Get your patient on Zilbrysq - Zilucoplan injection, Solution (Zilucoplan)
Zilbrysq - Zilucoplan injection, Solution prescribing information
WARNING: SERIOUS MENINGOCOCCAL INFECTIONS
ZILBRYSQ, a complement inhibitor, increases the risk of serious infections caused by Neisseria meningitidis [see Warnings and Precautions (5.1) ] . Life-threatening and fatal meningococcal infections have occurred in patients treated with complement inhibitors. These infections may become rapidly life-threatening or fatal if not recognized and treated early.
- Complete or update vaccination for meningococcal bacteria (for serogroups A, C, W, Y, and B) at least 2 weeks prior to the first dose of ZILBRYSQ, unless the risks of delaying therapy outweigh the risk of developing a serious infection. Comply with the most current Advisory Committee on Immunization Practices (ACIP) recommendations for vaccination against meningococcal bacteria in patients receiving a complement inhibitor. See Warnings and Precautions (5.1) for additional guidance on the management of the risk of serious infections caused by meningococcal bacteria.
- Patients receiving ZILBRYSQ are at increased risk for invasive disease caused by Neisseria meningitidis , even if they develop antibodies following vaccination. Monitor patients for early signs and symptoms of serious meningococcal infections and evaluate immediately if infection is suspected.
Because of the risk of serious meningococcal infections, ZILBRYSQ is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called ZILBRYSQ REMS [see Warnings and Precautions (5.2) ] .
INDICATIONS AND USAGE
ZILBRYSQ is indicated for the treatment of generalized myasthenia gravis (gMG) in adult patients who are anti-acetylcholine receptor (AChR) antibody positive.
DOSAGE AND ADMINISTRATION
- Obtain baseline amylase and lipase. (2.2 )
- For subcutaneous injection only. (2.3 )
- Recommended dosage (2.3 ):
Body Weight Once Daily Dosage Plunger Rod Color of Prefilled Syringe Less than 56 kg 16.6 mg RUBINE RED 56 kg to less than 77 kg 23 mg ORANGE 77 kg and above 32.4 mg DARK BLUE - See Full Prescribing Information for instructions on dosage, preparation, and administration. (2.4 , 2.5 )
Recommended Vaccination and Prophylaxis for Meningococcal Infection
Vaccinate patients against meningococcal infection (serogroups A, C, W, Y, and B) according to current ACIP recommendations at least 2 weeks prior to initiation of ZILBRYSQ [see Warnings and Precautions (5.1) ] .
If urgent ZILBRYSQ therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer these vaccines as soon as possible [see Warnings and Precautions (5.1) ] .
Healthcare providers who prescribe ZILBRYSQ must enroll in the ZILBRYSQ REMS [see Warnings and Precautions (5.2) ] .
Recommended Testing Before Initiating ZILBRYSQ
Before initiating ZILBRYSQ, obtain baseline lipase and amylase levels [see Warnings and Precautions (5.4) ] .
Recommended Dosage
The recommended dosage of ZILBRYSQ is given once daily as a subcutaneous injection and is dependent on actual body weight (see Table 1 ).
| Body Weight | Once Daily Dosage | Plunger Rod Color of Prefilled Syringe |
|---|---|---|
| Less than 56 kg | 16.6 mg | RUBINE RED |
| 56 kg to less than 77 kg | 23 mg | ORANGE |
| 77 kg and above | 32.4 mg | DARK BLUE |
Preparation Instructions
- ZILBRYSQ prefilled syringes can be stored in a refrigerator in the original carton. ZILBRYSQ can also be stored at room temperature in the original carton for up to 3 months or until the expiration date, whichever occurs first. If stored in the refrigerator:
- Before injecting, take 1 ZILBRYSQ prefilled syringe out of the refrigerator and place it on a clean, flat surface. Allow ZILBRYSQ to reach room temperature out of direct sunlight (30 to 45 minutes). Do not heat or place in microwave.
- Immediately return the carton with the other prefilled syringes to the refrigerator.
- Remove 1 ZILBRYSQ prefilled syringe from the carton.
- Do not return ZILBRYSQ to the refrigerator after it has been stored at room temperature.
- Visually inspect ZILBRYSQ for particulate matter and discoloration prior to administration. ZILBRYSQ is a clear to slightly opalescent, colorless solution. Do not use if the solution contains visible particles, is cloudy, or if foreign particulate matter is present. ZILBRYSQ does not contain preservatives; unused portions of drug remaining in the syringe should be discarded.
- Each prefilled syringe is single-dose only.
Administration Instructions
- ZILBRYSQ is intended for use under the guidance and supervision of a healthcare professional. Patients may self-inject ZILBRYSQ after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of ZILBRYSQ according to the "Instructions for Use" [see Instructions for Use ] .
- Administer ZILBRYSQ subcutaneously into areas of the abdomen, thighs, or back of the upper arms that are not tender, bruised, red, or hard. Avoid injecting into areas with scars or stretch marks. Rotate injection sites for each administration. Administration of ZILBRYSQ in the upper, outer arm should be performed by a caregiver.
- When using ZILBRYSQ prefilled syringes, inject the full contents of the single-dose prefilled syringe.
- Discard ZILBRYSQ prefilled syringe after use. Do not reuse.
- Instruct the patient that the daily dose should be administered at approximately the same time each day.
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time. Do not administer more than 1 dose per day.
DOSAGE FORMS AND STRENGTHS
Injection: 16.6 mg/0.416 mL, 23 mg/0.574 mL, or 32.4 mg/0.81 mL of zilucoplan as a clear to slightly opalescent, colorless solution in single-dose prefilled syringes.
USE IN SPECIFIC POPULATIONS
Pregnancy: Based on animal data, may cause fetal harm. (8.1 )
Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ZILBRYSQ during pregnancy. Patients or healthcare providers may contact UCBCares at 1-844-599-CARE (2273) or email ucbcares@ucb.com, so that information about the exposure of ZILBRYSQ during pregnancy and/or breastfeeding can be collected.
Risk Summary
There are no available data on ZILBRYSQ use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Administration of zilucoplan to pregnant monkeys resulted in increases in embryofetal death at maternal exposures similar to those in humans at therapeutic doses (see Animal Data ) .
