No data are available regarding the presence of eluxadoline in human milk, the effects of eluxadoline on the breastfed infant, or the effects of eluxadoline on milk production. However, eluxadoline is present in rat milk [see Use in Specific Populations (8.1 ) ] .
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VIBERZI and any potential adverse effects on the breastfed infant from VIBERZI or from the underlying maternal condition.
8.000000000000000e+00 4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Juvenile Toxicology Data
Eluxadoline was orally administered to juvenile rats at 500, 750, and 1500 mg/kg/day (about 16, 54 and 30 times, respectively, the human AUC of 24 ng.h/mL after a single oral dose of 100 mg) for 4 weeks. There were no adverse physiologic effects related to eluxadoline. Based on these results, the NOAEL for male and female juvenile rats was 1500 mg/kg/day (about 30 times the human AUC of 24 ng.h/mL after a single oral dose of 100 mg).
8.000000000000000e+00 5 Geriatric Use
Of 1795 IBS-D patients in clinical trials of VIBERZI who received 75 mg or 100 mg twice daily, 139 (7.7%) were at least 65 years of age, while 15 (0.8%) were at least 75 years old. No overall differences in effectiveness were observed between these patients and younger patients. There were no overall differences in the types of adverse reactions observed between elderly and younger patients; however, a higher proportion of elderly patients than younger patients experienced adverse reactions (66% vs 59%), serious adverse reactions (9% vs 4%), and gastrointestinal adverse reactions (39% vs 28%).
8.000000000000000e+00 6 Hepatic Impairment
Plasma concentrations of eluxadoline increase in patients with hepatic impairment [see Clinical Pharmacology (12.3 )] .
VIBERZI is contraindicated in patients with severe hepatic impairment (Child-Pugh Class C) as plasma concentrations of eluxadoline increase significantly (16-fold) and there is no information to support the safety of VIBERZI in these patients.
In patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment, plasma concentrations of eluxadoline increase to a lesser extent (6- and 4-fold, respectively). Administer VIBERZI at a reduced dose of 75 mg twice daily to these patients [see Dosage and Administration (2 )] . Monitor patients with any degree of hepatic impairment for impaired mental or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery and for other eluxadoline-related adverse reactions [see Adverse Reactions (6.1 )] .
8.7 Renal Impairment
In patients with severe renal impairment (eGFR 15 to 30 mL/min/1.73 m 2 ) and ESRD not yet on dialysis (eGFR less than 15 mL/min/1.73 m 2 ), exposure of eluxadoline was increased compared to healthy subjects with normal renal function [see Clinical Pharmacology (12.3 )]. Administer VIBERZI at a reduced dose of 75 mg twice daily to patients with moderate or severe renal impairment and in patients with ESRD not yet on dialysis [see Dosage and Administration (2 )]. No dosage adjustment is needed for patients with mild renal impairment.
4 CONTRAINDICATIONS
VIBERZI is contraindicated in patients:
- Without a gallbladder. These patients are at increased risk of developing serious adverse reactions of pancreatitis and/or sphincter of Oddi spasm [see Warnings and Precautions (5.1 , 5.2 )]
- With known or suspected biliary duct obstruction; or sphincter of Oddi disease or dysfunction. These patients are at increased risk for sphincter of Oddi spasm [see Warnings and Precautions (5.1 )].
- With alcoholism, alcohol abuse or alcohol addiction, or in patients who drink more than 3 alcoholic beverages per day. These patients are at increased risk for acute pancreatitis [see Warnings and Precautions (5.1 )].
- With a history of pancreatitis; or structural diseases of the pancreas, including known or suspected pancreatic duct obstruction. These patients are at increased risk for acute pancreatitis [see Warnings and Precautions (5.1 )] .
- With a known hypersensitivity reaction to VIBERZI [see Warnings and Precautions (5.3 )].
- With severe hepatic impairment (Child-Pugh Class C). These patients are at risk for significantly increased plasma concentrations of eluxadoline [see Use in Specific Populations (8.6 )]
- With a history of chronic or severe constipation or sequelae from constipation, or known or suspected mechanical gastrointestinal obstruction. These patients may be at risk for severe complications of bowel obstruction [see Warnings and Precautions (5.4 )] .
