Get your patient on Ycanth - Cantharidin solution (Cantharidin)
Ycanth - Cantharidin solution prescribing information
INDICATIONS AND USAGE
YCANTH is indicated for the topical treatment of molluscum contagiosum in adult and pediatric patients 2 years of age and older.
DOSAGE AND ADMINISTRATION
- All healthcare professionals should receive instruction and training prior to preparation and administration of YCANTH. (2.1 )
- For topical use only. Not for oral, mucosal, or ophthalmic use. (2.1 )
- Apply a single application directly to each lesion every 3 weeks as needed. (2.2 )
- Do not use more than two applicators during a single treatment session. (2.2 )
- Remove with soap and water 24 hours after treatment. (2.2 )
- For additional instructions on preparation and administration of YCANTH, see Full Prescribing Information. (2.1 , 2.2 , 2.3 )
Important Administration Instructions
- All healthcare professionals should receive instruction and training prior to preparation and administration of YCANTH.
- YCANTH is for topical use only.
- YCANTH is not for oral, mucosal, or ophthalmic use. Avoid contact with the treatment area, including oral contact, after YCANTH treatment. Do not apply YCANTH near the eyes [see Warnings and Precautions (5.1) , Overdosage (10) ] .
- If YCANTH contacts any unintended surface, including healthy skin, immediately remove by wiping with a cotton swab or gauze. Avoid other topical products on treated areas until 24 hours after YCANTH treatment or until washing [see Warnings and Precautions (5.2) ].
- Avoid fire, flame or smoking near lesion(s) during treatment [see Warnings and Precautions (5.3) ] .
Dosage and Administration Overview
- Regarding YCANTH dosage and administration:
- Use nitrile or vinyl gloves and eye protection during preparation and administration.
- Apply topically as a single application to cover each lesion. Use no more than two YCANTH applicators during a single treatment session.
- Remove with soap and water 24 hours after treatment. Administer YCANTH every 3 weeks as needed.
- Do not cover any treated lesions with bandages.
- If severe blistering, severe pain, or other severe adverse reactions occur, remove YCANTH with soap and water prior to 24 hours after treatment [see Warnings and Precautions (5.2) ].
- Regarding use of the YCANTH applicator and break tool:
- Do not reuse the applicator. The applicator is for a single treatment session only.
- Do not attempt to use a clogged applicator.
- Do not cut or modify the applicator in any way; doing so could reduce dispensing control.
- Do not remove the applicator cap prior to breaking the glass ampule.
- If any damage or leaks are observed on the applicator, applicators should be discarded in a sharps container and handled in accordance with accepted medical practice and applicable law. The YCANTH Break Tool should be managed as solid waste and placed in plastic recycling containers or the general trash.
Dosage and Administration Instructions
Instructions for preparing the YCANTH Applicator and administering the YCANTH Solution are presented below.
YCANTH includes the following components [see How Supplied/Storage and Handling (16.1) ] :
Outer Protective Tube  | ||
The YCANTH Applicator 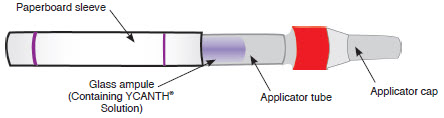 | ||
The YCANTH Break Tool  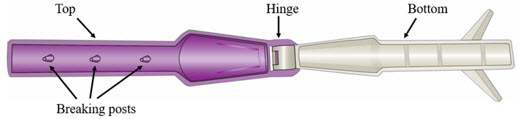 | ||
Step 1. Put on Personal Protective Equipment (PPE)
|  | |
Step 2. Remove the YCANTH Applicator from Outer Protective Tube
| 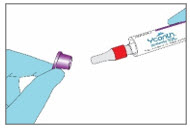 | |
Step 3. Remove the Paperboard Sleeve
| 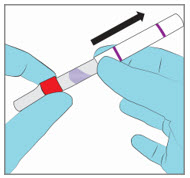 | |
Step 4. Inspect for Damage
| ||
Broken glass ampule  | Cut or cracked applicator tube  | No damage (for reference)  |
| ||
Step 5. Break Ampule using the YCANTH Break Tool
| 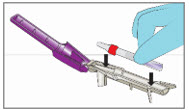 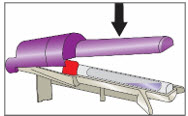 | |
Step 6. Tap YCANTH Applicator to move YCANTH Solution
| 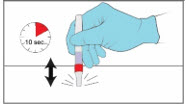 | |
Step 7. Test the YCANTH Applicator
|  | |
Step 8. Apply the YCANTH Solution
| 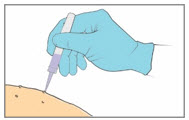 | |
Step 9. Allow YCANTH Solution to Dry
| 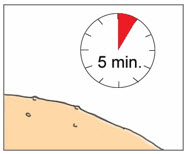 | |
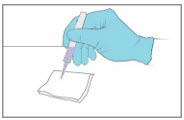
Step 10. Dispose of the YCANTH Applicator
| 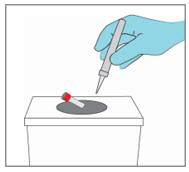 | |
YCANTH Break Tool Maintenance
- Wipe the surfaces of the YCANTH Break Tool with 70% isopropyl alcohol following each use.