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The background rate of major birth defects and miscarriage in the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Subcutaneous administration of zilucoplan (0, 1, 2, or 4 mg/kg/day) to pregnant monkeys throughout gestation resulted in an increase in embryofetal death at all doses, in the absence of maternal toxicity. A no effect dose for adverse developmental effects in monkeys was not identified. The lowest dose tested was associated with maternal exposures (AUC) similar to that in humans at the maximum recommended human dose of 32.4 mg/day.
Data from an ex vivo human placental transfer model demonstrated transfer of zilucoplan into the fetal compartment at a rate of 0.5% at a steady state plasma concentration of 10 µg/mL zilucoplan, which corresponds to a therapeutic dose of 0.3 mg/kg. The clinical significance of these data in human pregnancies is unknown.
Lactation
Risk Summary
There are no data on the presence of zilucoplan in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ZILBRYSQ and any potential adverse effects on the breastfed infant from ZILBRYSQ or from the underlying maternal condition.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatric Use
Clinical studies of ZILBRYSQ did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger adult patients.
CONTRAINDICATIONS
ZILBRYSQ is contraindicated for initiation in patients with unresolved serious Neisseria meningitidis infection [see Warnings and Precautions (5.1) ] .
WARNINGS AND PRECAUTIONS
- Other Infections: Use caution when administering ZILBRYSQ to patients with any other systemic infection. (5.3 )
- Pancreatitis and pancreatic cysts have been reported in patients treated with ZILBRYSQ. Discontinue ZILBRYSQ in patients with suspected pancreatitis and initiate appropriate management until pancreatitis is ruled out or has resolved. (5.4 )
Serious Meningococcal Infections
ZILBRYSQ, a complement inhibitor, increases a patient's susceptibility to serious, life-threatening, or fatal infections caused by meningococcal bacteria (septicemia and/or meningitis) in any serogroup, including non-groupable strains. Life-threatening and fatal meningococcal infections have occurred in both vaccinated and unvaccinated patients treated with complement inhibitors. The initiation of ZILBRYSQ treatment is contraindicated in patients with unresolved serious Neisseria meningitidis infection.
Complete or update meningococcal vaccination (for serogroups A, C, W, Y and B) at least 2 weeks prior to administration of the first dose of ZILBRYSQ, according to current ACIP recommendations for patients receiving a complement inhibitor. Revaccinate patients in accordance with ACIP recommendations considering the duration of ZILBRYSQ therapy. Note that ACIP recommends an administration schedule in patients receiving complement inhibitors that differs from the administration schedule in the vaccine prescribing information.
If urgent ZILBRYSQ therapy is indicated in a patient who is not up to date with meningococcal vaccines according to ACIP recommendations, provide the patient with antibacterial drug prophylaxis and administer meningococcal vaccines as soon as possible. Various durations and regimens of antibacterial drug prophylaxis have been considered, but the optimal durations and drug regimens for prophylaxis and their efficacy have not been studied in unvaccinated or vaccinated patients receiving complement inhibitors, including ZILBRYSQ. The benefits and risks of treatment with ZILBRYSQ, as well as the benefits and risks of antibacterial drug prophylaxis in unvaccinated or vaccinated patients, must be considered against the known risks for serious infections caused by Neisseria meningitidis .
Vaccination does not eliminate the risk of meningococcal infections, despite development of antibodies following vaccination.
Closely monitor patients for early signs and symptoms of meningococcal infection and evaluate patients immediately if infection is suspected. Inform patients of these signs and symptoms and instruct patients to seek immediate medical care if these signs and symptoms occur. Promptly treat known infections. Meningococcal infection may become rapidly life-threatening or fatal if not recognized and treated early. Consider interruption of ZILBRYSQ in patients who are undergoing treatment for serious meningococcal infection, depending on the risks of interrupting treatment in the disease being treated [see Contraindications (4) ] .
ZILBRYSQ is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) [see Warnings and Precautions (5.2) ] .
ZILBRYSQ REMS
ZILBRYSQ is available only through a restricted program under a REMS called ZILBRYSQ REMS, because of the risk of serious meningococcal infections [see Warnings and Precautions (5.1) ] .
Notable requirements of the ZILBRYSQ REMS include the following:
- Prescribers must enroll in the REMS.
- Prescribers must counsel patients about the risk of serious meningococcal infection.
- Prescribers must provide the patients with the REMS educational materials.
- Prescribers must assess patient vaccination status for meningococcal vaccines (against serogroups A, C, W, Y, and B) and vaccinate if needed according to current ACIP recommendations two weeks prior to the first dose of ZILBRYSQ.
- Prescribers must provide a prescription for antibacterial drug prophylaxis if treatment must be started urgently and the patient is not up to date with meningococcal vaccines according to current ACIP recommendations at least two weeks prior to the first dose of ZILBRYSQ.
- Pharmacies that dispense ZILBRYSQ must be certified in the REMS and must verify prescribers are certified.
- Patients must receive counseling from the prescriber about the need to receive meningococcal vaccines per ACIP recommendations, the need to take antibiotics as directed by the prescriber, and the signs and symptoms of meningococcal infection.
- Patients must be instructed to carry the Patient Safety Card with them at all times during and for 2 months following treatment discontinuation with ZILBRYSQ.
Further information is available at www.ZILBRYSQREMS.com or 1-877-414-8353.
Other Infections
Serious infections with Neisseria species (other than Neisseria meningitidis ), including disseminated gonococcal infections, have been reported in patients treated with complement inhibitors.
ZILBRYSQ blocks terminal complement activation; therefore, patients may have increased susceptibility to infections, especially with encapsulated bacteria, such as infections caused by Neisseria meningitidis but also Streptococcus pneumoniae , Haemophilus influenzae , and to a lesser extent, Neisseria gonorrhoeae . Administer vaccinations for the prevention of Streptococcus pneumoniae infection according to ACIP recommendations. Patients receiving ZILBRYSQ are at increased risk for infections due to these organisms, even if they develop antibodies following vaccination.
Pancreatitis and Other Pancreatic Conditions
Pancreatitis and pancreatic cysts have been reported in patients treated with ZILBRYSQ.
During the open-label extension studies, seven (3.3%) patients experienced pancreatic events, including 4 (1.9%) patients with pancreatitis and 3 (1.4%) with pancreatic cysts.