5 WARNINGS AND PRECAUTIONS
- Pancreatitis and Sphincter of Oddi Spasm : Monitor patients for new or worsening abdominal pain, with or without nausea and vomiting, or acute biliary pain with liver or pancreatic enzyme elevations; immediately discontinue VIBERZI and seek medical attention if symptoms develop. (5.1 , 5.2 )
- Hypersensitivity Reactions, including anaphylaxis : Immediately discontinue VIBERZI and seek medical attention if symptoms develop. (4 , 5.3 )
- Constipation: Instruct patients to stop VIBERZI and immediately contact their healthcare provider if they develop severe constipation. Avoid use with other drugs that may cause constipation (5.4 , 7 )
5.1 Pancreatitis
Pancreatitis, with or without sphincter of Oddi spasm [ see Warnings and Precautions (5.1 ) ] , has been reported in patients taking either the 75 mg or 100 mg dosage of VIBERZI, including serious cases resulting in hospitalization, primarily in patients without a gallbladder. Fatal cases have also been reported in patients without a gallbladder. VIBERZI is contraindicated in patients without a gallbladder [ see Contraindications (4 ) ] . Most of the reported cases of serious pancreatitis occurred within a week of starting treatment with VIBERZI and some developed symptoms after one to two doses.
In patients with a gallbladder, evaluate a patient’s alcohol intake prior to starting VIBERZI. Instruct patients to avoid chronic or acute excessive alcohol use while taking VIBERZI. Monitor for new or worsening abdominal pain that may radiate to the back or shoulder, with or without nausea and vomiting. Instruct patients to immediately stop VIBERZI and seek medical attention if they experience symptoms suggestive of pancreatitis such as acute abdominal or epigastric pain radiating to the back or shoulder associated with elevations of pancreatic enzymes with or without nausea and vomiting [ see Contraindications (4 ) ] .
5.2 Sphincter of Oddi Spasm
There is a risk of sphincter of Oddi spasm, resulting in pancreatitis or hepatic enzyme elevation associated with acute abdominal pain (e.g., biliary-type pain) in patients taking VIBERZI [ see Adverse Reactions (6.1 ) ] . Postmarketing serious adverse reactions of sphincter of Oddi spasm with or without pancreatitis resulting in hospitalization have been reported, primarily in patients without a gallbladder [ see Warnings and Precautions (5.1 ) ] . Most of the reported cases of serious sphincter of Oddi spasm occurred within a week of starting treatment with VIBERZI and some developed symptoms after one to two doses. VIBERZI is contraindicated in patients without a gallbladder [ see Contraindications (4 ) ] .
Instruct patients to immediately stop VIBERZI and seek medical attention if they experience symptoms suggestive of sphincter of Oddi spasm such as acute worsening of abdominal pain, (e.g. acute epigastric or biliary [i.e., right upper quadrant] pain), that may radiate to the back or shoulder with or without nausea and vomiting, associated with elevations of pancreatic enzymes or liver transaminases. Do not restart VIBERZI in patients who developed biliary duct obstruction or sphincter of Oddi spasm while taking VIBERZI [ see Contraindications (4 ) ] .
5.3 Hypersensitivity Reactions
In postmarketing experience, serious hypersensitivity reactions (including anaphylaxis) have been reported following VIBERZI administration. Some of these reactions occurred after the first one or two doses of VIBERZI [ see Adverse Reactions (6.2 )] .
Instruct patients to immediately stop VIBERZI and seek medical attention if they experience symptoms suggestive of a hypersensitivity reaction [ see Contraindications (4 ) ] .
5.4 Constipation
Constipation, sometimes requiring hospitalization, has been reported following VIBERZI administration. In postmarketing experience, severe cases with development of intestinal obstruction, intestinal perforation, and fecal impaction, requiring intervention, have also been reported. Instruct patients to stop VIBERZI and immediately contact their healthcare provider if they experience severe constipation. Avoid use with other drugs that may cause constipation [ see Adverse Reactions (6.1 ), Drug Interactions (7 ) ] .
6 ADVERSE REACTIONS
The following adverse reactions described below and elsewhere in the labeling include:
6.1 Clinical Trial s Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Over 1700 patients with IBS-D have been treated with 75 or 100 mg of VIBERZI twice daily in controlled trials. Exposures from placebo-controlled clinical trials in adult patients with IBS-D included 1391 exposed for 3 months, 1001 exposed for 6 months and 488 exposed for one year.
