Get your patient on Vitamin D - Ergocalciferol capsule (Ergocalciferol)
Vitamin D - Ergocalciferol capsule prescribing information
INDICATIONS AND USAGE
Ergocalciferol Capsules, USP are indicated for use in the treatment of hypoparathyroidism, refractory rickets, also known as vitamin D resistant rickets, and familial hypophosphatemia.
DOSAGE AND ADMINISTRATION
THE RANGE BETWEEN THERAPEUTIC AND TOXIC DOSES IS NARROW.
Vitamin D Resistant Rickets: 12,000 to 500,000 USP Units daily.
Hypoparathyroidism: 50,000 to 200,000 USP Units daily concomitantly with calcium lactate 4 g, six times per day.
DOSAGE MUST BE INDIVIDUALIZED UNDER CLOSE MEDICAL SUPERVISION.
Calcium intake should be adequate. Blood calcium and phosphorus determinations must be made every 2 weeks or more frequently if necessary.
X-rays of the bones should be taken every month until condition is corrected and stabilized.
CONTRAINDICATIONS
Ergocalciferol Capsules, USP are contraindicated in patients with hypercalcemia, malabsorption syndrome, abnormal sensitivity to the toxic effects of vitamin D, and hypervitaminosis D.
ADVERSE REACTIONS
Hypervitaminosis D is characterized by effects on the following organ system:
Renal: Impairment of renal function with polyuria, nocturia, polydipsia, hypercalciuria, reversible azotemia, hypertension, nephrocalcinosis, generalized vascular calcification, or irreversible renal insufficiency which may result in death.
CNS: Mental retardation.
Soft Tissues: Widespread calcification of the soft tissues, including the heart, blood vessels, renal tubules, and lungs.
Skeletal: Bone demineralization (osteoporosis) in adults occurs concomitantly.
Decline in the average rate of linear growth and increased mineralization of bones in infants and children (dwarfism) vague aches, stiffness, and weakness.
Gastrointestinal: Nausea, anorexia, constipation.
Metabolic: Mild acidosis, anemia, weight loss.
Call your doctor for medical advice about side effects. You may report side effects to Avet Pharmaceuticals Inc. at 1-866-901-DRUG (3784) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Mineral oil interferes with the absorption of fat-soluble vitamins, including vitamin D preparations.
Administration of thiazide diuretics to hypoparathyroid patients who are concurrently being treated with Ergocalciferol Capsules, USP may cause hypercalcemia.
DESCRIPTION
Ergocalciferol Capsules, USP are a synthetic calcium regulator for oral administration.
Ergocalciferol is a white, colorless crystal, insoluble in water, soluble in organic solvents, and slightly soluble in vegetable oils. It is affected by air and by light. Ergosterol or provitamin D 2 is found in plants and yeast and has no antirachitic activity.
There are more than 10 substances belonging to a group of steroid compounds, classified as having vitamin D or antirachitic activity.
One USP unit of vitamin D 2 is equivalent to one International Unit (IU), and 1 mcg of vitamin D 2 is equal to 40 USP Units.
Each capsule contains Vitamin D (Ergocalciferol) 1.25 mg equivalent to 50,000 USP Units in an edible vegetable oil.
Ergocalciferol, also called vitamin D 2 , is 9,10-secoergosta-5,7,10(19), 22-tetraen-3-ol, (3β,5Z,7E,22 E )-; (C 28 H 44 O) with a molecular weight of 396.65, and has the following structural formula:
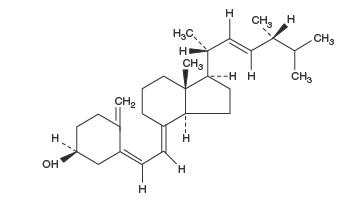
Inactive Ingredients : FD&C Blue #1, FD&C Yellow #5, Gelatin, Glycerin, Lecithin, Medium chain triglyceride, Purified water, Shellac glaze, N-Butyl alcohol, Simethicone, Soybean oil and Titanium dioxide.
CLINICAL PHARMACOLOGY
The in vivo synthesis of the major biologically active metabolites of vitamin D occurs in two steps. The first hydroxylation of ergocalciferol takes place in the liver (to 25-hydroxyvitamin D) and the second in the kidneys (to 1, 25-dihydroxyvitamin D). Vitamin D metabolites promote the active absorption of calcium and phosphorus by the small intestine, thus elevating serum calcium and phosphate levels sufficiently to permit bone mineralization. Vitamin D metabolites also mobilize calcium and phosphate from bone and probably increase the reabsorption of calcium and perhaps also of phosphate by the renal tubules.
There is a time lag of 10 to 24 hours between the administration of vitamin D and the initiation of its action in the body due to the necessity of synthesis of the active metabolites in the liver and kidneys. Parathyroid hormone is responsible for the regulation of this metabolism in the kidneys.
HOW SUPPLIED
Each capsule contains Vitamin D (Ergocalciferol) 1.25 mg equivalent to 50,000 USP Units. The green colored oval shaped transparent soft gelatin capsules are imprinted with '194' in white and contain clear light yellow oily liquid.
HDPE Bottles of 100 capsules (NDC 23155-809-01)
Store at 20 to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Protect from light.
Manufactured by:
Strides Pharma Science Limited
Bengaluru - 562106, India.
Manufactured for:
Avet Pharmaceuticals Inc.
East Brunswick, NJ 08816
1.866.901.DRUG(3784)
Rx Only
Revised: 11/2021