- Inspect the YCANTH Break Tool for damage (e.g., cracks in plastic, missing or broken breaking posts) and function (e.g., hinge) prior to each use.
- After 12 uses or if any damage is observed, the YCANTH Break Tool should be managed as solid waste and placed in plastic recycling containers or the general trash.
- If needed, contact Verrica Pharmaceuticals Inc. at 1-973-298-1390 to request additional YCANTH Break Tools.
DOSAGE FORMS AND STRENGTHS
Topical solution 0.7%: Each mL of YCANTH contains 7 mg (0.7%) of cantharidin in a light violet to dark purple, slightly viscous liquid.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no available data with use of YCANTH in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Animal reproduction studies were not conducted with cantharidin. Given that systemic exposure to cantharidin following topical administration is low, maternal use is not expected to result in fetal exposure to the drug [see Clinical Pharmacology (12.3) ].
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Lactation
Risk Summary
Avoid application of YCANTH topical solution to areas with increased risk for potential ingestion by or ocular exposure to the breastfeeding child. There are no data on the presence of cantharidin in either human or animal milk, or the effects on the breastfed infant or on milk production. Breastfeeding is not expected to result in exposure of the child to the drug due to the low systemic absorption of YCANTH following topical administration [see Clinical Pharmacology (12.3) ]. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for YCANTH topical solution and any potential adverse effects on the breastfeeding child from YCANTH topical solution or from the underlying maternal condition.
Pediatric Use
Risk Summary
The safety and effectiveness of YCANTH for the treatment of molluscum contagiosum have been established in pediatric patients aged 2 years and older. The use of YCANTH in pediatric patients is supported by results from adequate and well-controlled trials in patients 2 years of age and older; although the safety and efficacy of drug use for longer than 12 weeks has not been established [see Clinical Studies (14) ].
The safety and efficacy in pediatric patients below the age of 2 years have not been established .
Geriatric Use
Risk Summary
YCANTH has not been studied in geriatric patients.
CONTRAINDICATIONS
None
WARNINGS AND PRECAUTIONS
- Toxicities Associated with Inappropriate Administration: Life threatening or fatal toxicities can occur if administered orally. Avoid contact with the treatment area, including oral contact, after treatment. Ocular toxicity can occur if YCANTH comes in contact with eyes. If YCANTH gets in eyes, flush eyes with water for at least 15 minutes. (5.1 )
- Local Skin Reactions : Reactions at the application site have included vesiculation, pruritus, pain, discoloration, and erythema. Avoid application near eyes and mucosal tissue, and to healthy skin. If YCANTH contacts any unintended surface, or healthy skin, immediately remove. If severe local skin reactions occur, remove prior to 24 hours after treatment. (5.2 )
- Flammability : YCANTH is flammable, even after drying. Avoid fire, flame or smoking near lesion(s) during treatment and after application until removed. (5.3 )
Toxicities Associated with Inappropriate Administration
YCANTH is for topical use only. YCANTH is not for oral, mucosal, or ophthalmic use.
Life threatening or fatal toxicities can occur if YCANTH is administered orally [see Overdosage (10) ]. Adverse reactions to oral ingestion of cantharidin have included renal failure, blistering and severe damage to the gastrointestinal tract, coagulopathy, seizures, and flaccid paralysis. Advise patients and/or caregivers to avoid oral contact and to avoid touching lesions after YCANTH treatment and to seek medical attention immediately if YCANTH is accidently ingested.
Ocular toxicity can occur if YCANTH comes in contact with the eyes. Adverse reactions from contact of YCANTH with the eyes can include corneal necrosis, ocular perforation, and deep ocular injuries. Do not apply YCANTH near or to the eyes. If YCANTH comes in contact with the eyes, flush eyes with water for at least 15 minutes and seek medical attention immediately.