Demographic characteristics were comparable between the treatment groups [see Clinical Studies (14 )] . Data described below represent pooled data compared to placebo across the randomized trials.
Pancreatitis
Cases of pancreatitis, not associated with sphincter of Oddi spasm, were reported in 2/807 (0.2%) of patients receiving 75 mg and 3/1032 (0.3%) of patients receiving 100 mg VIBERZI twice daily in clinical trials. Of these 5 cases, 3 were associated with excessive alcohol intake, one was associated with biliary sludge, and in one case the patient discontinued VIBERZI 2 weeks prior to the onset of symptoms. All pancreatic events resolved with lipase normalization upon discontinuation of VIBERZI, with 80% (4/5) resolving within 1 week of treatment discontinuation. The case of sphincter of Oddi spasm-induced pancreatitis resolved within 24 hours of discontinuation.
Sp h incter of Oddi Spasm
In clinical trials, sphincter of Oddi spasm occurred in 0.2% (2/807) of patients receiving 75 mg and 0.8% (8/1032) of patients receiving 100 mg VIBERZI twice daily.
- Among patients receiving 75 mg, 1/807 (0.1%) patient experienced a sphincter of Oddi spasm presenting with abdominal pain but with lipase elevation less than 3 times the upper limit of normal (ULN) and 1/ 807 (0.1%) patient experienced a sphincter of Oddi spasm manifested as elevated hepatic enzymes associated with abdominal pain
- Among patients receiving 100 mg, 1/1032 (0.1%) patient experienced a sphincter of Oddi spasm manifested as pancreatitis and 7/1032 (0.7%) patients experienced sphincter of Oddi spasm manifested as elevated hepatic enzymes associated with abdominal pain
Of those patients who experienced a sphincter of Oddi spasm, 80% (8/10) reported their first onset of symptoms within the first week of treatment. The case of sphincter of Oddi spasm-induced pancreatitis occurred within minutes of taking the first dose of VIBERZI. No cases of sphincter of Oddi spasm occurred greater than 1 month after treatment onset. All events resolved upon discontinuation of VIBERZI, with symptoms typically improved by the following day.
Common Adverse Reactions
Table 1 provides the incidence of common adverse reactions reported in > 2% of IBS-D patients in either VIBERZI treatment group and at an incidence greater than in the placebo group.
| Adverse Reactions | VIBERZI 100 mg twice daily (N= 1032) % | VIBERZI 75 mg twice daily (N=807) % | Placebo (N=975) % |
| Constipation | 8 | 7 | 2 |
| Nausea | 7 | 8 | 5 |
| Abdominal Pain•• | 7 | 6 | 4 |
| Upper Respiratory Tract Infection | 5 | 3 | 4 |
| Vomiting | 4 | 4 | 1 |
| Nasopharyngitis | 3 | 4 | 3 |
| Abdominal Distention | 3 | 3 | 2 |
| Bronchitis | 3 | 3 | 2 |
| Dizziness | 3 | 3 | 2 |
| Flatulence | 3 | 3 | 2 |
| Rash••• | 3 | 3 | 2 |
| Increased ALT | 3 | 2 | 1 |
| Fatigue | 2 | 3 | 2 |
| Viral gastroenteritis | 1 | 3 | 2 |
• Reported in > 2% of VIBERZI-treated patients at either dose and at an incidence greater than in placebo-treated patients
•• " Abdominal Pain" term includes: abdominal pain, abdominal pain lower, and abdominal pain upper
••• " Rash" term includes: dermatitis, dermatitis allergic, rash, rash erythematous, rash generalized, rash maculo-papular, rash papular, rash pruritic, urticaria, and idiopathic urticaria
Constipation was the most commonly reported adverse reaction in VIBERZI-treated patients in these trials. Approximately 50% of constipation events occurred within the first 2 weeks of treatment while the majority occurred within the first 3 months of therapy. Rates of severe constipation were less than 1% in patients receiving 75 mg and 100 mg VIBERZI. Similar rates of constipation occurred between the active and placebo arms beyond 3 months of treatment.