In the 3-month, double-blind Study 1, adverse reactions of increased lipase were reported in six (6.9%) patients treated with ZILBRYSQ compared to no patients on placebo, and adverse reactions of increased amylase were reported in four (4.7%) patients treated with ZILBRYSQ compared to one (1.1%) patient on placebo. Lipase levels exceeded three times the upper limit of normal in six (7%) patients after being started on ZILBRYSQ compared to no patients on placebo.
Patients should be informed of this risk before starting ZILBRYSQ. Obtain lipase and amylase levels at baseline before starting treatment with ZILBRYSQ. Discontinue ZILBRYSQ in patients with suspected pancreatitis and initiate appropriate management until pancreatitis is ruled out or has resolved.
ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Serious Meningococcal Infections [see Warnings and Precautions (5.1) ]
- Other Infections [see Warnings and Precautions (5.3) ]
- Pancreatitis and Other Pancreatic Conditions [see Warnings and Precautions (5.4) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 212 patients were treated with ZILBRYSQ 0.3 mg/kg in clinical studies in gMG. Of these, 137 patients were exposed for at least 6 months, and 87 were exposed for at least 1 year.
In a placebo-controlled study (Study 1) in patients with gMG, 86 patients received ZILBRYSQ 0.3 mg/kg [see Clinical Studies (14) ] . Of these 86 patients, approximately 61% were female, 77% were White, 8% were Asian, and 8% were of Hispanic or Latino ethnicity. The mean age at study entry was 52.6 years (range 21 to 75 years). Table 2 summarizes the adverse reactions reported in at least 5% of patients treated with ZILBRYSQ and more frequently than placebo. The most common adverse reactions (reported in at least 10% of patients treated with ZILBRYSQ) were injection site reactions, upper respiratory tract infections, and diarrhea.
| Adverse Reaction | ZILBRYSQ 0.3 mg/kg (n=86) % | Placebo (n=88) % |
|---|---|---|
| Injection site reactions | 29 | 16 |
| Upper respiratory tract infections | 14 | 7 |
| Diarrhea | 11 | 2 |
| Urinary tract infection | 8 | 5 |
| Nausea or vomiting | 8 | 7 |
| Lipase increased | 7 | 0 |
| Amylase increased | 5 | 1 |
Pancreatic Events
In addition to increases in amylase and lipase observed in Study 1, pancreatic events, including pancreatitis and pancreatic cysts have been observed in patients taking ZILBRYSQ [see Warnings and Precautions (5.4) ] .
Adverse Laboratory Changes in Clinical Trials
Additional laboratory abnormalities included transient elevations of blood eosinophils, which were of uncertain clinical significance.
Postmarketing Experience
Adverse Reactions from Observational Studies
Morphea
In the open-label extension studies, which included 213 patients, morphea was observed in 10 (5%) patients; most cases had a time to onset longer than one year after start of treatment and were mild to moderate in severity. One patient discontinued ZILBRYSQ because of morphea.
DESCRIPTION
Zilucoplan, a complement inhibitor, is a 15 amino-acid, synthetic macrocyclic peptide.
The molecular formula of zilucoplan is C 172 H 278 N 24 O 55 in free acid form and its molecular weight is 3562.23 Daltons (free acid form).
The chemical name for zilucoplan sodium is: acetyl‐[L-lysyl 1 -L-valyl 2 -L-glutamyl 3 -L-arginyl 4 -L phenylalanyl 5 -L aspartyl 6 ]- N -methyl L-aspartyl 7 -L tert-leucyl 8 -L tyrosyl 9 -L-7-azatryptophyl 10 -L glutamyl 11 -L-tyrosyl 12 -L prolyl 13 -L cyclohexylglycyl 14 -[L-lysyl 15 , N ε -palmitoyl-γ-L-glutamyl-(1-amino-3,6,9,12,15,18,21,24,27,30,33,36,39,42,45,48,51,54,57,60,63,66,69,72-tetracosaoxapentaheptacontan-75-oyl)], cyclic (Lactam 1-6), tetra sodium.
The primary structure for zilucoplan sodium is shown below:

ZILBRYSQ injection is a sterile, clear to slightly opalescent, colorless, preservative-free, buffered solution of zilucoplan (as zilucoplan sodium) for subcutaneous injection, in single-dose prefilled syringes. The solution pH is between 6.5 and 7.5. ZILBRYSQ is supplied in three dose strengths containing 16.6 mg /0.416 mL, 23 mg /0.574 mL, and 32.4 mg/0.81 mL of zilucoplan free acid equivalent to 17 mg, 23.6 mg, and 33.2 mg of zilucoplan sodium, respectively. Additionally, each mL of the solution contains dibasic sodium phosphate, anhydrous (4.11 mg); monobasic sodium phosphate, monohydrate (2.9 mg); sodium chloride (4.42 mg); and water for injection.
CLINICAL PHARMACOLOGY
Mechanism of Action
Zilucoplan binds to the complement protein C5 and inhibits its cleavage to C5a and C5b, preventing the generation of the terminal complement complex, C5b-9.
The precise mechanism by which zilucoplan exerts its therapeutic effect in generalized myasthenia gravis is unknown but is presumed to involve reduction of C5b-9 deposition at the neuromuscular junction.
Pharmacodynamics
In a placebo-controlled Phase 2 study, patients received zilucoplan 0.3 mg/kg or 0.1 mg/kg for 12 weeks. There was a dose dependent inhibition of complement. Complement inhibition as represented by inhibition of sheep red blood cell (sRBC) lysis was 89.1% within 3 hours after the first administration and 94.9% after the 12-week treatment period in patients receiving ZILBRYSQ 0.3 mg/kg.
In Study 1 [see Clinical Studies (14) ], complement inhibition of 97.5% was observed by the end of the first week and sustained throughout the 12-week treatment period for gMG patients treated with the recommended dosage regimen of ZILBRYSQ.
Cardiac Electrophysiology
At a dose two times the maximum approved recommended dose, ZILBRYSQ does not cause clinically significant QTc interval prolongation.
Pharmacokinetics
The population pharmacokinetics (PK) analysis showed that zilucoplan PK was not time dependent. Following single daily subcutaneous administration of ZILBRYSQ (0.05 mg/kg, 0.1 mg/kg, 0.2 mg/kg, and 0.4 mg/kg) in healthy subjects, increase in peak plasma concentration was approximately dose proportional and increase in area under the curve was less than dose proportional.