Local Skin Reactions
YCANTH is a vesicant. Local skin reactions at the application site were observed in 97% of subjects treated with YCANTH during clinical trials. Local skin reactions included vesiculation, pruritus, pain, discoloration, and erythema [see Adverse Reactions (6.1) ] .
Avoid application near the eyes and mucosal tissues, and to adjacent healthy skin. If YCANTH contacts any unintended surface, including healthy skin, immediately remove by wiping with a cotton swab or gauze .
Avoid other topical products (e.g. creams, lotions, or sunscreen) on treated areas until 24 hours after YCANTH treatment or until washing. Application of other topical products could spread YCANTH and cause blistering or other adverse reactions to healthy skin.
If severe blistering, severe pain or other severe adverse reactions occur, remove YCANTH prior to the recommended 24 hours after administration by washing with soap and water .
Flammability
YCANTH is a flammable liquid, even after drying. Avoid fire, flame or smoking near lesion(s) during treatment and after application until removed.
ADVERSE REACTIONS
The following adverse reactions are described elsewhere in the labeling:
- Local Skin Reactions [see Warnings and Precautions (5.2) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
YCANTH was studied in two randomized, double-blind, placebo-controlled phase 3 trials, Trial 1 (NCT03377790) and Trial 2 (NCT03377803) (n = 266, and n = 262, respectively) in subjects with molluscum contagiosum. Most patients received a single 24-hour dermal administration of YCANTH or vehicle for each lesion every 3 weeks for up to 4 treatments. YCANTH Solution or vehicle were removed prior to the 24-hour timepoint in 109/311 (35%) subjects treated with YCANTH Solution and 46/216 (21%) subjects treated with vehicle due to treatment-emergent adverse events.
Table 1 presents the percentage of subjects with selected adverse reactions (incidence ≥ 1%) by the most severe grade reported during Trial 1 and Trial 2. Adverse reactions were primarily local skin reactions at the application site. Local skin reactions at the application site were observed in 97% of subjects treated with YCANTH during both trials.
| YCANTH N=311 | Vehicle N=216 | |||||
|---|---|---|---|---|---|---|
| Preferred Term Name | Mild | Moderate | Severe | Mild | Moderate | Severe |
| Application site vesicles | 60% | 32% | 4% | 27% | 2% | 0% |
| Application site pain and pain | 41% | 20% | 2% | 16% | 1% | 0% |
| Application site pruritus and pruritus | 47% | 8% | 1% | 30% | 7% | 0% |
| Application site scab and scab | 39% | 9% | 0% | 20% | 1% | 0% |
| Application site erythema and erythema | 24% | 21% | <1% | 20% | 7% | 0% |
| Application site discoloration | 28% | 4% | <1% | 12% | 1% | 0% |
| Application site dryness | 19% | 2% | 0% | 14% | 1% | 0% |
| Application site edema | 7% | 3% | 0% | 3% | 1% | 0% |
| Application site erosion | 6% | 1% | 0% | 1% | 0% | 0% |
| Contact dermatitis | 0% | 1% | 0% | 0% | 0% | 0% |
There were no serious adverse reactions reported in the two controlled trials.
The discontinuation rate due to an adverse reaction was 2.3% among subjects treated with YCANTH and 0.5% among subjects treated with vehicle.
DESCRIPTION
YCANTH (cantharidin) topical solution is a light violet to dark purple, slightly viscous liquid for topical administration.
Each mL of YCANTH topical solution contains 7 mg of active ingredient cantharidin (0.7%), a lipophilic compound. Cantharidin is a white to off-white solid at room temperature and is only very slightly soluble in water.
The chemical name for cantharidin is 1,2-dimethyl-3,6-epoxyperhydrophthalic anhydride. The molecular weight is 196.20 g/mol and the molecular formula is C 10 H 12 O 4 . The structural formula for cantharidin is represented below:
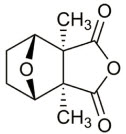
The inactive ingredients of YCANTH topical solution are acetone, camphor, castor oil, denatonium benzoate, ethanol (32%), gentian violet, hydroxypropyl cellulose, and nitrocellulose. The inactive ingredient denatonium benzoate is an oral deterrent to help mitigate the risk of accidental ingestion.
CLINICAL PHARMACOLOGY
Mechanism of Action
The mechanism of action of cantharidin in the treatment of molluscum contagiosum is unknown.
Pharmacodynamics
Cantharidin is a vesicant. The pharmacodynamics of cantharidin in the treatment of molluscum contagiosum are unknown.
Pharmacokinetics
Pharmacokinetics of cantharidin were assessed in 16 subjects aged 2 years and older with at least 21 molluscum contagiosum lesions at baseline. Subjects were administered a single, dermal application of cantharidin solution to each lesion. The average number of lesions treated for subjects in the exposure treatment group was 47.4 (median was 35.0).