Adverse Reactions Leading to Discontinuation
Eight percent of patients treated with 75 mg, 8% of patients treated with 100 mg VIBERZI and 4% of patients treated with placebo discontinued prematurely due to adverse reactions. In the VIBERZI treatment groups, the most common reasons for discontinuation due to adverse reactions were constipation (1% for 75 mg and 2% for 100 mg) and abdominal pain (1% for both 75 mg and 100 mg). In comparison, less than 1% of patients in the placebo group withdrew due to constipation or abdominal pain.
Less Common Adverse Reactions
Adverse reactions that were reported in ≤ 2% of VIBERZI-treated patients are listed below by body system.
Gastrointestinal : gastroesophageal reflux disease General Disorders and administration site conditions : feeling drunk Investigations: increased AST Nervous system : sedation, somnolence Psychiatric disorders : euphoric mood Respiratory : asthma, bronchospasm, respiratory failure, wheezing
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of VIBERZI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity : anaphylaxis, angioedema (e.g. swollen face and throat), dyspnea, throat tightness, and chest pain/tightness [see Warnings and Precautions (5.3 )] .
7 DRUG INTERACTIONS
Tables 2 and 3 include drugs which demonstrated a clinically important drug interaction with VIBERZI or which potentially may result in clinically relevant interactions.
| OATP1B1 Inhibitors | |
| Clinical Impact: | Increased exposure to eluxadoline when coadministered with cyclosporine [ see Clinical Pharmacology (12.3 ) ] |
| Intervention: | Administer VIBERZI at a dose of 75 mg twice daily [see Dosage and Administration (2 )] and monitor patients for impaired mental or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery and for other eluxadoline-related adverse reactions [see Adverse Reactions (6.1 )] . |
| Examples: | cyclosporine, gemfibrozil, antiretrovirals (atazanavir, lopinavir, ritonavir, saquinavir, tipranavir), rifampin, eltrombopag |
| Drugs that Cause Constipation | |
| Clinical Impact: | Increased risk for constipation related adverse reactions and potential for constipation related serious adverse reactions |
| Intervention: | Avoid use with other drugs that may cause constipation (see below); loperamide may be used occasionally for acute management of severe diarrhea but avoid chronic use. Discontinue loperamide immediately if constipation occurs. |
| Examples: | alosetron, anticholinergics, opioids |
| OATP1B1 and BCRP Substrate | |
| Clinical Impact: | VIBERZI may increase the exposure of co-administered OATP1B1 and BCRP substrates. Increased exposure to rosuvastatin when co-administered with VIBERZI with a potential for increased risk of myopathy/rhabdomyolysis [ see Clinical Pharmacology (12.3 ) ] |
| Intervention: | Use the lowest effective dose of rosuvastatin (see prescribing information of rosuvastatin for additional information on recommended dosing). |
11 DESCRIPTION
The active ingredient in VIBERZI is eluxadoline, a mu-opioid receptor agonist.
The full chemical name is 5-[[[(2S)-2-amino-3-[4-(aminocarbonyl)-2,6-dimethylphenyl]-1-oxopropyl][(1S)-1-(4-phenyl-1H-imidazol-2-yl)ethyl]amino]methyl]-2-methoxybenzoic acid.
Eluxadoline has a molecular weight of 569.65 and a molecular formula of C 32 H 35 N 5 O 5 . The chemical structure of eluxadoline is:
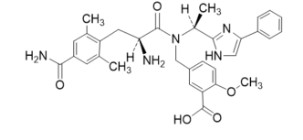
VIBERZI is available as 75 mg and 100 mg tablets for oral administration. In addition to the active ingredient, eluxadoline, each tablet contains the following inactive ingredients: silicified microcrystalline cellulose, colloidal silica, crospovidone, mannitol, magnesium stearate, and Opadry II (partially hydrolyzed polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, iron oxide yellow, and iron oxide red).
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Eluxadoline is a mu-opioid receptor agonist; eluxadoline is also a delta opioid receptor antagonist and a kappa opioid receptor agonist. The binding affinities (Ki) of eluxadoline for the human mu and delta opioid receptors are 1.8 nM and 430 nM, respectively. The binding affinity (Ki) of eluxadoline for the human kappa opioid receptor has not been determined; however, the Ki for guinea pig cerebellum kappa opioid receptor is 55 nM. In animals, eluxadoline interacts with opioid receptors in the gut.