Absorption
Following single and multiple daily subcutaneous administration of ZILBRYSQ 0.3 mg/kg in healthy subjects, zilucoplan reached peak plasma concentration generally between 3 to 6 hours post-dose. Following daily subcutaneous dosing of ZILBRYSQ 0.3 mg/kg for 14 days in healthy subjects, both the peak plasma concentration and exposure (AUC tau ) increased by approximately 3-fold.
In Study 1 [see Clinical Studies (14) ] , after daily repeated subcutaneous administration of ZILBRYSQ 0.3 mg/kg, plasma concentrations of zilucoplan were consistent, with steady state trough concentrations being reached by four weeks of treatment with ZILBRYSQ through twelve weeks.
Distribution
The mean volume of distribution at steady state was 3.51 L in the population pharmacokinetics analysis for adult patients with gMG. Zilucoplan and its 2 major metabolites are highly bound to plasma proteins (>99%).
Elimination
The mean plasma terminal half-life of zilucoplan was approximately 172 hours (7 to 8 days).
Metabolism
As a peptide, ZILBRYSQ is expected to be degraded into small peptides and amino acids via catabolic pathways.
In plasma, two major metabolites, RA103488 and RA102758, were detected. The metabolite RA103488, formed mainly because of cytochrome CYP450 4F2, has pharmacological activity similar to zilucoplan but is present at a much lower concentration compared to zilucoplan. The metabolite RA102758, formed by protease mediated degradation, is pharmacologically inactive. The AUCs of both metabolites were approximately 10% of the parent AUC. The contribution of RA103488 to pharmacological activity is therefore expected to be low.
Excretion
Excretion of zilucoplan and its metabolite in urine and feces was negligible (<1% of the dose).
Specific Populations
Age, Sex, and Race
A population pharmacokinetics analysis assessing the effects of age, sex, and race did not suggest any clinically significant impact of these covariates on zilucoplan exposures.
Patients with Renal Impairment
A dedicated clinical study compared the pharmacokinetics of a single subcutaneous dose of ZILBRYSQ 0.3 mg/kg in subjects with severe renal impairment (as defined by a creatinine clearance <30 mL/min estimated by Cockcroft-Gault formula) to that of matched healthy subjects with normal renal function. A decrease in zilucoplan exposure (AUC 0-inf ) of 13% was observed. This change in zilucoplan exposures is not expected to be clinically significant. No dose adjustment is required in patients with renal impairment.
Patients with Hepatic Impairment
A dedicated clinical study compared the pharmacokinetics of a single subcutaneous dose of ZILBRYSQ 0.3 mg/kg in subjects with moderate hepatic impairment (as indicated by a Child-Pugh category of moderate [score of 7 to 9]) to that of matched healthy subjects with normal hepatic function. A decrease in zilucoplan exposure (AUC 0-inf ) of 24% was observed. This change in zilucoplan exposures is not expected to be clinically significant. No dose adjustment is required in patients with mild or moderate hepatic impairment. The pharmacokinetics of zilucoplan in patients with severe hepatic impairment have not been studied.
Drug Interactions
Clinical drug interaction studies have not been performed with zilucoplan.
In vitro studies have shown that zilucoplan is not a substrate of major cytochrome P450 (CYP) enzymes (1A2, 2B6, 2C8, 2C9, 2C19, 2D6, and 3A) or transporters (P-gp, BCRP, OATP1B1, and OATP1B3). Based on the results from in vitro drug interaction testing, clinically relevant interactions between zilucoplan and substrates of CYP enzymes (1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 3A, and 4F), uridine diphosphoglucuronosyl transferases (UGTs; 1A1, 1A3, 1A4, 1A6, 1A9, 2B7, and 2B15), and transporters (P-gp, BCRP, OATP1B1, OATP1B3, OAT1, OAT3, MATE1, MATE2-K, OCT1, and OCT2) is unlikely.
Immunogenicity
As with all therapeutic peptides, there is a potential for immunogenicity. The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies or of other products. The sensitivity of the assay is not known although ADA was detectable in the tested samples.
In up to 12 weeks of treatment in Study 1, 2.3% (2/86) of patients treated with ZILBRYSQ developed anti-drug antibodies (ADA). A total of 9.3% (8/86) of ZILBRYSQ treated patients developed anti-polyethylene glycol (anti-PEG) antibodies. Because of the low occurrence of anti-drug antibodies and anti-PEG antibodies, the available data are too limited to make definitive conclusions regarding immunogenicity and its effect on pharmacokinetics, pharmacodynamics, safety, or efficacy of ZILBRYSQ.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Studies to assess the carcinogenic potential of zilucoplan have not been conducted.
Mutagenesis
Zilucoplan was not genotoxic in in vitro (Ames and chromosomal aberration) and in vivo (rat micronucleus) assays.
Impairment of Fertility
Subcutaneous administration of zilucoplan (0, 1, 2, or 4 mg/kg/day) to male monkeys resulted in testicular germ cell depletion/degeneration at all doses at the end of the 13-week dosing period and after the 8-week recovery period, indicating lack of reversibility.
A no-effect dose for testicular germ cell degeneration was not identified. At the lowest dose tested, plasma exposures (AUC) were similar to that in humans at the maximum recommended human dose of 32.4 mg/day.
CLINICAL STUDIES
The efficacy of ZILBRYSQ for the treatment of generalized myasthenia gravis (gMG) in adult patients who are anti-AChR antibody positive was established in a 12-week, multicenter, randomized, double-blind placebo-controlled study (Study 1; NCT04115293).
Study 1 enrolled patients who met the following criteria at screening:
- Myasthenia Gravis Foundation of America (MGFA) clinical classification class II to IV,
- Positive serology for AChR binding autoantibodies,
- MG-Activities of Daily Living (MG-ADL) total score of ≥6,
- Those on MG therapy prior to screening (including acetylcholinesterase (AChE) inhibitors, steroids, or non-steroidal immunosuppressive therapies (NSISTs), either in combination or alone), needed to maintain a stable dose.
A total of 174 patients were enrolled in Study 1 and were randomized 1:1 to receive either ZILBRYSQ 0.3 mg/kg (n=86) or placebo (n=88) once daily by subcutaneous injection. Baseline characteristics were similar between treatment groups. Patients had a mean age of 53.0 years and a mean time since diagnosis of 9 years. Fifty-seven percent of the patients were female, 74% were White, 12% were Asian, 8% were Black, 1% were American Indian or Alaska Native, and 6% did not have race reported. The mean baseline MG-ADL total score was 10.6 (range 6 to 19).