Cantharidin was not detectable in 15 out of 16 subjects. Only one subject had a detectable cantharidin plasma level of 3.391 ng/mL at the 2-hour post-dose.
Drug Interaction Studies
No studies evaluating the drug interaction potential of cantharidin have been conducted.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with cantharidin.
Cantharidin was not mutagenic in a bacteria reverse mutation (Ames) assay. An in vitro chromosomal aberration assay in human lymphocytes with cantharidin was inconclusive. Cantharidin was positive in an in vitro micronucleus assay in TK6 cells, primarily through an aneugenic mechanism.
The effects of cantharidin on fertility have not been evaluated.
CLINICAL STUDIES
In two double-blind, randomized, placebo-controlled trials (Trial 1 [NCT03377790] and Trial 2 [NCT03377803]), 528 subjects ages 2 years and older with molluscum contagiosum were randomized by household to treatment with either YCANTH or vehicle. Subjects ranged from 2 to 60 years of age, with a median age of 6. 51% of subjects were male and 91% were Caucasian. The baseline lesion count among subjects ranged from 1 to 184, with a mean of 23 and 19 lesions among subjects in Trial 1 and Trial 2, respectively.
Subjects' lesions were treated with either YCANTH or vehicle at intervals of approximately 21 days until complete clearance of the lesion or for a maximum of 4 applications (on Days 1, 21, 42, and 63). Study drug solution was applied and left on the lesions for approximately 24 hours before the lesions were washed with soap and water.
A healthcare professional who was blinded to the treatment group counted the number of lesions at each visit. The primary efficacy endpoint was the proportion of patients achieving complete clearance of all treated molluscum contagiosum lesions by Day 84. The secondary efficacy endpoints were the proportions of patients achieving complete clearance of all treated molluscum contagiosum lesions at Day 63, Day 42, and Day 21. Table 2 presents the efficacy results for Trial 1 and Trial 2.
| Trial 1 | Trial 2 | |||||
|---|---|---|---|---|---|---|
| YCANTH N = 160 | Vehicle N = 106 | Treatment Difference (95% CI) Treatment difference and 95% CI based on Generalized Estimating Equations (GEE) model for logistic regression with an exchangeable working correlation structure, a factor for treatment, and repeated measurements allowed for a household. Subjects with missing data are imputed as non-responders. | YCANTH N = 150 | Vehicle N = 112 | Treatment Difference (95% CI) | |
| CI = confidence interval. | ||||||
| Day 84 | 46% | 18% | 29% (19%, 38%) | 54% | 13% | 40% (30%, 51%) |
| Day 63 | 32% | 17% | 15% (4%, 25%) | 28% | 5% | 23% (15%, 32%) |
| Day 42 | 21% | 9% | 10% (2%, 19%) | 13% | 4% | 9% (3%, 16%) |
| Day 21 | 11% | 4% | 8% (2%, 14%) | 5% | 2% | 3% (-1%, 8%) |
HOW SUPPLIED/ STORAGE AND HANDLING
How Supplied
YCANTH topical solution is supplied in a sealed glass ampule contained within a single-use applicator and enclosed in a protective paperboard sleeve. Each ampule of YCANTH contains approximately 0.45 mL of 0.7% cantharidin solution. Each mL of YCANTH contains 7 mg cantharidin (0.7%). A YCANTH Break Tool is co-packaged as 2 units per each 6 count and 12 count carton of applicators and 1 unit per each 1 count carton with 1 applicator.
A listing of the available carton packages is provided in Table 3.
| Dosage Strength | Single-Use Applicator | Single Applicator NDC (Each) | Number of YCANTH Applicators (Each) per Carton | YCANTH Break Tool Part Reference Number | Number of YCANTH Break Tools (Each) per Carton | NDC |
|---|---|---|---|---|---|---|
| 0.7% (7 mg/mL) | 1 each | 71349-070-01 | 1 | 71349-000-01 | 1 | 71349-070-11 |
| 6 | 71349-000-01 | 2 | 71349-070-06 | |||
| 12 | 71349-000-01 | 2 | 71349-070-12 |
Individual YCANTH Break Tools are available separately (Part Reference Number 71349-000-01).
To request additional YCANTH Break Tools, contact Verrica Pharmaceuticals at 1-973-298-1390.
Storage and Handling
Store at 20°C to 25°C (68° to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature], protect from light.
Mechanism of Action
The mechanism of action of cantharidin in the treatment of molluscum contagiosum is unknown.