12.2 Pharmacodynamics
Cardiac Electrophysiology
At a dose 10 times the maximum recommended dose (100 mg), VIBERZI does not prolong the QT interval to any clinically relevant extent.
12.3 Pharmacokinetics
Following oral administration of 100 mg VIBERZI in healthy subjects, the C max of eluxadoline was approximately 2 to 4 ng/mL and AUC was 12 to 22 ng.h/mL. Eluxadoline has approximately linear pharmacokinetics with no accumulation upon repeated twice daily dosing. The variability of eluxadoline pharmacokinetic parameters ranges from 51% to 98%.
Absorption
Absolute bioavailability of eluxadoline has not been determined.
Effect of Food
The median T max value was 1.5 hours (range: 1 to 8 hours) under fed conditions and 2 hours (range: 0.5 to 6 hours) under fasting conditions [see Dosage and Administration (2 )] .
The administration of VIBERZI with a high fat meal that contained approximately 800 to 1000 total calories, with 50% of calories being derived from fat content decreased the C max of eluxadoline by 50% and AUC by 60%.
Distribution
Plasma protein binding of eluxadoline was 81%.
Elimination
The mean plasma elimination half-life of eluxadoline ranged from 3.7 hours to 6 hours.
Metabolism
Cytochrome P450 (CYP) and UGT pathways have minimal involvement in the metabolism of eluxadoline. It is unlikely that metabolism of eluxadoline by these enzymes has a clinically meaningful impact on systemic exposure.
Excretion
Following a single oral dose of 300 mg [ 14 C] eluxadoline in healthy male subjects, 82.2% of the total radioactivity was recovered in feces within 336 hours and less than 1% was recovered in urine within 192 hours.
Speci fic Populations
Patients with Hepatic Impairment
Following a single oral 100–mg dose in subjects with varying degrees of liver impairment and healthy subjects, mean eluxadoline plasma exposure was 6-fold, 4-fold, and 16-fold higher in mild, moderate, and severe hepatically impaired subjects (Child Pugh Class A, B, C), respectively, compared to the subjects with normal liver function [see Dosage and Administration (2 ) , Contraindications (4 ), Use in Specific Population s (8.6 ) ] .
P a ti e nts with R e n a l I mp a i r m e nt
Following a single oral 100 mg dose, the mean eluxadoline C max and AUC 0-t were 3.7-fold and 4.7-fold higher, respectively, in subjects with severe renal impairment (eGFR 15 to 30 mL/min/1.73 m 2 , n=2) and 2.4-fold and 5.9-fold higher, respectively, in subjects with ESRD not yet on dialysis (eGFR less than 15 mL/min/1.73 m 2 , n=6) compared to healthy subjects with normal renal function (n=6) [see Dosage and Administration (2 ), Use in Specific Populations (8.7 )].
Drug Interaction Studies
In Vitro Assessment of Drug Interactions
In vitro studies indicate that eluxadoline is neither an inducer of CYP1A2, CYP2C9, CYP2C19, and CYP3A4, nor an inhibitor of CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, and CYP2D6 at clinically relevant systemic concentrations. Although CYP2E1 was slightly inhibited by eluxadoline (IC 50 of approximately 20 micromolar [11 mcg/mL]), clinically meaningful interactions are unlikely. Despite mechanism-based inhibition of CYP3A4 in vitro , eluxadoline did not have a clinically meaningful drug-drug interaction with the CYP3A4 substrate midazolam.
I n vitro studies suggest that eluxadoline is a substrate for OAT3, OATP1B1, BSEP and MRP2, but not for OCT1, OCT2, OAT1, OATP1B3, P-gp and BCRP. Based on the in vitro studies, clinically meaningful interaction via inhibition of OCT1, OCT2, OAT1, OAT3, OATP1B3, BSEP and MRP2 by eluxadoline is unlikely.
In Vivo Assessment of Drug Interactions
The following drug interactions were studied in healthy subjects:
Oral Contraceptives
Coadministration of multiple doses of 100 mg VIBERZI with multiple dose administration of an oral contraceptive (norethindrone 0.5 mg/ethinyl estradiol 0.035 mg) does not change the exposure of either drug.