At baseline, approximately 85% of patients in each group received cholinesterase inhibitors, 63% received steroids, and 51% received NSISTs, at stable doses.
The primary efficacy endpoint was a comparison of the change from baseline between treatment groups in the Myasthenia Gravis-Specific Activities of Daily Living scale (MG-ADL) total score after twelve weeks of treatment. The MG-ADL assesses the impact of gMG on daily functions of 8 signs or symptoms that are typically affected in gMG. Each item is assessed on a 4-point scale where a score of 0 represents normal function and a score of 3 represents loss of ability to perform that function. A total score ranges from 0 to 24, with the higher scores indicating more impairment.
The efficacy of ZILBRYSQ was also measured using the Quantitative Myasthenia Gravis (QMG) total score which is a 13-item categorical grading system that assesses muscle weakness. Each item is assessed on a 4-point scale where a score of 0 represents no weakness and a score of 3 represents severe weakness. A total possible score ranges from 0 to 39, where higher scores indicate more severe impairment.
Other secondary endpoints included the proportion of patients with improvements of at least 3 and 5 points in the MG-ADL total score and QMG total score, respectively, at week 12 without rescue therapy.
At week 12, treatment with ZILBRYSQ demonstrated a statistically significant improvement from baseline compared to placebo for MG-ADL total score and QMG total score (Table 3; Figure 1).
| Efficacy Endpoints: LS Mean (95% CI) | ZILBRYSQ (n = 86) | Placebo (n = 88) | ZILBRYSQ change LS mean difference vs. placebo (95% CI) | p-value• |
|---|---|---|---|---|
| Abbreviations: CI = confidence interval; MG-ADL, myasthenia gravis activities of daily living scale; QMG, quantitative myasthenia gravis; LS = least square | ||||
| MG-ADL Total Score | -4.39 (-5.28, -3.50) | -2.30 (-3.17, -1.43) | -2.09 (-3.24, -0.95) | < 0.001 |
| QMG Total Score | -6.19 (-7.29, -5.08) | -3.25 (-4.32, -2.17) | -2.94 (-4.39, -1.49) | < 0.001 |
| •Analysis based on MMRM ANCOVA model |
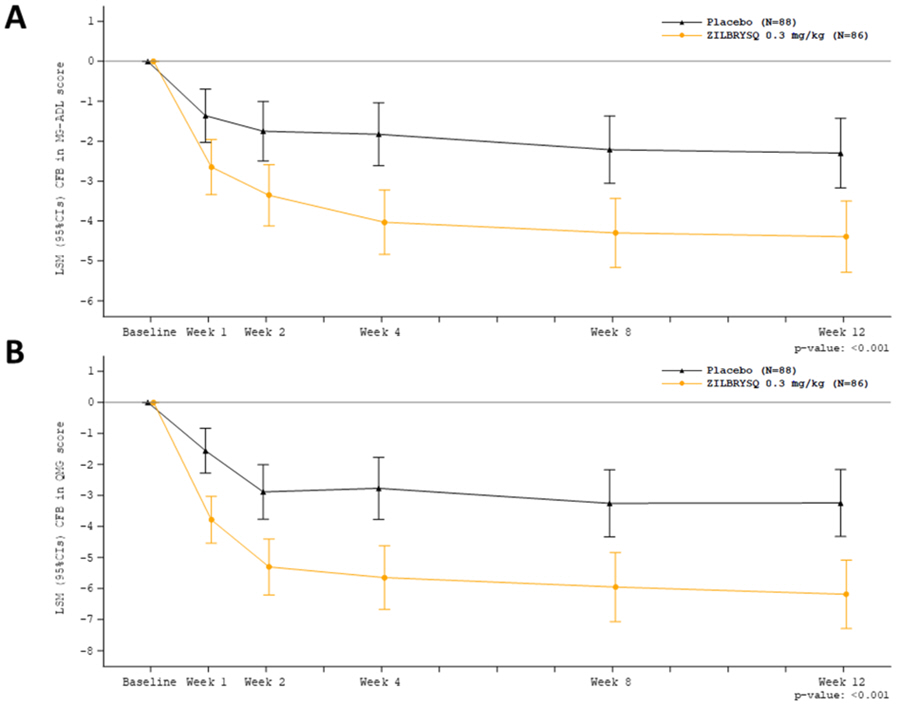 |
The proportion of MG-ADL responders with at least a 3-point improvement at week 12 was greater for ZILBRYSQ (73.1%) compared to placebo (46.1%) (p<0.001). The proportion of QMG responders with at least a 5-point improvement was also greater for ZILBRYSQ (58%) compared to placebo (33%) (p = 0.0012). The proportion of clinical responders at higher response thresholds was consistently greater for ZILBRYSQ compared to placebo.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
ZILBRYSQ (zilucoplan) injection prefilled syringe contains a sterile, preservative-free, clear to slightly opalescent, colorless solution. Each single-dose prefilled syringe consists of a 1 mL glass syringe with a 29-gauge ½ inch needle, a needle safety guard, and a needle cover. The ZILBRYSQ prefilled syringe components are not made with natural rubber latex.
ZILBRYSQ is available as follows:
| NDC | Carton Pack Size | Strength | Plunger Color |
|---|---|---|---|
| 50474-990-80 | 28 single-dose prefilled syringes (4 cartons each containing 7 syringes for a total of 28 syringes) | 16.6 mg/0.416 mL | RUBINE RED |
| 50474-991-80 | 28 single-dose prefilled syringes (4 cartons each containing 7 syringes for a total of 28 syringes) | 23 mg/0.574 mL | ORANGE |
| 50474-992-80 | 28 single-dose prefilled syringes (4 cartons each containing 7 syringes for a total of 28 syringes) | 32.4 mg/0.81 mL | DARK BLUE |
Storage and Handling
Pharmacy Prior to Dispensing
Store ZILBRYSQ prefilled syringes refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton until dispensing. Do not freeze.
Storage for Patients or Caregivers After Dispensing
Storage conditions after dispensing by pharmacist are summarized in Table 4:
| Temperature | Refrigeration 2°C to 8°C (36°F to 46°F) | Room Temperature Up to 30°C (86°F) |
| Time Period | Until expiration date on the carton | Up to 3 months after removing from refrigerator or until expiration date on the carton, whichever occurs first |
- Do not freeze.