Cyclosporine
Coadministration of a single dose of 100 mg VIBERZI with a single dose of 600 mg cyclosporine resulted in 4.4-fold and 6.2-fold increase in AUC and C max of eluxadoline, respectively, compared to administration of VIBERZI alone [see Drug Interactions (7 )] .
Proben e cid
Coadministration of a single dose of 100 mg VIBERZI with a single dose of 500 mg probenecid resulted in a 35% and 31% increase in eluxadoline AUC and C max , respectively, compared to administration of VIBERZI alone. This change in eluxadoline exposures is not expected to be clinically meaningful.
Rosuvastatin
Coadministration of multiple doses of 100 mg VIBERZI twice daily with a single dose 20 mg rosuvastatin resulted in an increase in the AUC (40%) and C max (18%) of rosuvastatin compared to administration of rosuvastatin alone. Similar results were observed with the active, major metabolite, n-desmethyl rosuvastatin [see Drug Interaction s (7 ) ] .
Midazolam
Coadministration of multiple doses of 100 mg VIBERZI twice daily with single dose administration of 4 mg midazolam did not affect midazolam pharmacokinetics in humans, suggesting that eluxadoline will not affect the exposure of concomitantly administered CYP3A4 substrates.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Two-year oral carcinogenicity studies have been conducted with eluxadoline in CD-1 mice at doses up to 1500 mg/kg/day (about 14 times the human AUC of 24 ng.h/mL after a single oral dose of 100 mg) and in Sprague Dawley rats at oral doses up to 1500 mg/kg/day (about 36 times the human AUC of 24 ng.h/mL after a single oral dose of 100 mg). Oral administration of eluxadoline for 104 weeks did not produce tumors in mice and rats.
Mutagenesis
Eluxadoline was negative in the Ames test, chromosome aberration test in human lymphocytes, in the mouse lymphoma cell (L5178Y/TK +/- ) forward mutation test and in the in vivo rat bone marrow micronucleus test.
Impairment of Fertility
Eluxadoline at oral doses up to 1000 mg/kg/day (about 10 times the human AUC of 24 ng.h/mL after a single oral dose of 100 mg) was found to have no adverse effect on fertility and reproductive performance of male and female rats.
14 CLINICAL STUDIES
The efficacy and safety of VIBERZI in IBS-D patients was established in two randomized, multi-center, multi-national, double-blind, placebo-controlled trials (Studies 1 and 2). A total of 1281 patients in Study 1 and 1145 patients in Study 2 received treatment with VIBERZI 75 mg, VIBERZI 100 mg or placebo twice daily [overall, patients had a mean age of 45 years (range 18 to 80 years with 10% at least 65 years of age or older), 66% female, 86% white, 11% black, and 27% Hispanic].
All patients met Rome III criteria for IBS-D (loose [mushy] or watery stools ≥25% and hard or lumpy stools <25% of bowel movements) and were required to meet both of the following criteria:
- an average of worst abdominal pain scores in the past 24 hours of >3.0 on a 0 to 10 scale over the week prior to randomization.
- an average daily stool consistency score (Bristol Stool Scale or BSS) of ≥5.5 and at least 5 days with a BSS score ≥5 on a 1 to 7 scale over the week prior to randomization.
Pertinent exclusion criteria included: prior pancreatitis, alcohol abuse, cholecystitis prior 6 months, sphincter of Oddi dysfunction, inflammatory bowel disease, intestinal obstruction, gastrointestinal infection or diverticulitis within prior 3 months, lipase greater than 2 xULN, ALT or AST greater than 3 xULN.
Study 1 and Study 2 included identical 26-week double-blind, placebo-controlled treatment periods. Study 1 continued double-blinded for an additional 26 weeks for long-term safety (total of 52 weeks of treatment), followed by a 2-week follow-up. Study 2 included a 4-week single-blinded, placebo-withdrawal period upon completion of the 26-week treatment period. During the double-blind treatment phase and the single-blinded placebo withdrawal phase, patients were allowed to take loperamide rescue medication for the acute treatment of uncontrolled diarrhea, but were not allowed to take any other antidiarrheal, antispasmodic agent or rifaximin for their diarrhea. Additionally, patients were allowed to take aspirin-containing medications or nonsteroidal anti-inflammatory drugs, but no narcotic or opioid containing agents.