- Store ZILBRYSQ prefilled syringes in the original carton to protect them from light until time of use.
- ZILBRYSQ prefilled syringes may be stored at room temperature in the original carton for a single period of up to 3 months.
- When storing ZILBRYSQ prefilled syringes at room temperature, write the date removed from the refrigerator in the space provided on the carton and discard if not used within 3 months or if the expiration date has passed, whichever occurs first.
- Do not return ZILBRYSQ to the refrigerator after it has been stored at room temperature.
- ZILBRYSQ does not contain a preservative; discard any unused portion.
INSTRUCTIONS FOR USE
ZILBRYSQ ® (ZIL-brisk) (zilucoplan) injection, for subcutaneous use Single-Dose Prefilled Syringe
This Instructions for Use contains information on how to inject ZILBRYSQ.
Understanding Your ZILBRYSQ Prefilled Syringe
Read this Instructions for Use before using the ZILBRYSQ prefilled syringe. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. See the Medication Guide that comes with ZILBRYSQ for important information.
Your healthcare provider should show you how to prepare and inject ZILBRYSQ prefilled syringe properly before you use it for the first time. Do not inject yourself or someone else until you have been shown how to inject ZILBRYSQ correctly.
Keep this Instructions for Use and refer to it as needed until you have used all of the ZILBRYSQ prefilled syringes in the packaging.
For general questions or help, please call your healthcare provider or UCBCares at 1-844-599-CARE (2273).
How Should I Store ZILBRYSQ Prefilled Syringes?
- Store ZILBRYSQ prefilled syringes in a refrigerator between 36°F to 46°F (2°C to 8°C) until the expiration date on the carton. Do not freeze ZILBRYSQ.
- ZILBRYSQ prefilled syringes may be stored at room temperature up to 86°F (30°C) in the original carton for a single period of up to 3 months. Write the date you removed the prefilled syringes from the refrigerator in the space provided on the carton. After ZILBRYSQ prefilled syringes have been stored at room temperature, do not place them back in the refrigerator. Throw them away if not used within 3 months or if the expiration date has passed, whichever occurs first.
- Keep the ZILBRYSQ prefilled syringes in the original carton before use.
- Keep ZILBRYSQ prefilled syringes and all medicines out of the reach of children.
Important Information You Need to Know Before You Inject ZILBRYSQ
- Do not use ZILBRYSQ if the expiration date on the packaging has passed or the carton seals have been broken.
- Do not reuse the ZILBRYSQ prefilled syringe. The prefilled syringe is for 1-time (single use) only. You may get an infection.
- Do not inject ZILBRYSQ more than 1 time per day.
- Do not miss any doses of ZILBRYSQ. If you miss your ZILBRYSQ dose, inject a dose as soon as possible. Then, inject the next dose at your scheduled time. Do not inject more than 1 dose each day.
- Do not use the ZILBRYSQ prefilled syringe if it has been dropped.
- Do not remove the needle cap from the ZILBRYSQ prefilled syringe until you are ready to inject.
- Do not insert the needle into the skin more than 1 time because this may bend or break the needle, causing trauma to the tissue.
- Do not pull back on the ZILBRYSQ prefilled syringe plunger head at any time because this can break the prefilled syringe.
- Do not touch the needle guard activation clips at any time because this can cause the early activation of the needle guard.
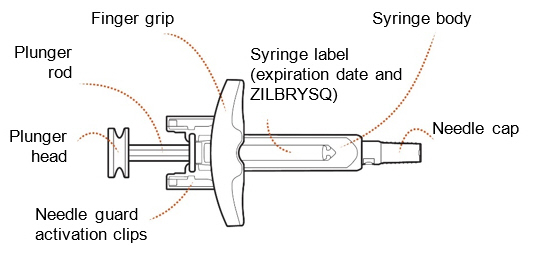
ZILBRYSQ Prefilled Syringe Guide to Parts (Figure A):
| Before use | |||
| |||
| After use | |||
| |||
| Figure A | |||
Preparing for Injection
Step 1: Take out the ZILBRYSQ prefilled syringe
If the ZILBRYSQ prefilled syringes are stored in the refrigerator:
- Take the ZILBRYSQ prefilled syringes carton out of the refrigerator and remove 1 prefilled syringe from the carton. Grasp the prefilled syringe body. Carefully lift the prefilled syringe straight up out of the tray (Figures B.1 and B.2) . Put the rest of the prefilled syringes in the carton back in the refrigerator.
- Before you inject ZILBRYSQ, let the prefilled syringe warm up to room temperature on a clean flat surface for 30 to 45 minutes . This will help to reduce discomfort when injecting.
- Do not warm the ZILBRYSQ prefilled syringe in any other way (for example in a microwave, in hot water, or in direct sunlight).
- Do not remove the needle cap from the prefilled syringe until you are ready to inject.
If the ZILBRYSQ prefilled syringes are stored at room temperature:
- Remove 1 ZILBRYSQ prefilled syringe from the carton. Grasp the prefilled syringe body. Carefully lift the prefilled syringe straight up out of the tray ( Figures B.1 and B.2 ). Any remaining prefilled syringes in the carton should not be placed in the refrigerator after it has been stored at room temperature.
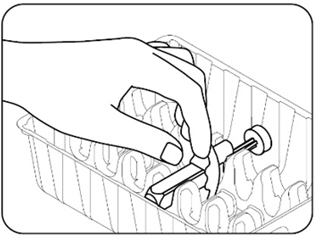 | 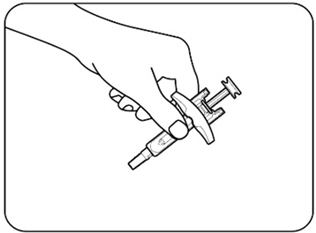 |
| Figure B.1 | Figure B.2 |
Step 2: Inspect the ZILBRYSQ prefilled syringe
- Check the prefilled syringe for damage (Figure C) .
- Do not use if any part of the prefilled syringe appears to be cracked, leaking, or broken.
- Check that the needle cap is not cracked or broken and attached to the prefilled syringe (Figure C) .
- Do not use if the needle cap is missing or not securely attached.
- Check the expiration date and medicine name (ZILBRYSQ) on the prefilled syringe label (Figure C) .
- Do not use if the expiration date printed on the prefilled syringe has passed.
- Do not use if the word ZILBRYSQ does not appear on the prefilled syringe.
- Check the dose appearing on the label (Figure C) and make sure it matches your prescribed dose.
- Do not use if the dose does not match your prescribed dose.
- Check the medicine inside the prefilled syringe (Figure C) . The medicine should be clear to almost clear and colorless. It is normal to see air bubbles in the prefilled syringe.
- Do not use if the medicine is cloudy, discolored, or contains floating particles.
Figure C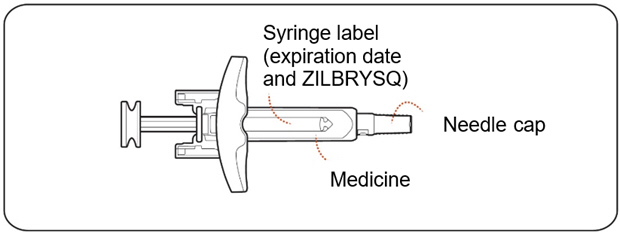
Step 3: Gather supplies
Wash your hands with soap and water and dry them with a clean towel.
Gather the following supplies on a clean, flat surface (Figure D) :
- 1 ZILBRYSQ prefilled syringe
- 1 alcohol wipe (not supplied)
- 1 cotton ball or gauze pad (not supplied)
- 1 adhesive bandage (not supplied)
- 1 sharps disposal or puncture-resistant container (not supplied). See Step 12 for instructions on throwing away the syringe.
Figure D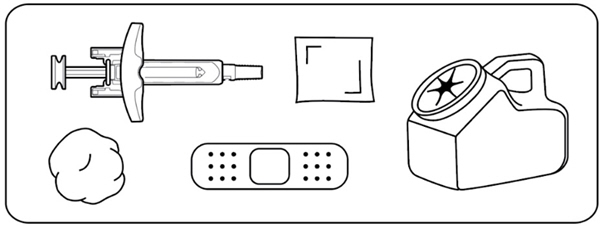
Step 4: Choose your injection site
Choose an injection site from the following areas (Figures E.1 and E.2) :
- The stomach (abdomen), except for the 2-inch area around the belly button (navel) (Figure E.1)
- The front of the thighs (Figure E.1)
Figure E.1 – abdomen and thighs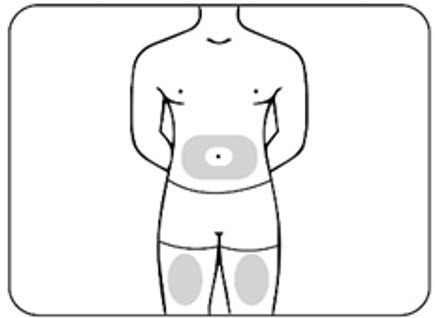
- The back of the upper arms (only if someone else is giving you the injection) (Figure E.2)
Figure E.2 – upper arms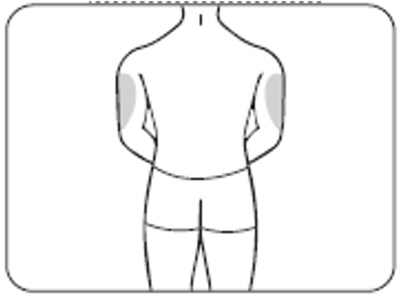
Choose a different site each time you give yourself an injection. If you want to use the same injection site, make sure it is at least 1-inch from a spot you used before.
Do not inject ZILBRYSQ into an area that is red, tender, bruised, swollen, hard or that has scars or stretch marks.
Step 5: Clean your injection site
Clean the injection site using an alcohol wipe (Figure F) .
Let the skin dry for 10 seconds before injecting.
Do not touch the injection site again before giving your injection.
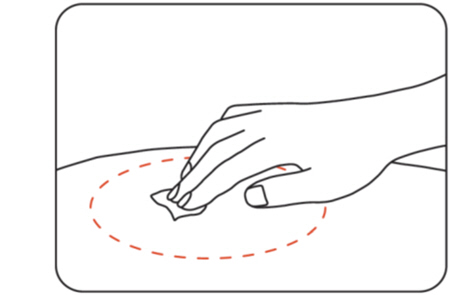 Figure F
Figure F
Injecting the Medicine
Step 6: Remove the needle cap
Hold the body of the ZILBRYSQ prefilled syringe with one hand and pull the needle cap straight off with your other hand (Figure G) .
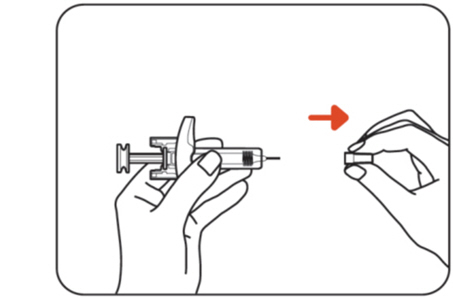 Figure G
Figure G
Throw away (discard) the needle cap into your household trash or a sharps disposal container.
Do not touch the needle or let it touch anything.
Do not recap the needle at any time to avoid injury.
Do not try to remove any air bubbles from the ZILBRYSQ prefilled syringe. Air bubbles will not affect your dose and will not harm you. This is normal. You can continue to take your injection.
Step 7: Pinch your injection site
Use your other hand to pinch the area of cleaned skin and hold it firmly (Figure H) .
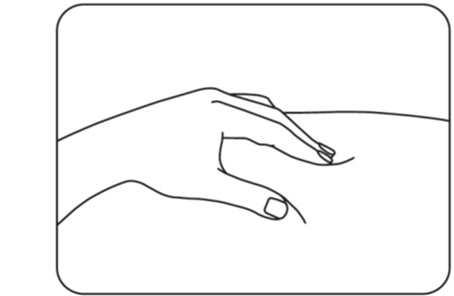 Figure H
Figure H
Step 8: Insert the needle
Insert the entire needle into the pinched skin at a 45° to 90° angle. When the needle is fully inserted, hold the ZILBRYSQ prefilled syringe in place (Figure I) .
Do not pull back on the plunger head at any time because this could break the prefilled syringe.
Do not touch the needle guard activation clips.