Efficacy of VIBERZI was assessed in both trials using an overall composite responder primary endpoint. The primary endpoint was defined by the simultaneous improvement in the daily worst abdominal pain score by ≥30% as compared to the baseline weekly average AND a reduction in the BSS to <5 on at least 50% of the days within a 12-week time interval. Improvement in daily worst abdominal pain in the absence of a concurrent bowel movement was also considered a response day. Results for endpoints were based on electronic daily diary entries by patients.
The proportion of composite responders over 12 weeks is shown in Table 4 . In both trials, the proportion of patients who were composite responders to VIBERZI was statistically significantly higher than placebo for both doses. The proportion of patients who were composite responders to VIBERZI was similar for male and female patients in both trials.
| Study 1 | Study 2 | ||||||
| VIBERZI 100mg twice daily n=426 | VIBERZI 75mg twice daily n=427 | PBO n=427 | VIBERZI 100mg twice daily n=382 | VIBERZI 75mg twice daily n=381 | PBO n=382 | ||
| Composite 1 Response o ver 12 weeks | |||||||
| Responder rates | 25% | 24% | 17% | 30% | 29% | 16% | |
| Treatment difference | 8% 2 | 7% 4 | 13% 3 | 13% 3 | |||
| 95% CI (%) | (2.6, 13.5) | (1.4, 12.2) | (7.5, 19.2) | (6.8, 18.5) | |||
| Composite Response over 26 weeks | |||||||
| Responder rates | 29% | 23% | 19% | 33% | 30% | 20% | |
| Treatment difference | 10% | 4% | 13% | 10% | |||
| 95% CI (%) | (4.7, 16.1) | (-1.0, 9.9) | (6.4, 18.8) | (4.2, 16.4) | |||
| Abdominal Pain Response Improved ≥30% over 12 weeks | |||||||
| Responder rates | 43% | 42% | 40% | 51% | 48% | 45% | |
| Treatment difference | 4% | 3% | 6% | 3% | |||
| 95% CI (%) | (-3.0, 10.2) | (-3.8, 9.4) | (-1.3, 12.8) | (-4.3, 9.8) | |||
| BSS <5 Response over 12 weeks | |||||||
| Responder rates | 34% | 30% | 22% | 36% | 37% | 21% | |
| Treatment difference | 12% | 8% | 15% | 16% | |||
| 95% CI (%) | (6.3, 18.2) | (2.1, 13.8) | (8.4, 21.0) | (9.7, 22.4) | |||
1 Composite= Simultaneous improvement of Worst Abdominal Pain (WAP) by ≥30% and Bristol Stool Score (BSS) < 5 on the same day for ≥ 50% of days over the interval
2 P <0.01
3 P <0.001
4 P <0.05
Additionally, the proportion of patients who were composite responders to VIBERZI at each 4-week interval was numerically higher than placebo for both doses as early as month 1 through month 6 demonstrating that efficacy is maintained throughout the course of treatment.
During the 4 week single-blind withdrawal period in Study 2, no evidence of worsening of diarrhea or abdominal pain compared to baseline was demonstrated at either dose.
16 HOW SUPPLIED/STORAGE AND HANDLING
VIBERZI is available as:
- 75 mg tablets: capsule-shaped tablets, coated in pale-yellow to light tan color, debossed with “FX75” on one side. Bottle of 60: NDC 61874-075-60
- 100 mg tablets: capsule-shaped tablets, coated in pink-orange to peach color, debossed with “FX100” on one side. Bottle of 60: NDC 61874-100-60
Store VIBERZI tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature] .
12.1 Mechanism of Action
Eluxadoline is a mu-opioid receptor agonist; eluxadoline is also a delta opioid receptor antagonist and a kappa opioid receptor agonist. The binding affinities (Ki) of eluxadoline for the human mu and delta opioid receptors are 1.8 nM and 430 nM, respectively. The binding affinity (Ki) of eluxadoline for the human kappa opioid receptor has not been determined; however, the Ki for guinea pig cerebellum kappa opioid receptor is 55 nM. In animals, eluxadoline interacts with opioid receptors in the gut.