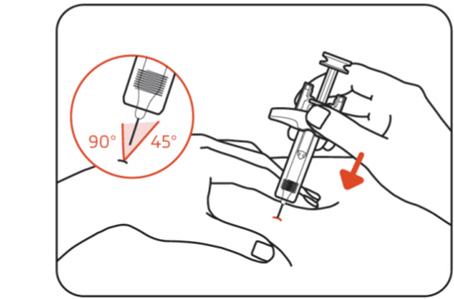 Figure I
Figure I
Step 9: Release the pinched skin
When the needle is fully inserted, hold the ZILBRYSQ prefilled syringe in place and release the pinched skin (Figure J) .
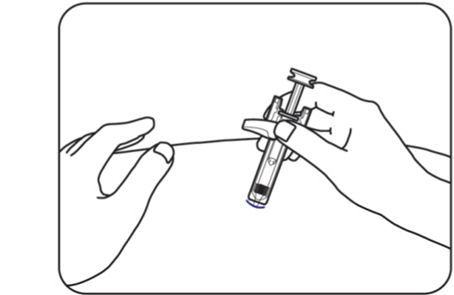 Figure J
Figure J
Do not reinsert the needle into the skin if the needle is pulled out when releasing the skin. If this happens, safely throw away (dispose of) the syringe in a sharps disposal container and get a new ZILBRYSQ prefilled syringe to give the injection.
Step 10: Inject the medicine
Push the plunger head all the way down while holding onto the finger grip to inject all the medicine (Figure K) .
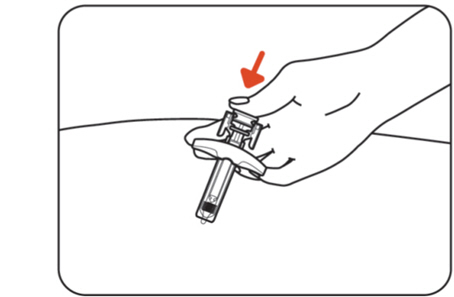 Figure K
Figure K
Step 11: Release the plunger head
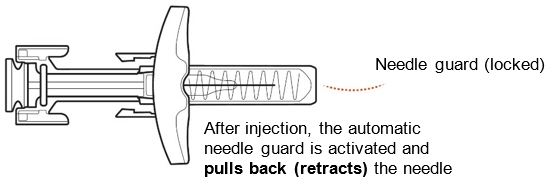
Slowly release the plunger head by lifting your thumb. After a complete injection, the needle guard will cover the needle and you may hear a click (Figure L) .
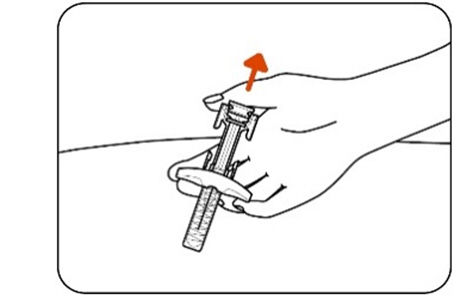 Figure L
Figure L
Step 12: Throw away (dispose of) the used ZILBRYSQ prefilled syringe
Throw away (dispose of) the used ZILBRYSQ prefilled syringe into a sharps disposal container (Figure M) right away.
Do not throw away (dispose of) the ZILBRYSQ prefilled syringe in your household trash.
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labelled to warn of hazardous waste inside the container.
Do not throw away (dispose of) your used sharps disposal container in your household trash unless your community guidelines permit this.
Do not recycle your used sharps disposal container.
Always keep the sharps disposal container out of the reach of children.
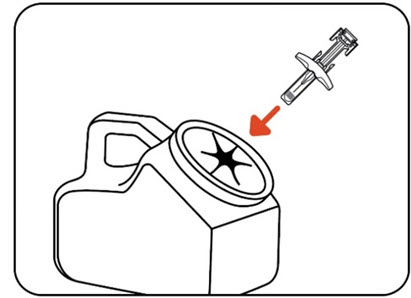 | Note: When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away your used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal. |
| Figure M |
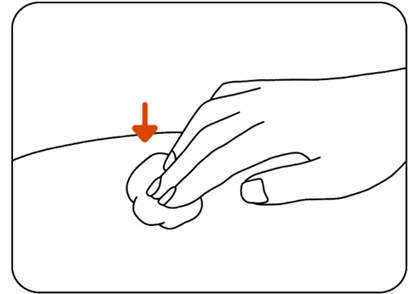
Step 13: Examine the injection site
Press a cotton ball or gauze pad over the injection site and hold it for 10 seconds (Figure N) .
Do not rub the injection site. You may have slight bleeding. This is normal. Apply an adhesive bandage if needed (Figure O).
 | 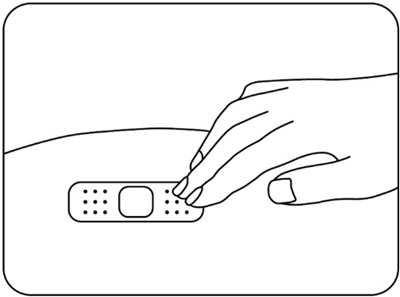 |
| Figure N | Figure O |
The injection is complete.
For more information or help, contact your healthcare provider.
If you have any questions, call UCBCares at 1-844-599-CARE (2273) or visit www.AskUCBCares.com.
Manufactured for: UCB, Inc. Smyrna, GA 30080
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Issued: 10/2023
Mechanism of Action
Zilucoplan binds to the complement protein C5 and inhibits its cleavage to C5a and C5b, preventing the generation of the terminal complement complex, C5b-9.
The precise mechanism by which zilucoplan exerts its therapeutic effect in generalized myasthenia gravis is unknown but is presumed to involve reduction of C5b-9 deposition at the neuromuscular junction.
Zilbrysq - Zilucoplan injection, Solution PubMed™ news
- Journal Article • 2026 MayNipocalimab (Imaavy) for myasthenia gravis.
- Journal Article • 2026 MayMeningococcal prophylaxis in neurological diseases treated with complement inhibitors: an expert consensus for Germany, Austria, and Switzerland.
- Journal Article • 2026 AprCombined inhibition of complement C5 and neonatal Fc receptor in refractory generalized myasthenia gravis: A report of two cases.
- Journal Article • 2026 AprTherapeutic approach to myasthenia gravis treatment in the era of new biologics - review of existing and upcoming therapies.
- Journal Article • 2026 AprEfficacy and safety of complement inhibitors and FcRn blockers in generalized AChR antibody-positive myasthenia gravis: a meta-analysis.